Interventional Glaucoma
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Interventional glaucoma describes a proactive treatment approach in which laser procedures, procedural pharmaceuticals, and minimally invasive surgery are used earlier in the disease course rather than after medications have failed. This article reviews the limitations of conventional glaucoma management, the evidence supporting earlier procedural intervention, and a practical framework for applying these principles from ocular hypertension through advanced disease.
Introduction
One of the most significant and evolving developments in the glaucoma field has been the concept of "interventional glaucoma." Interventional glaucoma refers to the management of glaucoma by using procedures early to lower intraocular pressure (IOP).[1] This approach involves proactive rather than reactive intervention to address glaucoma earlier in the disease process, including in both standalone and combination-cataract settings.
Conventionally, glaucoma treatment has followed a stepwise strategy beginning with medical therapy and progressing to surgery only when necessary.[2][3] However, this approach has shown several limitations, including patient adherence, ocular surface toxicity, and decreased quality of life.[4] In response to these challenges, surgical procedures that lower IOP, such as selective laser trabeculoplasty (SLT) and minimally invasive glaucoma surgery (MIGS), have emerged.[5] These procedures allow ophthalmologists to reduce both disease progression and dependence on chronic therapy.
By intervening earlier with minimally invasive surgical, laser, or drug-delivery treatments instead of prolonged topical medications, interventional glaucoma takes the burden of medication compliance off the patient. It also allows for standalone surgical interventions rather than letting cataract surgery dictate the glaucoma treatment plan. This article discusses the limitations of traditional glaucoma management, current practice and limitations of interventional glaucoma, and the rationale for its application in clinical practice.
Rationale for Early Procedural Intervention
Historical Basis for Conventional Glaucoma Management
Traditionally, incisional surgeries for glaucoma were associated with a high risk of postoperative complications, including endophthalmitis and conjunctival hemorrhage.[6] The IOP-lowering capacity of trabeculectomy, tube shunt implants, and the EX-PRESS shunt also declined over time, with 5-year reoperation rates of 15.1%, 14.0%, and 18.3%, respectively.[7][8][9] These limitations made topical medications the more practical option for long-term IOP control. Their effectiveness in lowering IOP, and the ability to adjust dosing incrementally, reinforced their place as first-line therapy.[10] Therefore, medical therapy was prioritized and surgical intervention was reserved for later stages of disease.
Limitations of Conventional Glaucoma Management Associated with Medications
Adherence Challenges
Nonadherence to topical glaucoma therapy is common, with published rates ranging from 30% to 80%, and adherence tends to worsen over time.[4] The causes are multifactorial and include forgetfulness, cognitive and physical impairment, regimen complexity, and difficulty with self-administration.[4] Correctly administering an eye drop requires coordination, dexterity, and adequate vision, skills that decline in the same elderly population most affected by glaucoma. Most patients receive no formal training in drop technique, and most make at least one error during self-administration.[4] Furthermore, between 40% and 75% require two or more medications for adequate IOP control, and each added agent reduces adherence to prior medications while introducing new side effects. [10] Nonadherence at any point in this regimen is directly linked to visual field progression.[11]
Detection of glaucoma presents with its own challenge. Patients who take their drops only in the days before a scheduled visit may appear controlled at each appointment while progressing between them.[4] By the time inadequate control becomes apparent in the clinic, irreversible damage may already have occurred.
Psychosocial and Emotional Considerations
Early procedural intervention offers significant emotional benefits by preserving visual function and alleviating the psychological burden of glaucoma.[12] Glaucoma-related vision loss is associated with depression and reduced confidence in daily activities.[13][14] Furthermore, patients often experience anxiety, low self-image, and reduced trust in healthcare due to feeling a lack of control over their condition.[15]
The reliance on eye drops is consistently linked to lower quality of life and patient dissatisfaction.[16][17][18][19] For instance, a study by Nordmann et al. found that nearly two-thirds of patients experienced local medication side effects, which were associated with reduced satisfaction and lower vision-related quality of life.[18] These issues, compounded by potential mental or physical comorbidities, further diminish the overall well-being of patients under current treatment paradigms.[20]
Consequently, a safe, procedural intervention is a desirable alternative, particularly to reduce the burden of medication. Studies support this, as Al Habash et al. reported that 79% of patients experienced improved quality of life following MIGS procedures.[21] Similarly, Pahljina et al. observed that reducing medication through XEN Gel Stent implantation correlated with better Glaucoma Symptom Scale (GSS) scores.[22] The LiGHT trial confirmed superior GSS scores for SLT over medication, while studies on iStent inject with phacoemulsification showed improved quality of life compared to cataract surgery alone, largely due to reduced medication dependence.[23][24]
Beyond the patient, the ongoing demands of treatment (medications and appointments) impose a significant burden on caregivers and family members, sometimes exceeding the burden of the disease itself.[25] This impact includes financial and psychological stress, as well as limitations in daily activities like driving and reading.[26]
These burdens, combined with a potential lack of motivation to use daily medication for an asymptomatic, long-term disease, can lead to poor compliance in patients with mild glaucoma.[25] Therefore, a patient’s understanding of the risk of irreversible vision loss is crucial when evaluating the benefits of surgical interventions. Research shows patients place an extremely high value on retaining sight, with some indicating they would give up a significant portion of their remaining life to avoid vision impairment.[27]
Ocular Surface Toxicity
Ocular surface disease (OSD) develops in 30% to 70% of patients using topical glaucoma medications, compared to 5% to 30% of the general population.[4] The primary driver is benzalkonium chloride (BAK), a preservative found in approximately 70% of ophthalmic formulations, which causes dose-dependent cytotoxic damage to corneal epithelial, conjunctival, trabecular meshwork, and ciliary epithelial cells.[4] Effects worsen with increasing numbers of medications, higher dosing frequency, and longer treatment duration.
