Distichiasis
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Definition
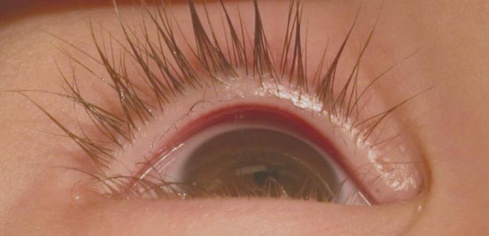
Distichiasis is a congenital or acquired condition where eyelashes arise from the meibomian glands along the posterior lamella of the eyelid margin.[1]
Disease Entity
ICD10CM: Q10.3 (Other congenital malformations of eyelid)
ICD9: 743.63 (Other specified congenital anomalies of eyelid)
Eyelid Anatomy
Eyelashes: There are approximately 100 cilia on each upper eyelid, and 50 on the lower eyelids.[2] The eye lash roots are located on the anterior surface of the tarsus. Cilia travel between the orbicularis oculi and the muscle of Riolan, and exit the skin at the anterior lid margin.[3]
Meibomian glands: Meibomian glands are specialized sebaceous glands that line the eyelid margin and secrete lipids that form the superficial layer of the tear film. There are approximately 25 glands in the upper eyelid, and 20 in the lower eyelid.[2] Meibomian glands and eyelashes differentiate from a common pilosebaceous unit during the 2nd month of gestation.[2] The dual potentiality of the pilosebaceous unit explains the etiology of both congenital and acquired forms of distichiasis.
Differential diagnosis
Trichiasis: Misdirected cilia that originate from normal eyelash-bearing area of eyelid. Consider other variants of trichiasis: Tristrichiasis - 3 rows of eyelashes and Tetrastrichiasis - 4 rows of eyelashes.[4]
Drug-induced eyelash trichomegaly: drug-related increase in the eyelashes' length with associated curling.[4]
Entropion: Inward rotation of the eyelid margin.
Congenital Distichiasis
Congenital distichiasis is a rare condition that occurs when a primary epithelium germ cell destined to differentiate into the meibomian glands of the tarsus develops into a complete (cilia and glandular structure) pilosebaceous unit.[3]
Signs
Partial or complete second row of lashes emerging at or slightly behind the Meibomian gland orifices. The aberrant lashes tend to be thinner and shorter than normal cilia, and are often directed posteriorly towards the globe. The lashes are typically soft and well tolerated during infancy, and typically do not become symptomatic until after the age of 5 years.[3]
Treatment
Treatment is indicated when the patient is symptomatic or if surface keratopathy develops.[2] Conservative therapy includes ocular lubricants and soft contact lenses. Aberrant eyelashes can be removed by epilation, electrolysis, laser ablation[5] and cryotherapy, although patients should be aware that often multiple treatments are necessary.[6] Manual epilation is temporary because eyelashes normally grow back in 2-3 weeks. More definitive surgical treatments include tarsotomy with eyelid margin rotation, direct tarsal excision,[7][8] and lamellar eyelid division with removal of the aberrant follicles.[9][10] Upper eyelid division and cryotherapy involves the following steps:[3][10]
- Incision along grey line to separate the anterior and posterior lamellae.
- Cryotherapy with double freeze thaw cycle to -20 °C to posterior lamellae and lash follicles.
- Surgical reapposition of the anterior and posterior lamellae.
Lymphedema-distichiasis syndrome
Lymphedema-distichiasis syndrome (LDS) is a rare genetic multisystem disorder characterized by the constellation of lower limb lymphedema and distichiasis. The condition inherited in an autosomal dominant pattern, and associated with pathogenic mutations of the Forkhead box protein C2 (FOXC2) transcription factor.[11]
Distichiasis is observed in 94% of affected individuals.[12] The degree of distichiasis can range from a single cilia to a full set of extra eyelashes. 75% of affected individuals have other ocular findings including corneal irritation, recurrent conjunctivitis, and photophobia.[13] Approximately 30% of individuals with LDS have unilateral or bilateral congenital ptosis.[14]
Lower extremity lymphedema is bilateral, confined to the lower limbs without involvement of external genitalia, and typically presents in late childhood or puberty.[11] Other associated systemic abnormalities include early onset varicose veins[13][14] congenital heart defects, cleft palate ± Pierre Robin sequence,[15] renal abnormalities[16], and scoliosis.
Clinical diagnosis for LDS requires either lymphedema and distichiasis, distichiasis and a family history of lower-limb lymphedema, or lower-limb lymphedema and a family history of distichiasis.[11] Identification of a heterozygous pathogenic variant in FOXC2 by a combination of gene-targeted testing and comprehensive genomic testing. The condition has high penetrance, but variable expressivity and severity is highly variable even within the same family.
