Primary Localized Conjunctival Amyloidosis
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Conjunctival amyloidosis is a unique localized process rarely associated with systemic involvement that usually occurs in middle-aged adults. It generally involves the palpebral conjunctiva but may occur anywhere in the conjunctiva. Patients may present with symptoms of pain or ptosis, or may remain completely asymptomatic.
Disease Entity
Classification
Amyloidosis is classified by extent. It may be localized to one organ or may be systemic. It can also be classified according by amyloid protein type, with AL and AA subtypes being the most common.
Pathophysiology
Amyloidosis is caused by the extracellular tissue deposition of amyloid fibrillar proteins. Amyloid fibrils are insoluble polymers comprised of low molecular weight subunit proteins, which adopt a beta-pleated sheet configuration. The deposition of these insoluble proteins can lead to tissue toxicity and disease. At least 27 different human protein precursors of amyloid fibrils are now known. The most frequent types are the AL and AA subtypes. Some types of amyloidosis are hereditary and have been linked to genetic mutations. For others there is no hereditary component.
Diagnosis
Symptoms
The most common presenting symptoms include pain and ptosis. However, some patients may remain asymptomatic and the lesion is discovered only incidentally. For patients with periocular or orbital amyloidosis proptosis, globe displacement, diplopia, and limited ocular motility may be observed.
Ocular Signs
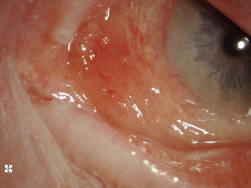
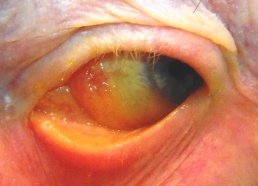
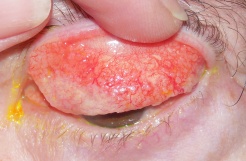
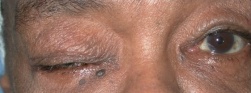
Conjunctival amyloidosis usually appears as confluent fusiform lesions or polypoidal papules that have a salmon-colored or yellow-pink color. Any conjunctival surface may be involved, but the superior fornix and tarsal conjunctiva are the most frequently affected. This deposition may result in ptosis. Patients may also present with recurrent subconjunctival hemorrhage from the accumulation of amyloid in the walls of the blood vessels. The eyelid, extraocular muscles, and lacrimal gland may also be affected by amyloidosis.
Figure 1a & 1b: Slit lamp photo of the conjunctival amyloidosis
Figure 2: Amyloidosis involving the upper eyelid
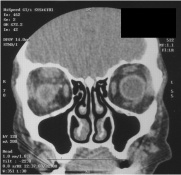
Figure 3a: Amyloidosis causing ptosis Figure 3b: CT scan showing amyloid infiltration of the levator and superior rectus of the right eye
Systemic Manifestations
Systemic amyloidosis most commonly involves the kidney, the heart, and the liver. Renal involvement may present as asymptomatic proteinuria or nephritic syndrome. End-stage renal disease may result in cases of severe disease. Cardiac involvement may lead to heart failure, arrhythmia, heart block, or even myocardial infarction from amyloid deposition in the coronary arteries. Hepatomegaly with or without splenomegaly may result from amyloid deposition in the liver. Amyloidosis may also result in neurologic, musculoskeletal, hematologic, pulmonary and cutaneous disease.
Clinical diagnosis
The diagnosis of conjunctival amyloidosis can only made with tissue biopsy and confirmed histopathology.
Pathology
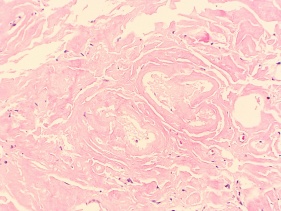
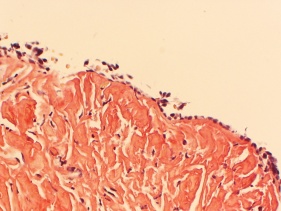
On hematoxylin-eosin staining, conjunctival amyloid demonstrates homogeneous, amorphous eosinophilic material in the conjunctival stroma. With Congo red staining, there is pathognomonic apple-green birefringence, which demonstrates dichroism with polarization microscopy.