Clinical manifestations include burning, tearing, punctate keratitis, reduced tear break-up time, and elevated Ocular Surface Disease Index (OSDI) scores.[4][28] In patients with well-controlled glaucoma, OSD symptoms may be the only ones experienced as the disease itself is typically silent until late. Fechtner et al. documented significant ocular surface complaints in patients on topical IOP-lowering therapy using validated OSDI scoring, and Ghosh et al. found that both signs and symptoms of OSD were substantially more prevalent in treated patients than in untreated individuals.[28][29] BAK-related surface damage also reduces the success rate of subsequent filtration surgery, meaning years of preserved topical therapy can compromise the conjunctival tissue required for later surgical intervention.[4]
Delayed Escalation
Even when topical medications fail to achieve adequate IOP control, escalation to procedural intervention is frequently delayed. A survey of German ophthalmologists by Vorwerk et al. found that clinicians managing patients with insufficient IOP control most commonly responded by adding or substituting medications rather than referring for surgery.[30] Stürmer and Faschinger observed that patients arriving for filtering surgery typically presented with advanced morphologic damage, far-advanced visual field defects, and decompensated IOP despite maximum tolerated medical therapy (MTMT), a pattern they attributed to systematic underreferral over years of conventional management.[31]
Adding topical medications is the most common escalation step in conventional management, though with real limitations. Adding a third or fourth agent carries only a 14% chance of achieving target IOP at 12 months, and adherence worsens with each additional medication.[4] When further medication is nonetheless appropriate, newer agents including latanoprostene bunod and netarsudil offer mechanisms beyond conventional prostaglandin therapy such as increasing trabecular outflow, reducing episcleral venous pressure, and providing dual-pathway IOP reduction that may yield additional lowering where standard agents have reached their ceiling.[32] Fixed combination formulations reduce bottle count and dosing complexity, which can improve tolerability.[32] These options are reasonable intermediate steps, but the diminishing returns of successive medications should factor into the decision alongside the evidence supporting earlier procedural intervention.[4][33]
Fear of surgical complications, therapeutic inertia, and the continued availability of additional drug classes to try all contribute to this delay. Procedural intervention is often introduced only after substantial irreversible optic nerve damage has occurred.
Role of Procedures
LiGHT Trial
The Laser in Glaucoma and Ocular Hypertension (LiGHT) trial is the most influential evidence base for SLT as a first-line treatment.[23][34] This multicenter randomized controlled trial (RCT) enrolled 718 treatment-naive patients with Open Angle Glaucoma (OAG) or Ocular Hypertension (OHT), randomized to primary SLT versus IOP-lowering eye drops. At 3 years, 74.2% of the SLT-first arm were drop-free, with target IOP achieved at 93% of patient visits and fewer trabeculectomies required than in the drops arm.[34] At 6 years, 72% of SLT-first patients remained drop-free, with sustained clinical and cost-effectiveness over topical therapy.[23] Based on these findings, the United Kingdom's National Institute for Health and Care Excellence (NICE) designated SLT as the preferred first-line treatment for the National Health Service, and both the European Glaucoma Society (EGS) and the American Academy of Ophthalmology (AAO) updated their guidelines accordingly.[4]
However, the effectiveness of SLT does wane. Success rates range from 45% to 87% at 1 year, falling to 18% to 62% at 3 years and approximately 25% at 5 years.[33] In the LiGHT trial, 44.5% of patients not on medications required repeat SLT.[33] Retreatment is an option, but each successive procedure may deliver diminishing benefit.
Minimally Invasive Glaucoma Surgery
MIGS procedures use an ab interno microinvasive approach with minimal tissue trauma, rapid recovery, and a safety profile substantially better than traditional filtration surgery.[33] Serious complications associated with trabeculectomy and tube shunts including hypotony, bleb-related infections, and corneal decompensation are far less common with MIGS.[33]
The iStent trabecular micro-bypass accounted for a negligible share of US glaucoma surgeries at its 2012 release and grew to 43.7% of all glaucoma surgeries by 2017; MIGS now represents the majority of glaucoma procedures performed in the United States.[33] In regions with higher MIGS adoption, trabeculectomy rates have declined.[33]
Multiple reviews support a consistent safety picture. Vinod and Gedde reviewed the safety profile of MIGS across multiple device types and found generally low rates of serious adverse events relative to incisional surgery.[35] A systematic review by Gottehrer and Davey of 30 RCTs found MIGS produces favorable IOP and medication reductions with a safety profile comparable to phacoemulsification alone.[36] Gillmann et al. confirmed an overall favorable safety profile across a decade of MIGS literature, while noting device-specific complication patterns that warrant individualized patient selection.[2]
Cataract Surgery
Phacoemulsification lowers IOP independently of any concurrent glaucoma procedure, though the effect is modest and not reliably durable. Wang et al. found a mean IOP decrease of 1.91 mmHg (8.89%) in eyes with glaucoma after standalone cataract surgery, with greater reductions in eyes with higher baseline pressures.[37] In the Ocular Hypertension Treatment Study (OHTS) cohort, cataract extraction produced a 4.0 mmHg (16.5%) reduction in patients with ocular hypertension, sustained for approximately one year before diminishing over the following two years.[38] However, the larger reduction in the OHTS likely reflects the higher untreated baseline IOP in that population compared to medically controlled glaucoma patients.
For most patients who require active glaucoma management, cataract surgery alone does not provide sufficient IOP control.[39] The timing of lens extraction does create an opportunity to add a MIGS procedure. Addressing both conditions in the same setting often achieves greater IOP reduction and lower medication burden than cataract surgery alone, and in some patients can alter the long-term treatment course.[33]
SLT versus Topical Medications as the Initial Treatment
The clinical case for SLT as first-line therapy has grown substantially over the past decade. A meta-analysis by Li et al. of five prospective comparative trials found no statistically significant difference in IOP reduction or success rates between SLT and topical medications.[40] The LiGHT trial then went further: at 3 years, 74.2% of SLT-first patients were drop-free with IOP at target at 93% of visits, no patient in the SLT arm required glaucoma surgery compared to 11 in the drops arm, and there was a 97% probability that SLT was more cost-effective than drops.[34] At 6 years, SLT-first patients had better GSS scores than the drops group (83.6 versus 81.3), the only quality-of-life measure to diverge between arms, suggesting that the symptomatic burden of long-term drop use accumulates in ways that standardized instruments may underestimate.[23] Organizations including NICE, EGS, and AAO have all updated their guidelines to list SLT as a first-line option.[4] Further, the 2020 AAO Primary Open Angle Glaucoma (POAG) Preferred Practice Pattern recommends treatment escalation whenever there is documented progression, regardless of whether measured IOP appears controlled.[41]
Additionally, the psychosocial dimension of this decision warrants more attention than it typically receives. Glaucoma is asymptomatic in its early and moderate stages, meaning patients experience no direct benefit from treatment, rather only the burden of it.[4] A systematic review and meta-analysis of 45 studies involving almost 5 million subjects found the pooled prevalence of depression in glaucoma patients to be 19% and anxiety to be 25%, both significantly higher than in patients without glaucoma.[42] Longer duration of diagnosis is independently associated with worsening psychological distress and reduced quality of life.[43] Daily preserved drops add medication side effects, dosing complexity, cost, and a persistent daily reminder of a progressive disease that cannot be felt or cured.[4] For many patients this starts on the first day of treatment and continues indefinitely. SLT removes that daily burden for most patients for several years, without preservative exposure or required behavioral change.[23][34]
In a chronic asymptomatic disease where multiple acceptable treatment options exist, shared decision-making is an ethical obligation, not a clinical courtesy.[44] Patients who understand the LiGHT outcomes and the limitations of long-term drop therapy often choose SLT. Katz et al. identified patient education about interventional options as critical to realizing the benefits of the interventional glaucoma approach, noting that this education extends beyond the patient to support networks, optometrists, payers, and policy-makers.[45]
Topical medications remain appropriate when a patient prefers to avoid a procedure, when angle anatomy limits SLT candidacy, when rapid IOP reduction is needed, or when laser access is unavailable.[46][47]The decision should reflect disease stage, surface health, adherence capacity, and what the patient values, but that conversation should happen with the patient is fully informed of both options and their evidence.[44][45]
The Interventional Glaucoma Approach
What distinguishes interventional glaucoma from conventional glaucoma management is not the procedures themselves – laser trabeculoplasty, MIGS, and filtration surgery have existed for decades – but their position in the treatment sequence. In the conventional approach, procedures followed medication failure. In the interventional approach, procedures lead. Medications serve as supplemental or bridge therapy, not anchors. This inversion of the traditional hierarchy is the defining clinical feature of interventional glaucoma.
