Ocular Trauma: Acute Evaluation, Cataract, Glaucoma
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
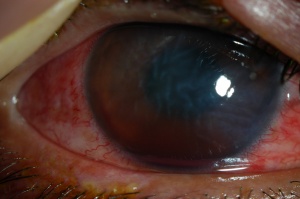
Ocular trauma is one of the most under-recognized causes of vision loss in the developed world. Blunt or penetrating ocular trauma can lead to vision loss through cataract or glaucoma (Figures 1 and 2). Etiologies of ocular injury differ in urban areas compared to other settings, and differ from country to country, between different regions of the world, and between differing demographic or socioeconomic classes. Epidemiological studies can encounter difficulty due to remote trauma and difficulty obtaining an accurate history. In addition, trials to better understand management can be limited by poor follow-up. Strategies for prevention of ocular trauma require knowledge of the cause or mechanism of injury, which may enable more appropriate targeting of resources toward preventing such injuries. Both eye trauma victims and society bear a large, potentially preventable burden.
Disease Entity
Disease
Cataract and glaucoma resulting from ocular trauma can occur in the setting of both penetrating and blunt injuries, thus both open and closed globe injuries.
Risk Factors and prevention
Although there are no specific guidelines for ocular trauma and its sequelae, evaluation and appropriate management at a center familiar with traumatic cataract and its comorbidities can facilitate more successful treatment and avoid complications associated with cataract surgery in these cases. Close follow-up after the inciting event can prevent the sequelae of trauma. In addition, the use of eye protection is critical in preventing eye injuries in many different fields of work.
Diagnosis
History
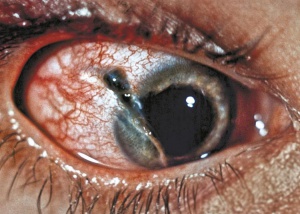
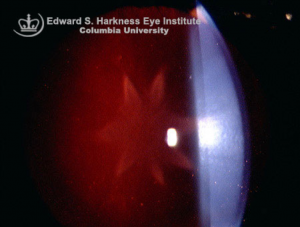
- Time course: Cataract may occur acutely after trauma, but more often a slowly progressive cataract develops. Evolution of the cataract can be monitored. Surgical intervention can typically be postponed until resolution of the typical intraocular inflammation and potentially increased intraocular pressure associated with trauma. Glaucoma, or damage to the optic nerve, can occur as soon as 2-3 weeks following the trauma event, depending upon the intraocular pressure and other factors, such as hyphema (Figure 3), and cyclodialysis, iridodialysis (Figure 4) and other distortions to intraocular structures.
- The development of glaucoma can also occur months to years later in the setting of angle-recession glaucoma.
- On initial exam, the patient’s consciousness level is assessed. It is important to establish mechanism of injury including blunt or penetrating, velocity of impact or projectile, point of impact, material, and any other details relevant to overall energy of impact.
- For example, was the patient attacked by a fist, a large blunt object with high velocity and force or a metal shaving, a small sharp object with high velocity.
- Was the event witnessed?
- Was the patient wearing protective eyewear (if applicable)? Were any visual changes noted soon after the trauma?
- Does the patient use anticoagulant medications (suggesting that a bleed may have formed in the anterior chamber)?
- Is there a personal or family history of sickle cell disease or trait?
- Any ocular history or previous ocular surgeries including implants, scleral buckling, filtration implants, previous wounds in the eye?
- Any other personal medical history or pertinent family history?
- Is the patient’s tetanus vaccination up-to-date?
- In pediatric or elderly patients is their concern for possible abuse?
Symptoms
1. Introduction: General and comprehensive evaluation of an injured patient and an injured eye is very important to save life and sight. Appropriate analgesia, sedation, and protection of open globe from further injury are an important part of the evaluation. Anti-emetics should be administered to avoid Valsalva maneuver if the patient has nausea or vomiting to prevent possible extrusion of intraocular contents.