Figure 4: H & E stain showing diffuse acellular, eosinophilic material in the conjunctiva
Figure 5: Amyloidosis stains strongly for Congo Red
Figure 6: "Apple-Green" Birefringence under polarized light
Systemic work-up
Systemic involvement should be ruled out in any patient who presents with an isolated conjunctival lesion. Amyloidosis can frequently affect the kidney, heart, and liver. Tests for cardiac, liver and kidney function including an echocardiogram and electrocardiogram as well as serum and urine protein electrophoresis should be obtained. More specific immunofixation and free light chain electrophoresis of both the serum and urine should be obtained as well. Other tests such as bone marrow biopsy, abdominal fat biopsy, and rectal biopsy should be considered. Imaging tests such as CT or MRI may be helpful in localizing and detecting the extent of orbital disease.
Differential diagnosis
The clinical differential diagnosis of conjunctival amyloidosis includes lymphoma, leukemia, squamous cell carcinoma, sebaceous cell carcinoma, sarcoidosis, and melanoma. The smooth salmon colored appearance of amyloidosis is similar to lymphoma.
Management
Management modalities include observation, judicious use of artificial tears, excision, liquid nitrogen cryotherapy or low dose external beam therapy [1]for localized conjunctival amyloidosis. For patients with systemic manifestations, stem cell transplant, chemotherapy, and steroids may be considered depending on the extent of the disease. Surgical debulking remains the standard treatment, but because complete excision is not always possible, the disease may recur. Demirci and Leibovitch report a recurrence rate of 21-27% after surgical debulking of localized orbital amyloidosis. Newer therapies such as cryotherapy may further decrease recurrence rate by decreasing blood supply to the surrounding tissue, but more research is needed. Radiotherapy for localized amyloidosis has also been reported but is not considered the standard of care.
For patients with systemic amyloidosis, treatment varies depending on the disease manifestations. Some patients may be considered for chemotherapy and steroids while others may qualify for stem cell transplant. There are multiple clinical trials studying the effect of different chemotherapy agents in treating systemic amyloidosis. Patients with end-stage renal disease may require dialysis or renal transplantation. For patients with hereditary amyloidosis in which abnormal proteins are produced by the liver, liver transplant may lead to regression of the disease.
References
- ↑ Copperman TS, Truong MT, Berk JL, Sobel RK, External beam radiation for localized periocular amyloidosis: a case series. Orbit. 2019 Jun;38(3):210-216. doi: 10.1080/01676830.2018.1483407. Epub 2018 Jun 11.
- Demirci H, Shields CL, Eagle RC Jr, et al. Conjunctival amyloidosis: report of six cases and review of the literature. Surv Ophthalmol. 2006 Jul-Aug;51(4):419-33.
- Do Socorro, Costa, da Silva M, Campos LR. Ocular sarcoidosis. Mayo Clin Proc 2010 Oct; 85(10):876.
- Fraunfelder FW. Liquid nitrogen cryotherapy for conjunctival amyloidosis. Arch Ophthal. 2009; 127(5):645-8.
- Gertz MA. Immunoglobulin light chain amyloidosis: 2011 update on diagnosis, risk-stratification, and management. Am J Hematol. 2011 Feb;86(2):180-6.
- Leibovitch I, Selva D, Goldberg RA, et al. Periocular and orbital amyloidosis: clinical characteristics, management, and outcome. Ophthalmology. 2006 Sep;113(9)
- Orbit, Eyelids, and Lacrimal System, Section 7. Basic and Clinical Science Course. San Francisco: American Academy of Ophthalmology; 2010.
- Taban M, Piva A, See RF, et al. Review: orbital amyloidosis. Ophthal Plast Reconstr Surg. 2004 Mar;20(2):162-5.