Descemet Membrane Anterior Keratoplasty (DMAK)
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Definition and Background
Descemet membrane anterior keratoplasty (DMAK) is a surgical procedure first described in 2024 by Dr. Joshua H. Hou in which a modified decellularized Descemet membrane (DM) is transplanted onto the anterior corneal surface, typically with anterior fetal banded layer side up, to serve as a long-term substrate for ocular surface rehabilitation in patients with impaired corneal healing.[1]
DM is a transparent basement membrane supporting the corneal endothelium on the posterior surface of the cornea. It is composed of type IV collagen, laminins, nidogens, and perlecan[2] and is highly resistant to collagenase degradation.[3] Intraocular transplantation of donor DM with associated corneal endothelium is a well-established procedure known as Descemet Membrane Endothelial Keratoplasty (DMEK).[4] This procedure is widely used for the treatment of endothelial diseases such as Fuchs’ corneal dystrophy or bullous keratopathy.[5][6] In DMEK, diseased DM and corneal endothelium are removed and replaced with healthy donor tissue, representing a direct anatomical replacement of tissue. In DMAK, the same tissue is decellularized and used ectopically on the ocular surface with the primary goal of supporting the corneal epithelium.[1] In both procedures DM functions as a basement membrane; however, in DMEK, corneal endothelium is supported, while in DMAK corneal epithelium is supported.
Experimental work has shown that the anterior fetal banded layer of DM expresses limbal basement membrane–specific proteins (i.e. vitronectin, osteonectin) and supports ex vivo limbal stem cell (LSC) expansion more effectively than human amniotic membrane (HAM).[3][7][8] In cultures, DM also helps maintain the stemness of LSCs and early transient amplifying cells (TACs) even as they migrate centripetally over the cornea and stratify.[3] Normally as LSCs migrate and stratify, they differentiate into mature corneal epithelium and lose their long-term proliferative potential in the process. However, ex vivo data suggests that when supported by DM, LSCs can persist in the basal layer of the corneal epithelium, potentially increasing the reservoir of proliferating cells in the central cornea. Thus, DM is a promising substrate for augmenting corneal epithelialization. DMAK leverages this to treat conditions of impair corneal healing by transplanting DM onto the ocular surface where it serves as a basement membrane for the epithelium.
Functionally, acellular DM is a potential alternative to HAM for several of its use cases in ocular surface procedures. HAM is commonly used as a scaffold for persistent epithelial defects (PEDs) and as a substrate in simple limbal epithelial transplantation (SLET). But HAM is relatively opaque, degrades rapidly on the ocular surface, and has limited long‑term benefit.[9][10][11] In contrast, DM is optically clear, highly resistant to enzymatic degradation, and can persist on the ocular surface even under inflammatory conditions, as evidenced by the behavior of descemetoceles.[2] As such, acellular DM has several key advantages over HAM on the ocular surface and to date DMAK has been reported as both an isolated procedure and a procedure combined with SLET for treating conditions of impaired corneal epithelialization.
Indications
Given the potential for DM to provide long-term support for migrating LSCs and early TACs on the cornea, DMAK is primarily indicated for treatment of partial limbal stem cell deficiency and persistent corneal epithelial defects.
Limbal stem cell deficiency (LSCD)
- Partial LSCD after ocular surface stem cell transplantation (OSSTx) failure.
- In a case report by Cheung, et al. (2025), a patient with PEDs following Keratolimbal allograft (KLAL) failure maintained intact corneal epithelium out to a final follow-up of 1.5 years after DMAK combined with allo-SLET.[1]
- Partial LSCD in the setting of neurotrophic keratopathy and aniridia.
- In a case series of four LSCD eyes with NK (3 eyes) or aniridia (1 eye) reported by Koseoglu, et al. (2025), DMAK resulted in good graft adhesion and no recurrent of central corneal neovascularization out to final follow-up.[12]
- Partial LSCD secondary to chemical burn.
- In a case report presented at the Holland Foundation 3rd Annual Ocular Surface Stem Cell Transplantation Education Symposium (2025) (Hou, et al.), DMAK, combined with symblepharolysis/conjunctival autograft for fornix reconstruction, was successful in treating progressive partial LSCD out to final follow-up of 10 months. (publication pending).
- Partial LSCD in congenital aniridia.
- In a prospective pilot clinical trial of DMAK for treatment of partial LSCD in congenital aniridia (NCT05909735), there was improvement in BCVA, central corneal staining, and central corneal neovascularization out to final follow-up of 6 months (n=19). (Hou, et al., submitted for publication)
Corneal epithelial healing
- PEDs after PKP in the setting of sclerocornea.