2. Systemic Evaluation:
- Vital signs should be monitored continuously. Entrapment of an intraocular muscle may lead to life-threatening bradycardia, particularly in children, therefore heart rate should be monitored.
- Brief assessment of other bony and soft tissue injuries must be performed. Once the patient is found to be stable, ophthalmic evaluation can proceed safely.
3. Initial ocular examination The ophthalmologist should have a high index of suspicion for damage to other parts of the eye when traumatic cataract and/or glaucoma are present. Concern for damage to the anterior chamber angle structures, zonular fibers, corneal endothelium, vitreous, and retina should be high. Attention should be paid to the integrity of the lens capsule.
4. Visual Acuity: Important predictor for the ocular trauma score, vision should be tested with correction and pinhole using Snellen’s/ETDRS chart. If vision is lower than this, finger counting may be recorded; if this is not possible, hand motion or light perception may be recorded. No light perception is an important consideration, as eyes with this level of vision may show some improvement following proper exploration and management.
5. Pupillary reaction: Relative afferent pupillary defect is an important negative prognostic factor for affected eye. This finding should raise suspicion for optic nerve damage, avulsion, and compartment syndrome if other signs are present.
6. Intraocular Pressure: IOP may be high, low or normal following injury. Low intraocular pressure or hyoptony should raise caution during exam for ruptured globe, and care should be taken to protect the eye from extrusion of contents. The eye should be shielded when not under examination. Leakage of lens protein from a non-intact lens capsule into the aqueous and/or vitreous, as well as non-inert intraocular foreign body, can elevate intraocular pressure as a result of inflammation. Presence of blood in the anterior chamber (hyphema) can decrease outflow facility of the trabecular meshwork and elevate IOP. Cyclodialysis cleft, ruptured globe, or retinal detachment can cause decreased intraocular pressure. Occult bleeding within the orbit posterior the globe can cause orbital compartment syndrome and elevated IOP. Tight eyelids, difficulty opening the eyelids, proptosis, periorbital edema, and an RAPD are classic signs of orbital compartment syndrome, which is a true ocular emergency.
7. Motility: It is important to assess ocular motility as there are chances of direct or indirect damage to extra ocular muscles. Extraocular muscle entrapment must be ruled out.
8. Field Testing: Central field can be evaluated by the Amsler grid and peripheral field by confrontation method, however, this is at best a gross evaluation and should be followed up by automated perimetry when possible/available.
9. Slit lamp examination:
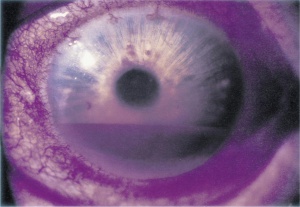
- Corneal endothelial damage will appear as the formation of Descemet membrane folds (Figure 5) and endothelial dysfunction can result in corneal edema.
- The iris is another commonly damaged ocular structure in the setting of ocular trauma. Examples include: sphincter tears, avulsion of the iris root, and bleeding leading to hyphema as this is a vascular structure. The pupil margin should be examined for irregularities.
- Zonular damage can occur after blunt or penetrating trauma to the eye and can be evaluated by tapping the slit lamp stand or asking the patient to glance quickly left-right then straight to observe movements of the phakic lens relative to the pupillary margin. This can be a sign of phacodonesis.
- Evaluation of cataract:
- Total cataract: No clear lens matter visible between the capsule and the nucleus
- Membranous cataract: The capsule and organized matter are fused and a membrane of varying density is formed.
- White soft fluffy cataract: Loose cortical material found in the anterior chamber together with a ruptured lens capsule.
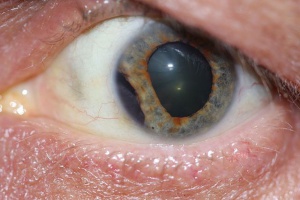
- Rosette cataract: Lens with a rosette pattern of opacity (Figure 6).
- Posterior segment: if a view to the posterior segment is available, the coexistence of retinal detachment, tear, and/or retinal dialysis should be ruled out by scleral depressed examination. This examination should be deferred until the operating room if ruptured globe is suspected even remotely and should not be performed acutely if a ruptured globe is found.
