Compressive Visual Field Defects
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Compressive Visual field defects
Disease
Mass effect is the compression of nearby structures by a mass (aneurysm, tumor, hematoma, abscess). When affecting the visual pathway, mass effect can cause a multitude of changes in a patient’s visual field. Specific visual field deficits can be attributed to the location of compression, which in turn can help clinicians localize lesions based on physical examination and diagnostic testing.
Etiology
The locations and associated tumors along the visual pathway include:
Orbit
The orbit can be divided into 3 different zones: intraconal, extraconal, and intercompartmental. The “cone” refers to the area created by the four rectus muscles and the intermuscular membrane that joins them and extends posteriorly to the insertion of the muscle tendons on the annulus of Zinn at the orbital apex[1].
- Intraconal
- Cavernous hemangioma
- Optic nerve sheath meningioma[2]
- Schwannoma
- Neurofibroma (localized)
- Fibrous histiocytoma
- Capillary hemangioma (children)
- Rhabdomyosarcoma
- Malignant rhabdoid tumor
- Primary orbital melanoma
- Leiomyoma
- Leiomyosarcoma
- Granular cell tumor
- Hemangiopericytoma
- Venous varix
- Arteriovenous malformation
- Extraconal
- Lymphoma
- Lymphangioma
- Metastasis
- Adults: breast, lung, unknown primary, prostate, melanoma
- Children: neuroblastoma, Ewing sarcoma, leukemia
- Rhabdomyosarcoma (embryonal type, children)
- Dermoid Epidermal inclusion cyst
- Paranasal sinus neoplasms
- Cavernous hemangioma
- Capillary hemangioma (children)
- Langerhans cell histiocytosis (<1% of all orbital masses)
- Fibromatosis (most common orbital lesion of infancy)
- Teratoma
- Hematic cyst
- Hemangiopericytoma
- Burkitt's lymphoma
- Granulocytic sarcoma
- Primary osseous neoplasm
- Ossifying fibroma
- Osteoma
- Plasmacytoma/multiple myeloma (0.5% of all orbital masses)
- Osteosarcoma
- Lacrimal gland neoplasms
- Epithelial (50%)
- Benign (50%) - Benign mixed tumor (pleomorphic adenoma)
- Malignant (50%) - Epithelial carcinoma, Adenoid cystic carcinoma, Malignant mixed tumor, Metastasis
- Lymphoproliferative/Inflammatory (50%)
- Lymphoma
- Pseudotumor
- Sjogren's
- Sarcoid
- Granulocytic sarcoma (chloroma)
- Lacrimal sac tumors
- Malignant (90%)
- Carcinoma
- Squamous
- Transitional
- Adenoid cystic
- Oncocytic adenocarcinoma
- Mucoepidermoid
- Melanoma
- Hemangiopericytoma
- Lymphoma
- Metastasis
- Carcinoma
- Benign
- Mucocele
- Polyp
- Fibroma
- Fibrous histiocytoma
- Oncocytoma
- Oxyphillic adenoma
- Malignant (90%)
- Epithelial (50%)
- Intercompartmental
- Lymphangioma
- Rhabdomyosarcoma
- Neurofibroma (plexiform and diffuse)
- Capillary hemangioma
- Venous varix
- Arteriovenous malformation
Intracranial optic nerve, junctional and optic chiasm
The optic chiasm is the point where the two optic nerves meet[3]. The chiasm lies in the suprasellar cistern above the sella turcica, and is surrounded by the hypothalamus superiorly and the infundibulum and pituitary gland inferiorly[3]. The most common cause of an optic chiasm lesion is from a compressive source, and this is often neoplastic in nature[4]. However, the etiology is less commonly from a congenital, traumatic, vascular, inflammatory, infectious, demyelinating, and infiltrative source[5]. Suprasellar and sellar lesions can produce optic neuropathies in one or both eyes; a junctional scotoma (ipsilateral visual loss and contralateral superotemporal visual field loss) or the junctional scotoma of Traquair (monocular hemianopic visual field loss); or chiasmal bitemporal hemianopsia (including paracentral bitemporal hemianopsia from crossing macular fiber in posterior chiasm).