In 2025, the Interventional Glaucoma Working Group, a group of 10 glaucoma experts committed to interventional glaucoma principles, reached a consensus and published a structured treatment protocol covering OHT through severe glaucoma.[48] The protocol provides the most detailed published clinical framework currently available for applying these principles in practice and forms the basis for much of this section.
Treatment Categories
The interventional glaucoma framework organizes available treatments into categories defined by four patient-centric characteristics:[48]
- the pre-procedure experience
- the procedure-day experience
- the post-procedure experience
- the impact on the patient's ability to undergo future procedures
This practical categorization differs from schemas organized by mechanism of action or outflow pathway. It yields the following groups, ordered from least to most ocular tissue impact:
Laser procedures include SLT, micropulse laser trabeculoplasty (MLT), and direct SLT. These are the preferred starting point in most disease stages because they require no incision, leave all tissue intact, and are repeatable. The LiGHT trial proved that starting with SLT is just as effective as using eye drops for for treatment-naive OAG and OHT.[23][34]
Procedural pharmaceuticals are intracameral sustained-release drug implants that deliver continuous IOP-lowering drug concentrations without depending on patient self-administration. The travoprost intracameral implant (iDose TR), FDA-approved in December 2023, achieved mean IOP reductions of 6.6 to 8.5 mmHg for the commercially marketed slow-eluting implant in Phase 3 randomized trials, with the primary efficacy endpoint evaluated through 3 months demonstrating non-inferiority to twice-daily timolol.[49] The bimatoprost implant (Durysta) provides similar sustained delivery via the same intracameral route.[48] These agents represent a qualitative shift: a pharmaceutical that behaves procedurally, requiring a single office-based administration rather than daily self-dosing.[50]
Tissue-sparing MIGS includes trabecular micro-bypass stents and canaloplasty procedures targeting the trabecular meshwork and schlemm's canal. These preserve the conjunctival tissue required for subsequent subconjunctival filtering surgery. In the HORIZON RCT, 556 patients with mild to moderate POAG undergoing cataract surgery were randomized 2:1 to receive a Hydrus Microstent or cataract surgery alone. At 5 years, 66% of the microstent group were medication-free compared to 46% of the cataract-alone group (P <0.001), 49.5% achieved IOP of 18 mmHg or lower without medications versus 33.8% (P = 0.003), and the microstent group experienced approximately 50% fewer secondary incisional glaucoma surgeries, with no adverse effect on corneal endothelium.[51]
Non-tissue-sparing MIGS includes goniotomy and trabeculotomy, which ablate or remove trabecular meshwork tissue, limiting future trabecular bypass options. These are reserved for when tissue-sparing options have been exhausted.[48]
Filtering surgery including trabeculectomy, tube shunt implantation, and the XEN gel stent achieves the largest IOP reductions and is appropriate when disease severity and the failure of prior interventions warrant it.[48]
Importantly, topical medications remain available throughout all stages of treatment as bridge and supplemental therapy. Maximum tolerated medical therapy, defined as ≥4 classes of IOP-lowering medications or fewer in cases of intolerance, marks the endpoint of medication escalation rather than the starting point of the treatment plan.[48]
Stage-Based Treatment Protocols
The consensus protocol assigns each disease stage its own stepwise treatment progression based on the degree of IOP reduction required and the acceptable risk of limiting future procedural options.[48] Severity categories follow the 2020 AAO POAG Preferred Practice Pattern definitions.[41] Figure 1 below summarizes the consensus protocol and was directly reproduced from Funke et al. 2025 under the CC BY-NC-ND 4.0 open access license.[48]
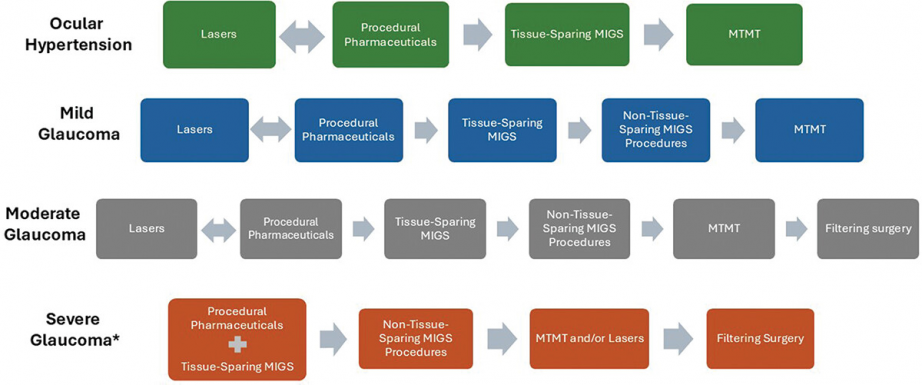
*In certain severe cases of glaucoma according to the surgeons’ discretion, interventions may be completed “out of sequence” compared to the above diagram. This framework is meant to serve as a guideline for the interventional glaucoma approach, but some patients may present with unique clinical characteristics and complexities requiring an individualized treatment plan that may not necessary follow the pathways outline in this diagram.[48]
Ocular Hypertension
Patients with OHT have elevated IOP without glaucomatous optic nerve damage or visual field loss. The OHTS established that IOP reduction significantly reduces conversion risk to POAG, identifying predictive factors including baseline IOP, age, cup-to-disc ratio, and central corneal thickness.[10] The priority at this stage is achieving sufficient IOP reduction while preserving all future procedural options. Non-tissue-sparing procedures and filtering surgery are not appropriate.