- When a patient is sitting upright at the slit lamp, it may be easier to detect a foreign body than when laying supine under the operating microscope.
10. Indirect ophthalmoscopy: If the ocular media is clear, a posterior segment foreign body can be observed by indirect ophthalmoscopy. Care must be taken to avoid pressure to the periocular tissue and/or globe itself, as if there is an intraocular foreign body, a ruptured globe exists. B-scan can be performed to characterize intraocular foreign bodies, but must be done with care. Computerized tomography is the modality of choice in most Emergency Department settings to characterize size, location and material of intraocular foreign bodies. Magnetic resonance imaging is typically not performed if metallic foreign body is suspected as this could cause movement of the object within the eye and further damage to ocular structures.
11. Gonioscopy: Angle recession will manifest as a tear in the ciliary body between the longitudinal and circular fibers (Figure 7). Acutely, classic gonioscopic findings include brown-colored, broad angle recess, glistening white scleral spur, and depression in overlying trabecular meshwork. Chronically, findings include peripheral anterior synechiae at the border of the recession or anywhere in the angle and damaged iris processes. In the setting of trauma, it is also necessary to assess the presence of a foreign body within the angle, however, gonioscopic examination should typically be deferred acutely in the setting of a ruptured globe or intraocular foreign body as inadvertent indentation pressure can cause extrusion of intraocular contents. CT imaging with 0.5-1 mm cuts through the orbital structures is a preferred modality of detecting an intraocular foreign body acutely, although small foreign bodies could be missed.
Diagnostic Procedures
Imaging
- Plain X ray: Plain X-ray AP and lateral views are essential to evaluate injured eye and orbit to assess intraocular and extra ocular injuries as well as fractures. It may also detect foreign bodies.
- Ultrasonography: B-scan ultrasound is an important investigation to detect intraocular and intraorbital damage, especially when ocular media is not clear. B-scan can be used to visualize vitreous hemorrhage, retinal detachment, intraocular foreign bodies, damage to the extraocular muscles, and scleral rupture. It can also offer information on the state of the posterior lens capsule and exudates in vitreous in case of endophthalmitis. It should be used with caution if ruptured globe is suspected, and typically deferred if ruptured globe is known.
- CT scan: In the case of orbital trauma, 1.5 to 2 mm cuts should be performed in axial and coronal plains (Figure 8). CT is considered superior to ultrasound for locating foreign bodies. This is a non-invasive method that does not require direct contact with the eye and may identify damage to traumatized eye, deformed eye wall, vitreous hemorrhage and retinal detachment.
- MRI: May be useful in case of patients with non-magnetic foreign bodies. Indications for use in those with pacemakers or implantable metallic hardware vary by type of hardware. It should be used with caution if ruptured globe is suspected, especially in cases of potential metallic foreign body. Can also be used in pregnant patients.
Electrophysiology
- Full-field electroretinogram: In eyes with no light perception, the full-field ERG is an important method to evaluate the vitality of photoreceptor function.
- Multifocal ERG: This modality may be used to detect which areas of the retina may have been affected by trauma.
Laboratory tests
A complete blood count, basic metabolic panel, international normalized ratio, prothrombin time (PT), and partial prothrombin time (PTT) may be required prior to surgery. Sickle cell testing should be considered in patients with hyphema.
Management
General treatment
- Management of post-traumatic cataract and glaucoma will depend upon time interval between injury and presentation of patient as well as clarity of ocular media, presence and morphology of cataract, and degree of IOP elevation if glaucoma is present.
- Indications for prompt surgical intervention include the following:
- Presence of cortical material in the anterior chamber is a sign of lens proteins being released, which can lead to glaucoma.
- Obscured visualization: if posterior segment injuries cannot be identified and treated due to cataract
Medical therapy
- The degree of intraocular inflammation dictates medical management. Topical, periocular and systemic corticosteroids are used in the setting of inflammation without infection.