- In a case reported by Elsharawi, et al. (2025) of high risk PKP in a pediatric patient with sclerocornea, PKP + DMAK resulted in faster corneal re-epithelialization (≤1 week) than PKP alone (1 month), which was performed in the contralateral eye.[13]
- Corneal re-epithelialization in ocular surface disease: PED, recurrent erosions, LSCD, and NK.
- In a retrospective multicenter, multi-surgeon series of 99 DMAK cases in eyes with ocular surface disease (including refractory PED, recurrent erosions, LSCD, and NK), who had failed treatment with a mean of 1.2 prior amniotic membrane transplants (AMT), corneal re-epithelialization was achieved in 84.8% of patients with a mean healing time of 28 days. (Hou et al., in press).
Surgical Technique
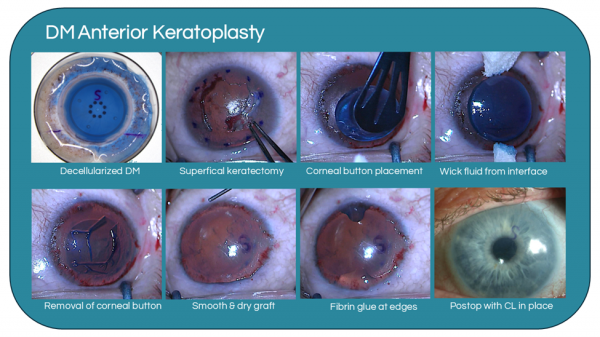
Graft Material
- All current clinical reports use a modified decellularized, pre‑stripped DM allograft supplied as a commercial product (BrightMEM, Brightstar Therapeutics). The graft is provided as DM attached to an anterior stromal cap on a corneal button and can be trephined intraoperatively to the desired diameter. The graft is pre-stained with trypan blue and marked with a reverse S‑stamp on the posterior (endothelial) side of the DM to aid in visualization and orientation, respectively.
Preparation of the recipient bed
- Superficial keratectomy is first performed to remove abnormal epithelium, fibrovascular tissue, and/or anterior stromal scarring. It is critical that the stromal surface is completely dry and cleared of debris after keratectomy to enhance DM adhesion. Preoperative phenylephrine drops and/or intraoperative sponges soaked in epinephrine 1:1000 can be utilized to achieve hemostasis.
- The DMAK trephine size is selected to be at least 1 mm smaller than the debrided area, leaving > 0.5 mm buffer so that the graft lies entirely within the prepared bed and avoids overlap onto irregular tissue.[1] Trephination should be performed using a vacuum trephine to minimize graft slippage during punch.
Positioning
- The posterior endothelial side of the donor graft is placed on the recipient stroma. This ensures that the fetal layer of DM, which expresses limbus-specific proteins), faces anteriorly to support migrating epithelial cells. Orientation is confirmed via the S‑stamp. Fluid is gently wicked from the graft–stromal interface using a cellulose spear or sponge. Repeat peripheral dabbing may be indicated. The graft is then allowed to dry in situ for approximately 1 minute to promote adhesion of the graft to the cornea and allow it to separate from the corneal button. The anterior corneal button is then peeled away, leaving a thin, crystal‑clear DM allograft on the corneal surface
Smoothing
- Any wrinkles in the graft should be smooth out using gentle sweeps using a 27-g or 30-g cannula with balanced salt solution.[1] Care should be taken to avoid dislodging the graft. If the graft is still highly mobile, additional peripheral dabbing to wick out interface fluid may also be indicated.
Securing the graft
- Once the graft is completely smooth without interface fluid or wrinkles, the graft is allowed to dry for 2 minutes to promote adhesion. A small amount of thrombin/fibrinogen glue is applied to seal the edges ensuring 360° coverage. A bandage contact lens (BCL) is then placed over the eye.
- If performing allo-SLET in combination, after placing the DMAK, limbal tissue slivers and pieces are prepared from an ABO-matched cornea and placed along the DMAK surface and then secured with tissue glue.
- A temporary tarsorrhaphy may also be considered to support contact lens retention and graft adhesion, limit exposure, and/or minimize loss of the allo-SLET pieces.
Postoperative care
- The postoperative drop regimen includes antibiotic eye drops 4 times daily (QID) x 1 week, then twice daily (BID) until BCL removal. Hypertonic saline QID is recommended until BCL removal to support adhesion. Steroid eye drops may be used as clinically appropriate or a standard regimen of QID with weekly taper can be utilized.
- Case reports have also used a combination of topical difluprednate 1% QID, tobramycin 0.3% QID, autologous serum tears 20% QID, cyclosporine 0.05% QID (off-label increased dosing), and preservative-free tears.[1]
- Data is still limited and postoperative drop regimen may be variable across surgeons.
- A bandage contact lens is recommended to stay in place for 4 weeks post-operatively.
- Protective measures including no eye rubbing and wearing shield at night should be emphasized to patients.
