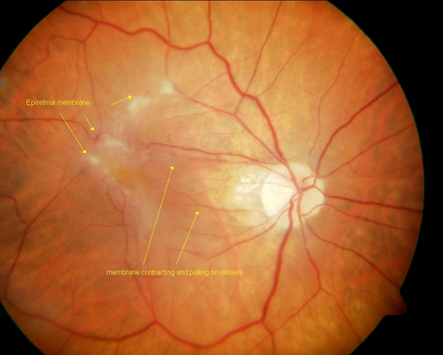
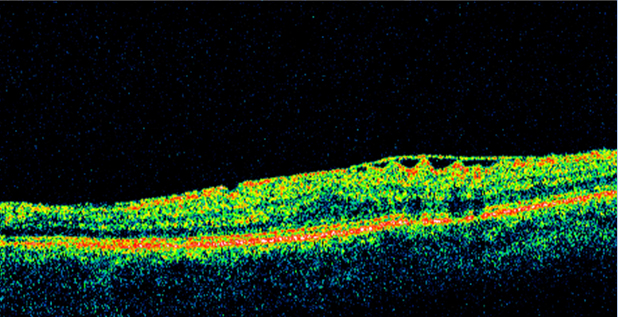
Epiretinal Membrane
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Numerous terms have been used to describe this condition, including epiretinal membrane (ERM), epimacular membrane, surface-wrinkling retinopathy, cellophane maculopathy, and preretinal macular fibrosis.
Disease
An ERM, aka "macular pucker," is pathologic fibrocellular tissue that is created by proliferation of myofibroblastic cells at the vitreoretinal interface. It is semi-translucent and proliferates on the surface of the internal limiting membrane (ILM).
Etiology and Risk Factors
Idiopathic ERM is the most common presentation and is noted in 95% of cases. Secondary ERMs occur in association with retinal vascular diseases (e.g., diabetic retinopathy), retinal vein occlusion, ocular inflammatory disease, trauma, intraocular surgery, intraocular tumors, and retinal tear or detachment. Other risk factors include posterior vitreous detachment and a history of ERM in the fellow eye.
Most patients with ERM are age >50 years; the mean age of ERM diagnosis is around 65 years.[1] The prevalence of ERM is reported to be between 7% and 11%, with as high as 17% in individuals aged >80 years.[2] The incidence rate of developing an ERM in the fellow eye is 2.7% per year.
General Pathology
Retinal glial and retinal pigment epithelial (RPE) cells are the major components of an ERM. Fibrous astrocytes, fibrocytes, myofibrocytes, and macrophages can also be identified on pathologic analysis.
Pathophysiology
It has been hypothesized that residual cortical vitreous secondary to a posterior vitreous detachment or partial separation of the posterior hyaloid can cause dehiscence of the ILM, allowing for migration of microglial cells to the retinal surface in the vitreoretinal space which then differentiate into fibroblast-like cells. When hyalocytes in the vitreous get in contact with microglial cells, they differentiate into myofibroblasts and form the ERM with the microglia-turned fibroblasts.
Inflammatory mediators also promote fibrocellular growth, especially in the setting of secondary ERM formation. Macrophages, RPE cells, and T- and B-cells are usually observed in secondary ERMs.[3]
Primary Prevention
There are no preventative measures for idiopathic ERMs. The risk of secondary ERMs can be reduced in some cases by appropriately managing the underlying cause.
Diagnosis
A clinical diagnosis is based on history and findings from clinical examinations. In some cases, optical coherence tomography (OCT) is useful in the diagnosis, quantification of retinal thickness, and management of ERMs.
Physical Examination
Slit lamp examination with dilated fundus examination and scleral depression (to rule out peripheral breaks/lesions), along with careful examination of the macula, are important for determining the presence of ERMs and evaluating their severity. Paying attention to the vitreous, retinal vasculature, and peripheral retina can provide insight as to the cause of the ERM in secondary cases.
Examination of the fellow eye is also recommended, given that ERMs are bilateral in approximately 10%–20% of patients.
Signs
A sheen or abnormal reflectivity of the macular surface is suggestive of an ERM. More advanced ERMs can become opaque.
Symptoms
Metamorphopsia, blurred vision, monocular diplopia, and micropsia can be noted with any macular pathology. The vast majority of patients with ERMs are asymptomatic.
Clinical Diagnosis
Idiopathic ERMs affect the architecture of the macula. There can be blunting of the foveal contour or wrinkling on the retinal surface from membrane contracture. Most commonly, ERMs involve the foveal and parafoveal area.
Macular edema and/or pseudohole can be seen in association with an ERM. As the name implies, a pseudohole is not a full-thickness macular hole, but rather an opening in the ERM that appears to be a retinal hole. The inner retina around the pseudohole is thickened. The pseudohole may not be exactly round and may have an oval or irregular shape. Other associated signs include vascular tortuosity in the region of the ERM and/or intraretinal hemorrhages.
A posterior vitreous detachment is often noted, which supports the pathophysiology of this entity.