- Cycloplegics, short-acting and long-acting, are to be used to control pain.
- IOP lowering agents, topical and oral, are to be used in the setting of IOP elevation. Carbonic Anhydrase Inhibitors should be avoided in patients with sickle cell disease.
- Topical, oral and intravenous antibiotics should be used depending on the injury, and subconjunctival or intraocular antibiotics should be considered in cases of ruptured globe.
Surgery
- Primary vs secondary procedure: In patients undergoing globe repair with an identified significant traumatic cataract, the decision needs to be made whether to manage both simultaneously or in a staged approach.
- Children: It is important to perform surgeries soon to avoid amblyopia. Inflammation is more pronounced in younger children, and examination is often difficult. In cases of traumatic cataract in children younger than 2 years of age, both lensectomy and vitrectomy via a pars plana route are often performed. Intraocular lens implantation as part of the primary procedure is often avoided in children younger than 2 years of age.
- Anesthesia: General, local, or local and monitored anesthesia may be used according to anesthetist and surgeon preference. Retrobulbar anesthesia should be avoided if there is a suspicion of a ruptured globe.
- Points to take into consideration when planning surgery:
- Synechiealysis and sphincter management: Those with anterior and posterior synechiae need close follow-up and management. Synechiae are membranes of various densities that attach to iris and lens capsules; peeling of these membranes can release traction and improve visual outcomes. Pupil expansion devices such as a Malyugin ring (Figure 9) may be used to expand the pupil in the setting of synechiae, pupil seclusion, and miosis due to fibrin membranes on the iris when performing cataract extraction.
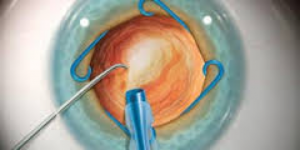 Figure 9: Malyugin Ring. Source: http://www.mst-surgical.com/
Figure 9: Malyugin Ring. Source: http://www.mst-surgical.com/ - Peripheral iridectomy: Pupillary block glaucoma can occur due to post-traumatic inflammation – a peripheral iridectomy may be considered in cases of significant synechiae causing pupillary seclusion.
- Ophthalmic viscosurgical device (OVD): OVD serves many functions and is essential in modern cataract surgery. OVDs are used to protect corneal endothelium and to improve view into the anterior segment, tamponade vessels by increasing IOP, marginalize bleeding, and can tamponade vitreous in areas of zonular incompetence
- Anterior capsule management: Status of the anterior capsule needs to be determined during surgery to plan for location of placement of the intraocular lens. Capsular breaks can extend during hydrodissection and manipulation of the lens. Hydrodisection should be avoided or performed slowly and carefully to minimize the chances of capsular disruption and having the lens fall into the posterior segment.
- Nucleus management: The surgical technique is to be selected according to morphology of the lens and the condition of tissues surrounding the lens. Phacoemulsification or small incision cataract surgery is to be done for cataracts with hard, large nuclei. With a lens that had either a white soft or rosette type of cataract, unimanual or bimanual aspiration is used especially if the patient is young. Membranectomy and anterior vitrectomy, either via an anterior or pars plana route, may need to performed.
- Intraocular lens: Location depends upon status of the posterior capsule. In the right conditions, a posterior chamber IOL should be implanted. The appropriate setting includes minimal intraocular inflammation and hemorrhage and good view of anterior segment structures. If capsular support is inadequate for a posterior chamber IOL, an anterior chamber IOL, sulcus IOL, transsclerally fixated IOL or iris fixated IOL may be appropriate choices. Inaccurate keratometry measurements can occur if the patient has scarring from a corneal laceration or if the corneal contour is changing. In these situations, leaving the patient aphakic might be a favorable option, with insertion of an IOL at a later time as a secondary procedure.
- Selection of IOL is discussed below:
- Patients with a history of uveitis should be spared from silicone lens implants, as inflammatory debris is prone to collecting on the surface of the optic, leading to impaired vision.
- Acrylic lenses are preferred in eyes that may have to undergo vitreoretinal surgery in the future or in eyes with a history of uveitis.