- Neoplasm is the most common cause of chiasmal syndrome and is from both direct compression and inflammatory disruption of the vascular circulation to the chiasm[4]
- Most common:
- Pituitary Adenoma - most common cause of chiasmal lesion
- Craniopharyngioma
- Meningioma (including optic nerve sheath meningioma)
- Optic glioma
- Less common[5]:
- Chordoma
- Germinoma
- Endodermal sinus tumor
- Leukemia
- Hodgkin’s and non-Hodgkin’s lymphoma
- Nasopharyngeal carcinoma
- Metastatic disease
- Non-neoplastic etiologies can also cause direct compression at the chiasm, including[5]:
- Sphenoid sinus mucocele
- Arachnoid cysts
- Rathke’s cleft cyst
- Epidermoid cysts
- Fibrous dysplasia
- Vascular sources may cause compression and/or ischemia to the chiasm[5][6].
- Apoplexy - from either vascular sources or tumors
- Aneurysm of anterior communicating artery or internal carotid artery
- Cavernous hemangiomas
- Arteriovenous malformations
- Trauma can rarely cause chiasmal syndrome from a frontal bone fracture and/or anterior skull base fracture and hematoma can act as a mass lesion[7]
- Infectious causes leading to mass effect[5]
- Syphilitic gumma
- Other granulomatous infectious disease
- Arachnoiditis
- Abscess
- Infiltrative disease
- Sarcoid granuloma
- Most common:
Retrochiasmal Lesions
The retrochiasmal afferent pathway extends posteriorly past the chiasm and includes the optic tract, lateral geniculate body, optic radiations, and the striate cortex[8]. The etiologies of retrochiasmal lesions are different between adults and children. Vascular and ischemic etiologies (i.e hemorrhage, aneurysm) are more frequent for adults, whereas neoplasms are more common for children[9][10].
Optic tract lesions are relatively uncommon[9], with lateral geniculate nucleus (LGN) lesions being even more rare but when seen, are due to ischemic, demyelinating, or traumatic etiologies.
Optic radiations exit dorsally from the LGN into two separate fascicles: Inferior (known as Meyer’s loop) and Superior (known as the parietal fascicles).
There can be several different etiologies affecting the optic pathway posterior to the chiasm:
- Hemorrhage[8]
- Hypertensive hemorrhage
- Amyloid angiopathy
- Vascular malformations
- Trauma
- Hemorrhagic infarction
- Secondary to anticoagulation
- Drug related
- Hemorrhage into tumors
- Primary Tumors[11]
- Gliomas
- Astrocytomas
- Ependymomas
- Oligodendrogliomas
- Gangliogliomas
- Intraventricular Tumors
- Choroid plexus papillomas
- Meningiomas
- Primary CNS B-cell lymphoma
- Metastatic Tumors[12]
- Lung
- Breast
- Melanomas
- Genitourinary
- Gastrointestinal
- Gynecologic
- Infectious/Abscess
- Inflammatory (sarcoidosis)
Clinical Manifestations
Depending on the patient’s lesion, the clinical manifestations can be variable. Patients may experience nonspecific symptoms such as headaches (from increased intracranial pressure), relative afferent pupillary defect, seizures, and inability to perform activities of daily living[13].
Orbit
The clinical manifestations of orbital tumors may include gaze-evoked amaurosis (due to reduction in the flow of blood to the optic nerve and retina), proptosis, optic disc swelling (direct compression of the optic nerve), optociliary shunt (actually retinochoroidal venous collateral) vessels, and choroidal folds[13]. Visual field manifestations can be variable within the orbit, depending on the location of compression and the fibers affected. There can be a progression of symptoms, including central scotomas, enlarged blind spots, and generalized constriction[13]. If there is a complete lesion or compression at the optic nerve, patients may experience total monocular vision loss in the affected eye.
Chiasm
The clinical manifestations of an optic chiasm lesion can manifest with varying visual field defects and decreased visual acuity. Due to the chiasm proximity to the pituitary gland, endocrine symptoms may present. Headache and photophobia have also been known symptoms[4].