The consensus progression is: Lasers → Procedural Pharmaceuticals → Tissue-Sparing MIGS → MTMT (Figure 1).[48]
Mild Glaucoma
Mild glaucoma involves structural change to the optic nerve or retinal nerve fiber layer without visual field loss. IOP reduction is more important than in OHT, and there is modestly greater willingness to accept procedural inconveniences or side effects. Non-tissue-sparing MIGS becomes available when earlier steps have not achieved adequate control. Filtering surgery remains inappropriate at this stage.
The consensus progression is: Lasers → Procedural Pharmaceuticals → Tissue-Sparing MIGS → Non-Tissue-Sparing MIGS → MTMT (Figure 1).[48]
Moderate Glaucoma
Moderate disease (prominent glaucomatous optic nerve damage and visual field loss in one hemifield outside 5 degrees of fixation) requires target IOP in the 12 to 15 mmHg range, and the degree of damage is sufficient to justify the risks of filtering surgery if all other options have failed.[48]
The protocol mirrors that of mild glaucoma with filtering surgery added as a final step: Lasers → Procedural Pharmaceuticals → Tissue-Sparing MIGS → Non-Tissue-Sparing MIGS → MTMT → Filtering Surgery (Figure 1).[48]
Severe Glaucoma
Severe disease (advanced glaucomatous optic nerve damage and visual field loss in two hemifields or within 5 degrees of fixation) demands the most aggressive IOP reduction available. Optic nerve reserve is minimal and the sequential step-by-step approach used in earlier stages is not appropriate here. SLT is not first-line given the need for more rapid and substantial IOP reduction.[48]
The consensus protocol initiates both a procedural pharmaceutical and a tissue-sparing MIGS procedure simultaneously as the first step, providing immediate dual-mechanism IOP reduction from the outset.[48] If IOP remains uncontrolled, non-tissue-sparing MIGS follows, then MTMT with or without SLT, then filtering surgery. In cases requiring urgent IOP reduction, direct escalation to incisional surgery may be appropriate regardless of the standard sequence (Figure 1).[48]
The Multi-Mechanism and Multi-Modal Approach
A central principle in interventional glaucoma involves combining procedures that target different mechanisms of aqueous outflow rather than cycling through single interventions in sequence. Micheletti et al. described this as a multi-mechanism and multi-modal approach, using SLT, procedural pharmaceuticals, and MIGS together or in close succession to target different outflow pathways simultaneously.[52]
Trabecular bypass stents address resistance at the trabecular meshwork and schlemm's canal. Procedural pharmaceuticals such as iDose TR reduce IOP via the uveoscleral pathway.[49] SLT stimulates trabecular meshwork cell activity, reducing trabecular resistance.[23] Combining these modalities targets multiple resistance sites at once, which may explain why combined approaches achieve lower IOPs than single interventions alone.[52]
This framework also supports earlier and more definitive intervention. Two precisely targeted procedures may achieve what three or four medications cannot, with better adherence, less surface toxicity, and more consistent 24-hour pressure control, a meaningful advantage given that IOP fluctuation between medication doses is a recognized driver of glaucomatous progression.[33][52]
Differences from Traditional Management
The practical differences between interventional and conventional management operate at every stage of the treatment pathway. In conventional management, medications are first-line and procedures follow failure. Surgery is reactive and intervention timing is driven by what has already failed as previously discussed.
In interventional management, procedures come first. Medications bridge and supplement. Surgery is planned based on disease stage. Intervention timing is driven by what the disease needs now.[48]
Yadgarov et al. described this specifically in the context of procedural pharmaceuticals: rather than a last resort, these represent early, reliable, adherence-independent IOP control that can be combined with other interventions for a multi-modal approach from the start of treatment.[50] The goal at each step is to achieve the target IOP with the minimum burden while preserving the maximum number of future procedural options.
For patients already on multiple medications, the interventional framework asks a different clinical question than conventional care does. Rather than asking what can be added to the current regimen, it asks whether the current regimen is actually achieving what it should, and whether years of preserved topical therapy have compromised the ocular surface or future surgical options in the process.[48] These are different questions and they often lead to different decisions.
Long-term Treatment Planning and Sequencing
Target IOP is assessed regularly, with Sihota et al. recommending reassessment every 6 to 12 months based on optic nerve stability and visual field change.[46][41] When a patient progresses despite apparently controlled IOP, the target should be lowered and treatment intensified regardless of measured IOP.[41][53] When a patient has remained stable for years with mild disease and faces competing medical priorities or limited life expectancy, treatment burden may reasonably be reduced.[41]
Patients move through treatment categories over time. When repeat SLT has been attempted and is no longer producing adequate IOP control, transition to procedural pharmaceuticals or tissue-sparing MIGS is appropriate. Prolonged use of preserved topical therapy in the interim compromises the conjunctival tissue that filtration surgery requires, reinforcing the value of earlier procedural transition.[4][33] A patient who has completed tissue-sparing MIGS but continues to progress is a candidate for non-tissue-sparing MIGS or, in moderate-to-severe disease, filtering surgery, while conjunctival tissue largely remains intact.[33][48]
The sequencing is individualized. The interventional principle is consistent: use the most appropriate intervention for the current disease stage, preserve future options, and do not defer effective treatment while the disease advances.[48]
Management in Patients with Prior Interventions
When initial SLT has been effective but its IOP-lowering effect has diminished, repeat SLT is a reasonable next step before escalating to medications or surgery. A systematic review and meta-analysis by Jang et al. found repeat SLT to be effective and safe for up to 24 months in patients with POAG.[54] Because SLT selectively targets pigmented trabecular meshwork cells without causing structural scarring, the meshwork remains susceptible to retreatment.[54] Success rates may be lower than initial SLT, and repeat treatment is best approached as a bridge rather than a reliable long-term solution.[33]
However, when initial SLT has waned and repeat SLT is no longer producing adequate response, the question shifts from retreatment to transition. Patients with declining and shorter-lasting SLT responses are better served by procedural pharmaceuticals or tissue-sparing MIGS than by further laser.[33] The trabecular meshwork tolerates repeated SLT, but this tolerance is not unlimited, and continued retreatment past the point of adequate response delays a more appropriate next step.[33][54]
Patients presenting on chronic medical therapy with progressive OSD, ongoing visual field loss, and inadequate IOP control are a common and predictable product of a treatment approach that reserved procedures for late-stage disease. Several signals warrant earlier transition to procedural management: persistent IOP above target despite maximum tolerated medical therapy; progressive OSD limiting tolerability or forcing dose reductions; documented visual field or structural progression.[41][47] Years of preserved topical therapy progressively compromise the conjunctival tissue that filtering surgery requires. Earlier transition to procedural management preserves that option before it is foreclosed.[4]
Role of Filtration Surgery in Advanced Disease
Filtration surgery remains the most effective means of achieving very low IOPs when severe disease demands it.[41] The interventional approach does not eliminate filtering surgery from the algorithm. It repositions it where it belongs: filtering surgery is reached only after earlier stages have been appropriately used, with better-preserved conjunctival tissue and a more favorable operative environment.[4][48]
Patients who arrive at filtration surgery after years of preserved topical therapy may have compromised conjunctiva that reduces surgical success rates. Earlier procedural management reduces this risk. The failure rates of filtering surgery (30.7% by 3 years and 46.9% by 5 years) and the associated reoperation rates of 15.1% for trabeculectomy and 14.0% for tube shunts at five years are not reasons to avoid it when indicated.[7][9][33] They are reasons to manage disease proactively enough that when filtering surgery is needed, it is performed under better conditions.