- A 3-piece posterior chamber intraocular lens may be placed in the ciliary sulcus of eyes with an intact anterior capsule but compromised posterior capsule.
- In eyes without capsular support, an intraocular lens may be sclerally fixated.
- An anterior chamber IOL may be implanted if there is adequate iris support.
- Surgeon’s experience with lens types and familiarity with implantation methods are ultimately the deciding factors in choice of IOL.
- An anterior vitrectomy must be performed before starting phacoemulsification or cortical aspiration if vitreous is present in the anterior chamber. Failure to do so may lead to vitreous aspiration and retinal traction.
- Primary posterior capsulotomy and vitrectomy: Recurrent inflammation is more likely found in patients who had undergone previous surgery for trauma. In such cases; the ocular media will turn hazy due to condensation of the anterior vitreous unless a vitrectomy is performed. Hence, capsulectomy and vitrectomy via an anterior/pars plana route are often performed in adults.
Surgical follow-up
- Appropriate follow-up is to be scheduled- often arranged for post-operative day 1, week 1, month 1, and month 3, depending on post-operative course. Intraoperative and postoperative complications may require more frequent follow-up.
- At every follow-up examination, visual acuity and anterior segment examination is required, and examination of the posterior segment is recommended.
- Periocular and systemic corticosteroids are to be titrated according to degree of inflammation and the risk of IOP spikes.
Glaucoma Associated with Trauma
- Angle recession glaucoma: occurs due to a tear between the longitudinal and circular muscle fibers, both of which make up the ciliary body, and often includes the trabecular meshwork. The development of glaucoma can occur soon after ocular trauma or may develop over the course of months to years. Glaucoma is chronic, typically unilateral and has some of the following gonioscopic findings: brown-colored broad angle recess, torn or absent iris processes, white scleral spur, PAS at the border of the recession, and depression in the overlying trabecular meshwork. Initial treatment is medical, although incisional glaucoma surgery may be needed.
- Lens particle glaucoma: can occur during the setting of surgical trauma, accidental trauma, or capsulotomy. Lens cortex particles obstruct the trabecular meshwork and can often be seen floating in the anterior chamber. This results in elevated IOP, anterior chamber reaction, microcystic corneal edema, and development of synechiae. This usually occurs within weeks of initial surgery or trauma, but can occur months to years later.
- Phacoantigenic glaucoma: occurs after surgical or accidental trauma, patients become sensitized to their own lens proteins resulting in granulomatous inflammation. Clinical signs include anterior chamber reaction with keratic precipitates on both the corneal endothelium and the anterior lens surface. Treatment is initially with corticosteroids and aqueous suppressants – if this is not successful, the lens material can be surgically removed.
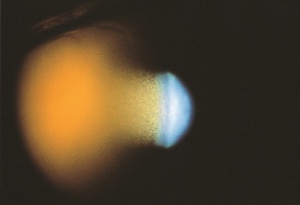
- Hemolytic or ghost cell glaucoma: may develop after vitreous hemorrhage. In hemolytic glaucoma, trabecular flow is blocked by hemoglobin-laden macrophages, while in ghost cell glaucoma, degenerated red blood cells gain access to the anterior chamber by bypassing a disrupted hyaloid face and obstruct the trabecular meshwork (Figure 10). Glaucoma clears once hemorrhage has resolved.
Prognosis
- Visual gain following surgery for traumatic cataracts is a complex process, because multiple ocular co-morbidities may be present. Electrophysiological and radio-imaging investigations are important tools for assessing co-morbidities associated with an opaque lens.
- Morphology of traumatic cataracts and surgical technique may influence final visual outcome.
- Ocular trauma score is validated value which may forecast visual outcome.