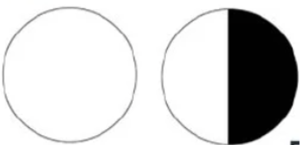
The visual field defect seen in the chiasmal syndrome is typically a bitemporal hemianopsia. However, other field defects, like a junctional scotoma or a junctional scotoma of Traquair, may occur[14]. To understand these field defects, it’s necessary to know how the eye processes the visual field. The retina processes the opposite side of the visual field. The temporal retinal processes the nasal visual field, while the nasal retina processes the temporal visual field. The same is true for the superior and inferior visual fields - the inferior retina processes the superior visual field, and the superior retina processes the inferior visual field[6]. The retinal ganglion cells all meet at the optic nerve head and form the optic nerve[3]. The optic nerve then courses posteriorly until the two optic nerves meet at the chiasm. At the chiasm, the temporal retinal nerve fibers remain uncrossed on the same side and travel ipsilaterally on the optic tract. However, the nasal retinal fibers decussate to the contralateral side for processing by the contralateral hemisphere of the brain[6]. The visual field defect from a chiasmal lesion thus depends on which fibers are compressed.
The bitemporal hemianopia field defect is the classic presentation of a chiasmal lesion. This presents with loss of both temporal fields and is due to compression of the bilateral decussating nasal fibers at the chiasm, since the nasal fibers correspond with the temporal visual fields[5]. Although it may present as a complete bitemporal hemianopia, most patients will have an asymmetric presentation on visual field testing[4].
The junctional scotoma (see Junctional Scotoma and Junctional Scotoma of Traquair) manifests as a central scotoma in the ipsilateral eye and a superior temporal visual field defect in the contralateral eye. This occurs due to a lesion of the ipsilateral optic nerve, causing an optic neuropathy, and of the contralateral decussating inferior nasal retinal fibers, causing loss of the superior and temporal field in the opposite eye. This is often from an anterior chiasmal lesion[6].
Lastly, the junctional scotoma of Traquair (see Junctional Scotoma and Junctional Scotoma of Traquair) may occur. This occurs from a lesion where the optic nerve and chiasm meet and can present with varying field defects in the monocular, ipsilateral eye[14]. At the junction of the optic nerve and the chiasm, there are two different types of fibers on the optic nerve that can be affected: the nasal crossing fibers and the temporal non-crossing fibers. Most commonly, the nasal crossing fibers are affected, manifesting with a monocular temporal field defect in the ipsilateral eye. Less commonly, the uncrossed temporal fibers are compressed, manifesting with a monocular nasal field defect in the ipsilateral eye. In both of these cases, the contralateral eye is unaffected.
Retro-chiasmal
Optic tract
Neoplasms in the region of the optic tract can produce contralateral incongruous homonymous hemianopia, often with a relative afferent pupillary defect and band atrophy of the optic nerve (in the eye with the temporal visual field defect). As the corticospinal motor tracts are often involved as well causing contralateral hemiparesis[13].
Temporal lobe
Contralateral homonymous superior quadrantanopia often called pie in the sky is seen when a mass lesion is in the temporal lobe. It represents damage to the inferior optic radiating fibers (Meyer’s loop). The associated features like aphasia and hallucinations will help to further localize lesions to the temporal lobe[13].
Parietal Lobe
Mass lesions in parietal lobe produces contralateral inferior homonymous quadrantic field defect also referred as “pie on the floor”. This visual defect is caused by damage to superior fibers of the optic radiations. Damage to the left parietal lobe can result in right-left confusion, agraphia and acalculia referred to as Gerstmann’s Syndrome. Damage to the right parietal lobe can produce contralateral neglect. A bilateral simultaneous confrontation test will elicit a consistent neglect of the visual field in these patients. Bi-lateral damage to parietal lobes can cause “Balint’s Syndrome,” a visual attention and motor syndrome[13].
Occipital lobes
Lesions of the occipital lobe will result in contralateral congruous homonymous hemianopia. The tips of the occipital cortices are supplied by both the MCA and posterior cerebral artery (PCA). So macular vision can be spared when only one of the major cerebral vessel distributions is affected. The monocular temporal crescent is represented in the most anterior mesial portion of the occipital lobe. Sparing or involvement of the monocular temporal crescent localizes to the occipital lobe[13].
Diagnostic Criteria
Automated perimetry (e.g., Humphrey Visual Field (10-2, 24-2, and 30-2)) is recommended for all patients with unexplained visual loss. Depending on the results of the visual field, patients can have further imaging performed.