Patient-Centered Considerations
Glaucoma management spans decades, and patient priorities change. A 55-year-old may prioritize medication freedom and active management; that same patient at 75, with significant competing medical problems, may reasonably prefer lighter monitoring and fewer procedures. Treatment decisions across this timeline require ongoing shared decision-making rather than a plan set once at diagnosis.[42][43]
Bhartiya describes shared decision-making as an ethical obligation in glaucoma care: in a chronic asymptomatic disease where multiple acceptable treatment arms exist, patient centricity is a clinical responsibility, not a courtesy.[44] Katz et al. found that realizing the benefits of the interventional approach depends on patients genuinely understanding their options, requiring consistent education across every clinical encounter from the optometrist to the glaucoma specialist.[45]
Approximately 19% of glaucoma patients develop depression and 25% develop anxiety, both substantially higher than in the general population, with longer duration of diagnosis independently associated with worsening psychological distress and reduced quality of life.[42][43]An approach that reduces daily medication demands, removes adherence as a constant variable, and gives patients a sense of active management rather than passive monitoring may reduce this burden even when IOP outcomes are equivalent.[44][45]
The goal is not to perform more procedures. It is to match the treatment to the disease proactively, transparently, and with the patient as a participant in that decision.[1][45]
Limitations and Ongoing Discussion
Longevity of IOP Control Following SLT and MIGS
SLT does not provide permanent IOP control. Success rates decline from 45% to 87% at one year to approximately 25% at five years, and 44.5% of LiGHT trial patients not on medications required repeat SLT during the study period.[23][33] Repeat SLT is effective for up to 24 months but produces progressively shorter and weaker responses with successive treatments.[54]
Long-term MIGS data are more limited and vary by device. The HORIZON trial provides five-year outcomes for the Hydrus microstent with maintained results.[51] Many other MIGS devices lack data extending beyond two years in pivotal trials, and durability cannot be assumed to apply uniformly across device categories.[2] For procedural pharmaceuticals, iDose TR Phase 3 primary efficacy data extend through three months.[49] Longer-term real-world effectiveness for this class remains to be established.
Variability in IOP Reduction Across Procedures
IOP reduction is not consistent across patients or procedures. Baseline IOP, angle anatomy, trabecular meshwork pigmentation, prior treatment history, and individual variation in outflow dynamics all influence response.[33] Gottehrer and Davey's systematic review of 30 RCTs found favorable overall outcomes for MIGS but meaningful variability across individual studies.[36] Gillmann et al. identified device-specific complication patterns that require individualized patient and procedure selection.[2] Patients should be counseled that outcomes cannot be guaranteed and that escalation may follow even a technically successful procedure.
Limited Evidence Base and Variation by Clinical Practice Setting
The interventional glaucoma approach as an integrated clinical philosophy has not been tested in a dedicated large randomized trial comparing systematic early procedural intervention against conventional stepwise medical management across a diverse population. The LiGHT trial supports SLT as first-line over drops for OAG and OHT.[23][34] Individual MIGS clinical trials support specific devices.[51] The Funke et al. 2025 consensus protocol represents expert opinion from 10 clinicians, not data from a RCT, and by the authors' own acknowledgment is a snapshot of the treatment landscape as of late 2024, subject to revision as evidence evolves.[48]
Many MIGS pivotal trials enrolled patients in combined cataract-surgery settings, making it difficult to isolate the MIGS contribution from the IOP effect of lens extraction.[33] Trial populations typically exclude patients with prior surgery, advanced disease, and significant comorbidities, limiting generalizability to the broader clinical population.[36]
Nevertheless, the evidence base for interventional glaucoma is substantive and continues to grow. It strongly challenges the traditional glaucoma treatment paradigm and the limitations of the approach may be viewed as opportunities for further investigation and forward-thinking progress.[48] Ultimately, the primary objective of glaucoma care is to preserve the patient’s vision and quality of life, necessitating the use of all available therapeutic options. The best patient-care considers a proactive and patient-focused treatment approach for glaucoma.
Cost, Access, and External Considerations
SLT is cost-effective relative to topical medications. The LiGHT trial demonstrated a 97% probability that SLT is more cost-effective than drops at a willingness-to-pay threshold of £20,000 per QALY.[34] There is strong evidence on the overall cost-effectiveness of MIGS and SLT procedures.[55][56][57][58][59] Higher disease severity (possibly due to lack of timely intervention) is associated with higher direct costs and ophthalmology-related resource consumption, including medications, ophthalmology visits, and glaucoma surgeries.[60][61] That said cost-effectiveness for MIGS varies by device, setting, and degree of sustained medication reduction achieved.[3] Further, procedural pharmaceuticals may introduce higher upfront costs and long-term cost-effectiveness for this class has not been established in published trials.[49]
Importantly, access to these therapies is not uniform. SLT requires laser equipment and trained operators. MIGS and procedural pharmaceuticals require specialized surgical training and, in the United States, often specific reimbursement structures. Many standalone MIGS procedures lack insurance coverage outside the combined cataract-surgery context, effectively restricting access to patients whose lens timing and glaucoma needs happen to align.[33][62] The interventional glaucoma approach as currently practiced primarily benefits patients with access to subspecialty glaucoma care. Expanded training, advocacy for standalone procedure reimbursement, and integration into underserved settings are necessary steps toward equitable access.