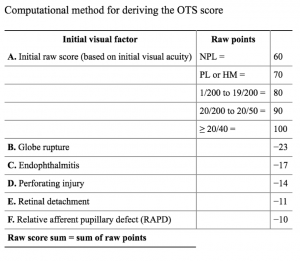
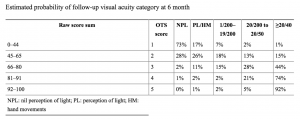
Ocular Trauma Score: The United States Eye Injury Registry (USEIR) developed the Ocular Trauma Score (OTS) with support from the Centers for Disease Control and Prevention (CDC)*. The OTS provides a single probability estimate of an eye trauma patient will obtain a specific visual range by six months after injury. The OTS can be used as an aid in the counseling and treatment of eye injury patients, and is able to direct attention toward resource needs and rehabilitation during the treatment process. The OTS is meant to be a continually evolving scoring system to be used by the clinician to facilitate patient counseling, treatment, rehabilitation, and research.
How to calculate the Ocular Trauma Score:(1) First, determine the patient’s initial visual acuity after the injury and their tissue diagnoses. Second, assign a raw point value for initial visual acuity from row A in Table 1. Then subtract the appropriate raw points for each diagnosis from rows B-F. (For example, a patient with an initial visual acuity of 1/200, scleral rupture, and retinal detachment would receive a raw OTS score of 80-23-11= 46.) Higher OTS scores tend to indicate a better prognosis. To provide an estimate of the patient’s probability of attaining a specific visual acuity range at a six-month follow-up, locate the row in Table 2 corresponding to the patient’s OTS. (A patient with a raw OTS score of 46 would have an OTS category score of 2.) Table 2 shows the estimated probability of all potential visual outcomes vision after 6 months.
Additional Resources
- Porter D, Jimenez EM. Corneal Laceration. American Academy of Ophthalmology. EyeSmart/Eye health. https://www.aao.org/eye-health/diseases/corneal-laceration. Accessed March 07, 2019.
References
- Khatri SK, Lewis AE, Schein OD, Thapa MD, Pradhan EK, Katz J, et al. The epidemiology of ocular trauma in rural Nepal. Br J Ophthalmol. 2004; 88:456-60.
- Abraham, D. I.; Vitale, S. I.; West, S. I.; Isseme, I. Epidemiology of eye injuries in rural Tanzania. Ophthalmic Epidemiol.1999; 6:85-94
- D. Virgil Alfaro, Eric P. Jablon, Monica Rodriguez Fontal, Simon J. Villalba, Robert E.Morris, Michael Grossman, Enrique Roig-Melo et al. Fishing-related ocular trauma. American Journal of Ophthalmology.2005; 139: 488-492
- Shah M, Shah S, Khandekar R. Ocular injuries and visual status before and after Their management in the tribal areas of Western India-A historical cohort study Grafes Arch Clin Exp Ophthalmol 2008; 246:191–197.
- Kuhn F, Morris R, Witherspoon CD, Mester V,) The Birmingham Eye Trauma Terminology system (BETT). J Fr Ophtalmol. 2004; 27:206-10.
- Vajpayee RB, Sharma N, Dada T, Gupta V, Kumar A, Dada VK. Management of posterior capsule tears. Surv Ophthalmol 2001;45:473-88.
- Krishnamachary M, Rathi V, Gupta S. Management of traumatic cataract in children. J Cataract Refract Surg 1997;23 Suppl 1:681-7.
- Churchill AJ, Noble BA, Etchells DE, George NJ. Factors affecting visual outcome in children following uniocular traumatic cataract. Eye (Lond) 1995;9 ( Pt 3):285-91
- Thylefors B, Chylack LT Jr, Konyama K, Sasaki K, Sperduto R, Taylor HR, et al. A simplified cataract grading system. Ophthalmic Epidemiol. 2002; 9:83-95.
- Weinand F, Plag M, Pavlovic S. (2003) Primary implantation of posterior chamber lenses after traumatic cataract penetration. Ophthalmology.100:843-6.
- Baykara M, Dogru M, Ozçetin H, Ertürk H (2002) Primary repair and intraocular lens implantation after perforating eye injury. J Cataract Refract Surg. 28:1832-35.