General treatment
The treatment of the visual field defect depends on the underlying etiology. In general, most mass lesions compressing the visual system require surgery both for diagnostic (pathologic confirmation) and therapeutic (relieve mass effect) indications. Clinicians should be aware of the localizing value of specific visual field defects and perform neuroimaging to exclude a structural lesion including mass lesions[15]. Multidisciplinary evaluation and treatment including orbital or neurosurgery may be necessary depending on the underlying lesion producing the visual field defect.
References
- ↑ Koeller KK, Smirniotopoulos JG. Orbital masses. Semin Ultrasound CT MR. 1998;19(3):272-291. doi:10.1016/s0887-2171(98)90012-9
- ↑ Douglas VP, Douglas KAA, Cestari DM. Optic nerve sheath meningioma. Curr Opin Ophthalmol. 2020;31(6):455-461. doi:10.1097/ICU.0000000000000700
- ↑ 3.0 3.1 3.2 De Moraes, C. G. (2013). Anatomy of the Visual Pathways. Journal of Glaucoma, 22 Suppl 5 Suppl 1, Proceedings of the 18th Annual Optic Nerve Rescue and Restoration Think Tank: “Glaucoma and the Central Nervous System” September 16–17, 2011 New York, NY Sponsored by The Glaucoma Foundation (TGF), S2–S7. https://doi.org/10.1097/IJG.0b013e3182934978
- ↑ 4.0 4.1 4.2 4.3 Takahashi, M., Goseki, T., Ishikawa, H., Hiroyasu, G., Hirasawa, K., & Shoji, N. (2018). Compressive Lesions of the Optic Chiasm: Subjective Symptoms and Visual Field Diagnostic Criteria. Neuro-Ophthalmology (Amsterdam : Aeolus Press. 1980), 42(6), 343–348. https://doi.org/10.1080/01658107.2018.1438477
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 Rubin, R. M., Sadun, A. A., & Piva, A. P. (2019). Lesions of the Optic Chiasm, Parasellar Region, and Pituitary Fossa. In Ophthalmology (Fifth Edition, pp. 909–917.e1). https://doi.org/10.1016/B978-0-323-52819-1.00237-1
- ↑ 6.0 6.1 6.2 6.3 Yoshihara MK, Lui F. Neuroanatomy, Bitemporal Hemianopsia. [Updated 2021 Aug 11]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK545213/
- ↑ Vellayan Mookan, L., Thomas, P. A., & Harwani, A. A. (2018). Traumatic chiasmal syndrome: A meta-analysis. American journal of ophthalmology case reports, 9, 119–123. https://doi.org/10.1016/j.ajoc.2018.01.029
- ↑ 8.0 8.1 Prasad, Sashank. “Retrochiasmal Disorders.” In: Liu, Volpe, and Galetta's Neuro-Ophthalmology: Diagnosis and Management, 3rd ed., Elsevier Inc., Edinburgh, 2019, pp. 293–339.
- ↑ 9.0 9.1 Zhang X, Kedar S, Lynn MJ, et al: Homonymous hemianopias: clinical-anatomic correlations in 904 cases. Neurology 2006; 66: pp. 906-910
- ↑ Kedar S, Zhang X, Lynn MJ, et al: Pediatric homonymous hemianopia. J AAPOS 2006; 10: pp. 249-252
- ↑ Wen PY, and Kesari S: Malignant gliomas in adults. N Engl J Med 2008; 359: pp. 492-507
- ↑ Posner JB: Intracranial metastases. In (eds): Neurologic Complications of Cancer. Philadelphia: F.A. Davis, 1995. pp. 77-110
- ↑ 13.0 13.1 13.2 13.3 13.4 13.5 13.6 Miller, Neil R., Newman, Nancy J. eds. Walsh & Hoyt's Clinical Neuro-Ophthalmology. 6th Edition. 530 Walnut Street, Philadelphia, Pennsylvania 19106 USA; 351 West Camden Street, Baltimore, Maryland 21201-2436 USA:Lippincott Williams & Wilkins; 2005.
- ↑ 14.0 14.1 Foroozan, R. (2003). Chiasmal syndromes. Current Opinion in Ophthalmology, 14(6), 325–331. https://doi.org/10.1097/00055735-200312000-00002
- ↑ Lee AG, Johnson MC, Policeni BA, Smoker WR. Imaging for neuro-ophthalmic and orbital disease - a review. Clin Exp Ophthalmol. 2009;37(1):30-53