From the patient's perspective, the fear of undergoing a procedure is also an important consideration, as there may be understandable hesitation around undergoing surgery rather than self-administering an eyedrop.[12] However, modern, MIGS treatments help ease these concerns. Typically, glaucoma procedures are often completed alongside cataract surgery, thus addressing two ocular concerns in a single visit to the operating room. Secondly, if cataract surgery isn't required, patients may find comfort in the high safety profile of minimally invasive lasers, MIGS, and procedural drugs, particularly when weighed against the common side effects of daily eye drops.
References
- ↑ 1.0 1.1 De Francesco T, Bacharach J, Smith O, Shah M. Early diagnostics and interventional glaucoma. Ther Adv Ophthalmol. 2024;16:25158414241287431. doi:10.1177/25158414241287431
- ↑ 2.0 2.1 2.2 2.3 Gillmann K, Baudouin C, Masood I, et al. A Systematic and Narrative Review of Safety and Complications in Minimally Invasive Glaucoma Surgery (MIGS) Between 2014–2024. Clin Ophthalmol. 2026;20:564425. doi:10.2147/OPTH.S564425
- ↑ 3.0 3.1 Bedrood S, Berdahl J, Sheybani A, Singh IP. Alternatives to Topical Glaucoma Medication for Glaucoma Management. Clin Ophthalmol. 2023;17:3899-3913. doi:10.2147/OPTH.S439457
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 4.12 4.13 4.14 4.15 4.16 4.17 Radcliffe NM, Shah M, Samuelson TW. Challenging the “Topical Medications-First” Approach to Glaucoma: A Treatment Paradigm in Evolution. Ophthalmol Ther. 2023;12(6):2823-2839. doi:10.1007/s40123-023-00831-9
- ↑ Richter GM, Coleman AL. Minimally invasive glaucoma surgery: current status and future prospects. Clin Ophthalmol. 2016;10:189-206. doi:10.2147/OPTH.S80490
- ↑ Wagner IV, Stewart MW, Dorairaj SK. Updates on the Diagnosis and Management of Glaucoma. Mayo Clin Proc Innov Qual Outcomes. 2022;6(6):618-635. doi:10.1016/j.mayocpiqo.2022.09.007
- ↑ 7.0 7.1 Gedde SJ, Herndon LW, Brandt JD, et al. Postoperative complications in the Tube Versus Trabeculectomy (TVT) study during five years of follow-up. Am J Ophthalmol. 2012;153(5):804-814.e1. doi:10.1016/j.ajo.2011.10.024
- ↑ Lee DA, Higginbotham EJ. Glaucoma and its treatment: a review. Am J Health Syst Pharm. 2005;62(7):691-699. doi:10.1093/ajhp/62.7.691
- ↑ 9.0 9.1 Craven ER, Singh IP, Yu TM, Rhoten S, Sadruddin OR, Sheybani A. Reoperation Rates and Disease Costs for Primary Open-Angle Glaucoma Patients in the United States Treated with Incisional Glaucoma Surgery. Ophthalmol Glaucoma. 2022;5(3):297-305. doi:10.1016/j.ogla.2021.10.011
- ↑ 10.0 10.1 10.2 Kass MA, Heuer DK, Higginbotham EJ, et al. The Ocular Hypertension Treatment Study: a randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open-angle glaucoma. Arch Ophthalmol. 2002;120(6):701-713; discussion 829-830. doi:10.1001/archopht.120.6.701
- ↑ Rossi GC, Pasinetti GM, Scudeller L, Radaelli R, Bianchi PE. Do adherence rates and glaucomatous visual field progression correlate?. Eur J Ophthalmol. 2011;21(4):410-414. doi:10.5301/EJO.2010.6112
- ↑ 12.0 12.1 Gallardo, M., Smith, O., Trubnik, V., & Reiss, G. (2024). Interventional glaucoma and the patient perspective. Expert Review of Ophthalmology, 19(5), 311–318. https://doi.org/10.1080/17469899.2024.2382149
- ↑ Prager AJ, Liebmann JM, Cioffi GA, et al. Self-reported function, health resource use, and total health care costs among medicare beneficiaries with glaucoma. JAMA Ophthalmol. 2016 Apr;134(4):357–365. doi: 10.1001/jamaophthalmol.2015.5479
- ↑ Nelson P, Aspinall P, O’Brien C. Patients’ perception of visual impairment in glaucoma: a pilot study. Br J Ophthalmol. 1999 May;83(5):546–552. doi: 10.1136/bjo.83.5.546
- ↑ Chan EW, Chiang PP, Liao J, et al. Glaucoma and associated visual acuity and field loss significantly affect glaucoma-specific psychosocial functioning. Ophthalmology. 2015 Mar;122(3):494–501. doi: 10.1016/j.ophtha.2014.09.030
- ↑ Rossi GC, Tinelli C, Pasinetti GM, et al. Dry eye syndrome-related quality of life in glaucoma patients. Eur J Ophthalmol. 2009 Jul;19(4):572–579. doi: 10.1177/112067210901900409
- ↑ Skalicky SE, Goldberg I, McCluskey P. Ocular surface disease and quality of life in patients with glaucoma. Am J Ophthalmol. 2012 Jan;153(1):1–9 e2. doi: 10.1016/j.ajo.2011.05.033
- ↑ 18.0 18.1 Nordmann JP, Auzanneau N, Ricard S, et al. Vision related quality of life and topical glaucoma treatment side effects. Health Qual Life Outcomes. 2003 Dec 10;1(1):75. doi: 10.1186/1477-7525-1-75
- ↑ Stalmans I, Lemij H, Clarke J, et al. Signs and symptoms of ocular surface disease: the reasons for patient dissatisfaction with glaucoma treatments. Clin Ophthalmol. 2020;14:3675–3680. doi: 10.2147/OPTH.S269586
- ↑ Serbin M, Devine B, Campbell J, et al. Assessing health care burden in glaucoma patients with and without physical or mental comorbidities. J Manag Care Spec Pharm. 2020 Mar;26(3):325–331. doi: 10.18553/jmcp.2020.26.3.325