- Mohammad pour M, Jafarinasab MR, Javadi MA (2007) Outcomes of acute postoperative inflammation after cataract surgery. Eur J Ophthalmol. 17:20-8.
- Khvatova AV, Kruglova TB. (1992) Intraocular correction in the restorative therapy of children with congenital and traumatic cataracts. Vestn Oftalmol.108:18-21.
- Corbett MC, Shilling JS, Holder GE. (1995) The assessment of clinical investigations: the Greenwich Grading System and its application to electro diagnostic testing in ophthalmology. Eye. 9:59-64.
- Segev Y, Goldstein M, Lazar M, Reider-Groswasser I (1995) CT appearance of a traumatic cataract. AJNR Am J Neuroradiol. 16:1174-1175.
- McWhae JA, Crichton AC, Rinke M (2003) Ultrasound Biomicroscopy for the assessment of zonules after ocular trauma. Ophthalmology. 110:1340-1343.
- Zhang Y, Zhang J, Shi S. (1998) Determination of posterior lens capsule status in traumatic cataract with B-Ultrasonography. Zhonghua Yan Ke Za Zhi.34:298-299.
- Synder A, Kobielska D, Omulecki W. (1999) Intraocular lens implantation in traumatic cataract. Klin Oczna. 101:343-6.
- Brar GS, Ram J, Pandav SS, Reddy GS, Singh U, Gupta A (2001) Postoperative complications and visual results in uniocular pediatric traumatic cataract. Ophthalmic Surg Lasers. 32:233-8.
- Wos M, Mirkiewicz-Sieradzka B. (2004) Traumatic cataract--treatment results. Klin Oczna.106:31-4.
- Gradin D, Yorston D. (2001) Intraocular lens implantation for traumatic cataract in children in East Africa. J Cataract Refract Surg. 27:2017-25.
- Baklouti K, Mhiri N, Mghaieth F, El Matri L. Traumatic cataract: clinical and therapeutic aspects. Bull Soc Belge Ophtalmol. 298:13-7.
- Lacmanovic Loncar V, Petric I. (2004) Surgical treatment, clinical outcomes, and complications of traumatic cataract: retrospective study. Croat Med J. 45:310-3.
- Vatavuk Z, Pentz A. (2004) Combined clear cornea phacoemulsification, vitrectomy, foreign body extraction, and intraocular lens implantation. Croat Med J. 45:295-8.
- Morgan KS. (1993) Cataract surgery and intraocular lens implantation in children. Curr Opin Ophthalmol. 4:54-60.
- American Society of Ocular Trauma. Ocular trauma Score (OTS) http://www.asotonline.org/ots.html accessed on 10/12/2008.
- Sternberg, P., Jr.; de Juan, E., Jr.; Michels, R. G.; Auer, C. (1984) Multivariate analysis of prognostic factors in penetrating ocular injuries. Am J Ophthalmol. 98:467-72
- Behbehani AM, Lotfy N, Ezzdean H, Albader S, Kamel M, Abul N. (2002) Open eye injuries in the pediatric population in Kuwait. Med Princ Pract.11:183-9.
- Cillino S, Casuccio A, Di Pace F, Pillitteri F, Cillino G.A (2008) five-year retrospective study of the epidemiological characteristics and visual outcomes of patients hospitalized for ocular trauma in a Mediterranean area. BMC Ophthalmol. 22;6-8.
- Smith AR, O'Hagan SB, Gole GA Epidemiology (2006) of open- and closed-globe trauma presenting to Cairns Base Hospital, Queensland. Clin Experiment Ophthalmol. 34:252-9.
- Mohammad pour M, Jafarinasab MR, Javadi MA. Outcomes of acute postoperative inflammation after cataract surgery. Eur J Ophthalmol. 2007; 17:20-8.
- Gupta A, Srinivasan R, Gulnar D, Sankar K, Mahalakshmi T. Risk factors for post-traumatic endophthalmitis in patients with positive intraocular cultures. Eur J Ophthalmol 2007; 17: 642-7.