Acquired distichiasis
Acquired distichiasis is attributed to metaplasia and de-differentiation of the meibomian glands into complete (gland and follicle) producing pilosebaceous units. Intense acute inflammation, trauma, or chronic irritation can cause cilia to develop from meibomian gland openings.[2] Common etiologies of acquired distichiasis include late-stage cicatrizing conjunctivitis associated with chemical injury, Stevens–Johnson syndrome, and ocular cicatricial pemphigoid, chronic rosacea and blepharitis.[17] Some systemic medications more notably: docetaxel, pertuzumab and trastuzumab, were associated with the development of distichiasis.[18]
Signs
Compared to congenital distichiasis the pattern of acquired distichiasis is less uniform with a variable number of lashes originating from each meibomian gland orifice. Also, the cilia tend to be non-pigmented and stunted compared to the normal cilia. Patients with corneal irritation secondary to the eyelashes are typically symptomatic with tearing, foreign body sensation and photophobia.
Treatment
Treatment of mild cases is with lubricating ointment, epilation, and electrolysis. Severe cases require lamellar eyelid division and cryotherapy to the posterior lamella as discussed above. Splitting of the eyelid lamella with cryotherapy was first introduced by Anderson et al. with a series of thirteen eyelids followed for 8-48 months. Seven eyelids (54%) remaining symptom free and six eyelids required either minor cryosurgical touch-ups, or occasional epilation of trichiatic lashes.[10] However, when applied to distichiasis secondary to cicatrizing ocular surface disease, cryotherapy resulted to variably poor outcomes with reports of worsened surface inflammation post-treatment. [19]
Additional Resources
References
- ↑ Scheie, H. G. & Albert, D. M. Distichiasis and trichiasis: origin and management. Am. J. Ophthalmol.61, 718–720 (1966).
- ↑ 2.0 2.1 2.2 2.3 2.4 American Academy of Ophthalmology. 2019-2020 BCSC: Basic and clinical science course. (2019).
- ↑ 3.0 3.1 3.2 3.3 Bowling, B. & Kanski, J. J. Kanski’s clinical ophthalmology: a systematic approach. (Elsevier, 2016).
- ↑ 4.0 4.1 Singh S. Distichiasis: An update on etiology, treatment and outcomes. Indian J Ophthalmol. 2022 Apr;70(4):1100-1106. doi: 10.4103/ijo.IJO_1141_21. PMID: 35325995; PMCID: PMC9240497.
- ↑ Ioannides J, Everson R, Matas Riera M, Dawson C. A description of blend electrolysis for treatment of canine distichiasis: 78 cases (2012-2017) Vet Rec. 2020;187:e102.
- ↑ O’Donnell, B. A. & Collin, J. R. Distichiasis: management with cryotherapy to the posterior lamella. Br J Ophthalmol77, 289–292 (1993).
- ↑ White, J. H. Correction of distichiasis by tarsal resection and mucous membrane grafting. Am. J. Ophthalmol.80, 507–508 (1975).
- ↑ Rozenberg, A., Pokroy, R., Langer, P., Tsumi, E. & Hartstein, M. E. Modified treatment of distichiasis with direct tarsal strip excision without mucosal graft. Orbit37, 341–343 (2018).
- ↑ Vaughn, G. L., Dortzbach, R. K., Sires, B. S. & Lemke, B. N. Eyelid splitting with excision or microhyfrecation for distichiasis. Arch. Ophthalmol.115, 282–284 (1997).
- ↑ 10.0 10.1 10.2 Anderson, R. L. & Harvey, J. T. Lid Splitting and Posterior Lamella Cryosurgery for Congenital and Acquired Distichiasis. Archives of Ophthalmology99, 631–634 (1981).
- ↑ 11.0 11.1 11.2 Mansour, S., Brice, G. W., Jeffery, S. & Mortimer, P. Lymphedema-Distichiasis Syndrome. in GeneReviews®(eds. Adam, M. P. et al.) (University of Washington, Seattle, 2019).
- ↑ Brice, G. et al.Analysis of the phenotypic abnormalities in lymphoedema-distichiasis syndrome in 74 patients with FOXC2 mutations or linkage to 16q24. J. Med. Genet.39, 478–483 (2002).
- ↑ 13.0 13.1 Mellor, R. H. et al.Mutations in FOXC2 are strongly associated with primary valve failure in veins of the lower limb. Circulation115, 1912–1920 (2007).
- ↑ 14.0 14.1 Lyons, O. et al.Human venous valve disease caused by mutations in FOXC2 and GJC2. J. Exp. Med.(2017). doi:10.1084/jem.20160875
- ↑ Papoff, P. et al.Early Mandibular Distraction to Relieve Robin Severe Airway Obstruction in Two Siblings with Lymphedema-Distichiasis Syndrome. J Maxillofac Oral Surg15, 384–389 (2016).
- ↑ Jones, G. E. et al.Renal anomalies and lymphedema distichiasis syndrome. A rare association? Am. J. Med. Genet. A173, 2251–2256 (2017).
- ↑ Khandekar R, Kidiyur S, Al-Raisi A. Distichiasis and dysplastic eyelashes in trachomatous trichiasis cases in Oman: A case series. East Mediterr Health J. 2004;10:192–7.
- ↑ Tas F, Ozturker C. Distichiasis in association with entropion in metastatic HER2-positive breast cancer treated by pertuzumab, trastuzumab, and docetaxel combination chemotherapy.
- ↑ Wood JR, Anderson RL. Complications of cryosurgery. Arch Ophthalmol. 1981;99:460–3.