- ↑ Al Habash A, Nagshbandi AA. Quality of life after combined cataract and minimally invasive glaucoma surgery in glaucoma patients. Clin Ophthalmol. 2020;14:3049–3056. doi: 10.2147/OPTH.S276124
- ↑ Pahljina C, Sarny S, Hoeflechner L, et al. Glaucoma medication and quality of life after phacoemulsification combined with a xen gel stent. J Clin Med. 2022 Jun 15;11(12):3450. doi: 10.3390/jcm11123450
- ↑ 23.0 23.1 23.2 23.3 23.4 23.5 23.6 23.7 23.8 Gazzard G, Konstantakopoulou E, Garway-Heath D, et al. Laser in Glaucoma and Ocular Hypertension (LiGHT) Trial: Six-Year Results of Primary Selective Laser Trabeculoplasty versus Eye Drops for the Treatment of Glaucoma and Ocular Hypertension. Ophthalmology. 2023;130(2):139-151. doi:10.1016/j.ophtha.2022.09.009
- ↑ Samuelson TW, Singh IP, Williamson BK, et al. Quality of life in primary open-angle glaucoma and cataract: an analysis of VFQ-25 and OSDI from the iStent inject(R) pivotal trial. Am J Ophthalmol. 2021 Sep;229:220–229. doi: 10.1016/j.ajo.2021.03.007
- ↑ 25.0 25.1 Stagg BC, Granger A, Guetterman TC, et al. The burden of caring for and treating glaucoma: the patient perspective. Ophthalmol Glaucoma. 2022 Jan;5(1):32–39. doi: 10.1016/j.ogla.2021.04.011
- ↑ Sotimehin AE, Ramulu PY. Measuring disability in glaucoma. J Glaucoma. 2018 Nov;27(11):939–949. doi: 10.1097/IJG.0000000000001068
- ↑ Jampel HD, Schwartz A, Pollack I, et al. Glaucoma patients’ assessment of their visual function and quality of life. J Glaucoma. 2002 Apr;11(2):154–163. doi: 10.1097/00061198-200204000-00012
- ↑ 28.0 28.1 Fechtner RD, Godfrey DG, Budenz D, Stewart JA, Stewart WC, Jasek MC. Prevalence of ocular surface complaints in patients with glaucoma using topical intraocular pressure-lowering medications. Cornea. 2010;29(6):618-621. doi:10.1097/ICO.0b013e3181c325b2
- ↑ Ghosh S, O'Hare F, Lamoureux E, Vajpayee RB, Crowston JG. Prevalence of signs and symptoms of ocular surface disease in individuals treated and not treated with glaucoma medication. Clin Exp Ophthalmol. 2012;40(7):675-681. doi:10.1111/j.1442-9071.2012.02781.x
- ↑ Vorwerk C, Thelen U, Buchholz P, Kimmich F. Treatment of glaucoma patients with insufficient intraocular pressure control: a survey of German ophthalmologists in private practice. Curr Med Res Opin. 2008;24(5):1295-1301. doi:10.1185/030079908x291976
- ↑ Stürmer JPE, Faschinger C. Operieren wir die Glaukome zu spät? [Do We Perform Glaucoma Surgery too Late?]. Klin Monbl Augenheilkd. 2018;235(11):1269-1277. doi:10.1055/s-0043-115902
- ↑ 32.0 32.1 Mehran NA, Sinha S, Razeghinejad R. New glaucoma medications: latanoprostene bunod, netarsudil, and fixed combination netarsudil-latanoprost. Eye (Lond). 2020;34(1):72-88. doi:10.1038/s41433-019-0671-0
- ↑ 33.00 33.01 33.02 33.03 33.04 33.05 33.06 33.07 33.08 33.09 33.10 33.11 33.12 33.13 33.14 33.15 33.16 33.17 33.18 Micheletti JM, Brink M, Brubaker JW, Ristvedt D, Sarkisian SR. Standalone interventional glaucoma: evolution from the combination-cataract paradigm. J Cataract Refract Surg. 2024;50(12):1284-1290. doi:10.1097/j.jcrs.0000000000001537
- ↑ 34.0 34.1 34.2 34.3 34.4 34.5 34.6 Gazzard G, Konstantakopoulou E, Garway-Heath D, et al. Selective laser trabeculoplasty versus eye drops for first-line treatment of ocular hypertension and glaucoma (LiGHT): a multicentre randomised controlled trial. Lancet. 2019;393(10180):1505-1516. doi:10.1016/S0140-6736(18)32213-X
- ↑ Vinod K, Gedde SJ. Safety profile of minimally invasive glaucoma surgery. Curr Opin Ophthalmol. 2021;32(2):160-168. doi:10.1097/ICU.0000000000000731
- ↑ 36.0 36.1 36.2 Gottehrer J, Davey PG. The Efficacy and Safety of Minimally Invasive Glaucoma Surgery for Primary Open-Angle Glaucoma: A Systematic Review. Healthcare (Basel). 2026;14(3):319. Published 2026 Jan 27. doi:10.3390/healthcare14030319
- ↑ Wang SY, Azad AD, Lin SC, Hernandez-Boussard T, Pershing S. Intraocular Pressure Changes after Cataract Surgery in Patients with and without Glaucoma: An Informatics-Based Approach. Ophthalmol Glaucoma. 2020;3(5):343-349. doi:10.1016/j.ogla.2020.06.002
- ↑ Mansberger SL, Gordon MO, Jampel H, et al. Reduction in intraocular pressure after cataract extraction: the Ocular Hypertension Treatment Study. Ophthalmology. 2012;119(9):1826-1831. doi:10.1016/j.ophtha.2012.02.050
- ↑ Ling JD, Bell NP. Role of Cataract Surgery in the Management of Glaucoma. Int Ophthalmol Clin. 2018;58(3):87-100. doi:10.1097/IIO.0000000000000234
- ↑ Li X, Wang W, Zhang X. Meta-analysis of selective laser trabeculoplasty versus topical medication in the treatment of open-angle glaucoma. BMC Ophthalmol. 2015;15:107. Published 2015 Aug 19. doi:10.1186/s12886-015-0091-2
- ↑ 41.0 41.1 41.2 41.3 41.4 41.5 41.6 Gedde SJ, Vinod K, Wright MM, et al. Primary Open-Angle Glaucoma Preferred Practice Pattern®. Ophthalmology. 2021;128(1):P71-P150. doi:10.1016/j.ophtha.2020.10.022