- Zhang Y, Zhang MN, Jiang CH, Yao Y, Zhang K. Endophthalmitis following open globe injury. Br J Ophthalmol;2010:94:111-4.
- Narang S, Gupta V, Simalandhi P, Gupta A, Raj S, Dogra MR. Paediatric open globe injuries. Visual outcome and risk factors for endophthalmitis. Indian J Ophthalmol 2004;52:29-34.
- Cebulla CM, Flynn HW, Jr. Endophthalmitis after open globe injuries. Am J Ophthalmol 2009: 147: 567-8.
- Andreoli CM, Andreoli MT, Kloek CE, et al. Low rate of endophthalmitis in a large series of open globe injuries.
- Jonas JB, Knorr HL, Budde WM. Prognostic factors in ocular injuries caused by intraocular or retrobulbar foreign bodies. Ophthalmology 2000;107:823-8.
- Jonas JB, Budde WM. Early versus late removal of retained intraocular foreign bodies. Retina 1999;19:193-7.
- Yang CS, Lu CK, Lee FL, Hsu WM, Lee YF, Lee SM. Treatment and outcome of traumatic endophthalmitis in open globe injury with retained intraocular foreign body. Ophthalmologica;2010:224:79-85.
- Unejo SA, Ahmed M, Alam M. (2010)Endophthalmitis in paediatric penetrating ocular injuries in Hyderabad. J Pak Med Assoc ; 60:532-535.
- Al-Mezaine HS, Osman EA, Kangave D, Abu El-Asrar AM.(2010) Risk factors for culture-positive endophthalmitis after repair of open globe injuries. Eur J Ophthalmol ; :201-208.
- Wade PD, Khan SS, Khan MD. (2009) Endophthalmitis: magnitude, treatment and visual outcome in northwest frontier province of Pakistan. Ann Afr Med ; 8:19-24.
- Viestenz A, Schrader W, Behrens-Baumann W. (2008) Traumatic Endophthalmitis Prevention Trial (TEPT)]. Klin Monbl Augenheilkd ; 225:941-946.
- Wykoff CC, Flynn HW, Jr., Miller D, Scott IU, Alfonso EC.(2008) Exogenous fungal endophthalmitis: microbiology and clinical outcomes. Ophthalmology ; 115:1501-1507, 07 e1-2.
- Al-Omran AM, Abboud EB, Abu El-Asrar AM.(2007) Microbiologic spectrum and visual outcome of posttraumatic endophthalmitis. Retina ; 27:236-242.
- Chhabra S, Kunimoto DY, Kazi L, et al.(2006) Endophthalmitis after open globe injury: microbiologic spectrum and susceptibilities of isolates. Am J Ophthalmol ; 142:852-854.
- Gupta A, Srinivasan R, Kaliaperumal S, Saha I.(2008) Post-traumatic fungal endophthalmitis--a prospective study. Eye (Lond) ; 22:13-17.
- Essex RW, Yi Q, Charles PG, Allen PJ.(2004) Post-traumatic endophthalmitis. Ophthalmology ; 111:2015-2022.
- Lieb DF, Scott IU, Flynn HW, Jr., Miller D, Feuer WJ.(2003) Open globe injuries with positive intraocular cultures: factors influencing final visual acuity outcomes. Ophthalmology ; 110:1560-1566.
- Sabaci G, Bayer A, Mutlu FM, Karagul S, Yildirim E.(2002) Endophthalmitis after deadly-weapon-related open-globe injuries: risk factors, value of prophylactic antibiotics, and visual outcomes. Am J Ophthalmol ; 133:62-69.
- Duch-Samper AM, Chaques-Alepuz V, Menezo JL, Hurtado-Sarrio M. (1998)Endophthalmitis following open-globe injuries. Curr Opin Ophthalmol ; 9:59-65.
- American Academy of Ophthalmology. Basic Clinical Science Course: Glaucoma. San Francisco: American Academy of Ophthalmology, 2015.
- Scott R. The Ocular Trauma Score. Community Eye Health. 2015;28(91):44-5.