- Clinical follow‑up focuses on epithelial integrity, graft adhesion (slit‑lamp and AS‑OCT when available), neovascularization, and signs of allo‑immune activity.
Case Examples
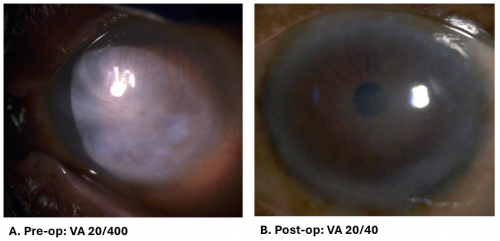
- Patient with partial limbal stem cell deficiency who underwent DMAK in their left eye. Uncorrected visual acuity improved from 20/400 pre-op to 20/40 at 6 month post-op visit.
Potential Complications and Limitations
- This is a new technique that is still evolving without a consensus standardized protocol.
- There are no randomized controlled trials comparing its efficacy against other ocular surface interventions.
- Late epithelial staining / partial surface recurrence (Cheung, et al.) as well as recurrence of neovascularization in ungrafted areas have been reported.[1]
- Intraoperative graft tearing (Koseoglu, et al. had one patient who needed a repeat DMAK due to tearing) has been reported.[12]
- Post-operative graft dislocation, epithelial ingrowth, and graft wrinkling have been reported. (publication pending)
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 Cheung AY, Reinisch CB, Hou JH. Decellularized Descemet Membrane Anterior Keratoplasty With Allogeneic Simple Limbal Epithelial Transplantation for Partial Limbal Stem Cell Deficiency Following Partial Keratolimbal Allograft Failure. Cornea. 2025;44(1):108-112.
- ↑ 2.0 2.1 de Oliveira RC, Wilson SE. Descemet's membrane development, structure, function and regeneration. Exp Eye Res. 2020;197:108090.
- ↑ 3.0 3.1 3.2 Bedard P, Yuan C, Lee S, Roehrich H, Hou JH. Descemet Membrane Versus Amniotic Membrane for Limbal Stem Cell Therapy. Cornea. Published online December 8, 2025
- ↑ Melles GR, Ong TS, Ververs B, van der Wees J. Descemet membrane endothelial keratoplasty (DMEK). Cornea. 2006;25(8):987-990.
- ↑ Ham L, Dapena I, van Luijk C, van der Wees J, Melles GR. Descemet membrane endothelial keratoplasty (DMEK) for Fuchs endothelial dystrophy: review of the first 50 consecutive cases. Eye (Lond). 2009;23(10):1990-1998.
- ↑ Woo JH, Ang M, Htoon HM, Tan D. Descemet membrane endothelial keratoplasty versus descemet stripping automated endothelial keratoplasty and penetrating keratoplasty. American journal of ophthalmology. 2019 Nov 1;207:288-303.
- ↑ Hou JH, Bedard P, Lee S, Yuan C. Ex vivo expansion of limbal stem cells using Descemet’s Membrane as a culture substrate. Invest Ophthalmol Vis Sci. 2019;60(9):4128-4128.
- ↑ Hou JH, Bedard P, Lee S, Yuan C. Comparison of Descemet’s membrane and human amniotic membrane as culture substrates for ex vivo expansion of limbal stem cells. Invest Ophthalmol Vis Sci. 2020;61(7):3267-3267.
- ↑ Lee SH, Tseng SCG. Amniotic membrane transplantation for persistent epithelial defects with ulceration. Am J Ophthalmol. 1997;123(3):303-312. doi:10.1016/S0002-9394(14)70125-4
- ↑ Sell S, de la Presa M, Thakur S, et al. Comparison of Persistent Epithelial Defect Treatment With Sutureless Cryopreserved and Dehydrated Amniotic Membrane. Am J Ophthalmol. 2023;251:32-42. doi:10.1016/j.ajo.2022.12.007
- ↑ Shanbhag S, Patel C, Goyal R, Donthineni P, Singh V, Basu S. Simple limbal epithelial transplantation (SLET): Review of indications, surgical technique, mechanism, outcomes, limitations, and impact. Indian J Ophthalmol. 2019;67(8):1265-1277.
- ↑ 12.0 12.1 Dilruba Koseoglu N, Hamrah P. Ocular Microbiology and Immunology Group Descemet’s Membrane Anterior Keratoplasty (DMAK) Outcomes in Limbal Stem Cell Deficiency Assessed with Anterior Segment Optical Coherence Tomography-A Case Series. 2025.
- ↑ Elsharawi R, Lee D, Mittal A, Chamberlain WD, Tauber J, Redd TK. Use of Decellularized Descemet Membrane Anterior Keratoplasty to Facilitate Epithelialization of Pediatric Penetrating Keratoplasty for Total Sclerocornea. Cornea. 2025;44(12):1543-1545.