Diagnostic Procedures
High-resolution OCT has become increasingly helpful in the diagnosis and management of ERM as it can allow for both 3-dimensional and cross-section evaluation of the macula. On an OCT image, ERM appears as a hyperreflective linear band on the inner retina. OCT can detect subtle ERMs as well as those associated with macular edema or other macular pathology. Some patients with ERM may have foveoschisis, a separation of the outer nuclear layer from the outer plexiform layer.[4]
OCT can also help guide patient management. Some cases of ERM with vitreomacular traction are subtle clinically and may be more easily detected with OCT. One of the great advantages of OCT is the assessment of the vitreoretinal interface. This can provide additional information regarding therapeutic options and prognosis. In surgical cases, evaluation of each scan can elucidate the best approach for removal. Spectral-domain OCT can help evaluate the outer retinal layers/structure, which may have bearing on the physiologic outcome after an ERM removal.
Fluorescein angiography can be helpful in secondary cases of ERM including retinal vascular occlusions or intraocular tumors. Macular edema can be confirmed with angiography, as well.
Differential Diagnosis
The clinical appearance of an ERM is fairly distinctive. However, macular hole, parafoveal telangiectasia, and macular edema must also be considered.
Management
The most important issue in the management of idiopathic ERMs is the presence of visual complaints. Visual symptoms can be variable and sometimes independent of clinical severity.
Medical Therapy
No medical therapy is indicated for idiopathic ERMs.
Surgery
Surgery is indicated if the patient has significant complaints of visual decline and /or metamorphopsia. Surgery involves pars plana vitrectomy (PPV) with membrane peel and removal of retinal traction. Since the ILM acts as a scaffold for proliferation of myofibroblasts, it may be removed in certain cases, potentially minimizing the risk of ERM recurrence. The goal of PPV should be to remove all ERM fragments.
The advent of intraoperative OCT allows the surgeon to use OCT to pick the best location for beginning the ERM peeling procedure and to check whether there is residual ERM, possibly leading to more complete removal of the ERM. ILM peeling with brilliant blue G dye may be effective in removing all of the ERM fragments.[5] The prospective DISCOVER study evaluated the utility of intraoperative OCT for ERM peeling, with the visual and anatomic results compared retrospectively to a series of ERM peeling without use of intraoperative OCT. The study showed no significant differences in visual acuity or retinal thickness at 12 months; however, at 6 months, central subfield thickness was lower in the intraoperative OCT group than in the conventional peeling group.[6]
Follow-up for ERM surgery is similar to follow-up for other PPV procedures. It is important to note that visual acuity improvement does not occur in some patients due to preoperative characteristics, ERM duration, and other factors. However, most patients improve by 3–6 months postoperatively, and some may experience improvement up to 1–2 years postoperatively.
Complications
Complications are similar those seen in all eyes undergoing PPV, including phakic cataract, retinal break, retinal detachment, and dissociated optic nerve fiber layer. Macular surgery–specific complications include intraoperative macular trauma and light toxicity.
Prognosis
A meta-analysis of 3 studies reporting surgical results following small-incision PPV found mean preoperative and postoperative visual acuity of 20/110 and 20/55, respectively.[7]
The recurrence rate of ERM has been estimated to be 1%.[7]
Additional Resources
- Boyd K. What is a macular pucker? American Academy of Ophthalmology. https://www.aao.org/eye-health/diseases/macular-pucker-list. Published October 31, 2024. Accessed April 16, 2025.
References
- ↑ Fraser-Bell S, Guzowski M, Rochtchina E, et al. Five-year cumulative incidence and progression of epiretinal membranes: the Blue Mountains Eye Study. Ophthalmology. 2003;110(1):34-40.
- ↑ Fung AT, Galvin J, Tran T. Epiretinal membrane: a review. Clin Exp Ophthalmol. 2021;49(3):289-308.
- ↑ Ueki M, Morishita S, Kohmoto R, et al. Comparison of histopathological findings between idiopathic and secondary epiretinal membranes. Int Ophthalmol. 2016;36(5):713-718.
- ↑ Hubschman JP, Govetto A, Spaide RF, et al. Optical coherence tomography-based consensus definition for lamellar macular hole. Br J Ophthalmol. 2020;104(12):1741-1747.
- ↑ Hikichi T, Kubo N, Tabata M. Epiretinal membrane fragments: the origin of recurrent membranes after epiretinal membrane peeling. Can J Ophthalmol. 2023;58(5):472-479.
- ↑ Tuifua TS, Sood AB, Abraham JR, et al. Epiretinal membrane surgery using intraoperative OCT-guided membrane removal in the DISCOVER study versus conventional membrane removal. Ophthalmol Retina. 2021;5(12):1254-1262.
- ↑ 7.0 7.1 Gupta OP, Brown GC, Brown MM. A value-based medicine cost-utility analysis of idiopathic epiretinal membrane surgery. Am J Ophthalmol. 2008;145(5):923-928.