- ↑ 42.0 42.1 42.2 Groff ML, Choi B, Lin T, Mcllraith I, Hutnik C, Malvankar-Mehta MS. Anxiety, depression, and sleep-related outcomes of glaucoma patients: systematic review and meta-analysis. Can J Ophthalmol. 2023;58(4):346-355. doi:10.1016/j.jcjo.2022.02.010
- ↑ 43.0 43.1 43.2 Kopilaš V, Kopilaš M. Quality of life and mental health status of glaucoma patients. Front Med (Lausanne). 2024;11:1402604. Published 2024 Jun 3. doi:10.3389/fmed.2024.1402604
- ↑ 44.0 44.1 44.2 44.3 Bhartiya S. Patient Centricity and the Ethics of Glaucoma Care. J Curr Glaucoma Pract. 2020;14(2):68-71. doi:10.5005/jp-journals-10078-1281
- ↑ 45.0 45.1 45.2 45.3 45.4 Katz LJ, Myers JS, Herndon LW, Kresch YS, Hengerer FH. Interventional Glaucoma: Improving the Patient-Provider Educational Exchange. Clin Ophthalmol. 2024;18:3365-3374. Published 2024 Nov 21. doi:10.2147/OPTH.S491287
- ↑ 46.0 46.1 Sihota R, Angmo D, Ramaswamy D, Dada T. Simplifying "target" intraocular pressure for different stages of primary open-angle glaucoma and primary angle-closure glaucoma. Indian J Ophthalmol. 2018;66(4):495-505. doi:10.4103/ijo.IJO_1130_17
- ↑ 47.0 47.1 Zhang X, Vadoothker S, Munir WM, Saeedi O. Ocular Surface Disease and Glaucoma Medications: A Clinical Approach. Eye Contact Lens. 2019;45(1):11-18. doi:10.1097/ICL.0000000000000544
- ↑ 48.00 48.01 48.02 48.03 48.04 48.05 48.06 48.07 48.08 48.09 48.10 48.11 48.12 48.13 48.14 48.15 48.16 48.17 48.18 48.19 48.20 48.21 48.22 Funke CM, Ristvedt D, Yadgarov A, Micheletti JM. Interventional glaucoma consensus treatment protocol. Expert Rev Ophthalmol. 2025;20(2):79-87. https://doi.org/10.1080/17469899.2025.2465330
- ↑ 49.0 49.1 49.2 49.3 Sarkisian SR Jr, Ang RE, Lee AM, et al. Phase 3 Randomized Clinical Trial of the Safety and Efficacy of Travoprost Intraocular Implant in Patients with Open-Angle Glaucoma or Ocular Hypertension. Ophthalmology. 2024;131(9):1021-1032. doi:10.1016/j.ophtha.2024.02.022
- ↑ 50.0 50.1 Yadgarov A, Provencher L, Shafer B, Funke C. Adopting Interventional Glaucoma Via Sustained-Release Therapies: The Wide-Ranging Impact of Procedural Pharmaceuticals in Ophthalmology. Ophthalmol Ther. 2024;13(11):2825-2838. doi:10.1007/s40123-024-01041-7
- ↑ 51.0 51.1 51.2 Ahmed IIK, De Francesco T, Rhee D, et al. Long-term Outcomes from the HORIZON Randomized Trial for a Schlemm's Canal Microstent in Combination Cataract and Glaucoma Surgery. Ophthalmology. 2022;129(7):742-751. doi:10.1016/j.ophtha.2022.02.021
- ↑ 52.0 52.1 52.2 Micheletti JM, Shultz M, Singh IP, Samuelson TW. An Emerging Multi-mechanism and Multi-modal Approach in Interventional Glaucoma Therapy. Ophthalmol Ther. 2025;14(1):13-22. doi:10.1007/s40123-024-01073-z
- ↑ Villasana GA, Bradley C, Ramulu P, Unberath M, Yohannan J. The Effect of Achieving Target Intraocular Pressure on Visual Field Worsening. Ophthalmology. 2022;129(1):35-44. doi:10.1016/j.ophtha.2021.08.025
- ↑ 54.0 54.1 54.2 54.3 Jang HJ, Yu B, Hodge W, Malvankar-Mehta MS. Repeat Selective Laser Trabeculoplasty for Glaucoma Patients: A Systematic Review and Meta-analysis. J Curr Glaucoma Pract. 2021;15(3):117-124. doi:10.5005/jp-journals-10078-1302
- ↑ Ngan K, Fraser E, Buller S, et al. A cost minimisation analysis comparing iStent accompanying cataract surgery and selective laser trabeculoplasty versus topical glaucoma medications in a public healthcare setting in New Zealand. Graefes Arch Clin Exp Ophthalmol. 2018 Nov;256(11):2181–2189. doi: 10.1007/s00417-018-4104-8
- ↑ Patel V, Ahmed I, Podbielski D, et al. Cost-effectiveness analysis of standalone trabecular micro-bypass stents in patients with mild-to-moderate open-angle glaucoma in Canada. J Med Econ. 2019 Apr;22(4):390–401. doi: 10.1080/13696998.2019.1572013
- ↑ Berdahl JP, Khatana AK, Katz LJ, et al. Cost-comparison of two trabecular micro-bypass stents versus selective laser trabeculoplasty or medications only for intraocular pressure control for patients with open-angle glaucoma. J Med Econ. 2017 Jul;20(7):760–766. doi: 10.1080/13696998.2017.1327439
- ↑ Yong MH, Che Hamzah J. Selective laser trabeculoplasty vs. topical medications for step-up treatment in primary open angle glaucoma: comparing clinical effectiveness, quality of life and cost-effectiveness. Med J Malaysia. 2020 Jul;75(4):342–348.
- ↑ Ruiz-Lozano RE, Alamillo-Velazquez J, Ortiz-Morales G, et al. Selective laser trabeculoplasty is safe and effective in patients previously treated with prostaglandin analogs: An evidence-based review. Int Ophthalmol. 2023 Feb;43(2):677–695. doi: 10.1007/s10792-022-02460-w
- ↑ Patel AR, Schwartz GF, Campbell JH, et al. Economic and Clinical Burden Associated With Intensification of Glaucoma Topical Therapy: A US Claims-based Analysis. J Glaucoma. 2021 Mar 1;30(3):242–250. doi: 10.1097/IJG.0000000000001730
- ↑ Lee PP, Walt JG, Doyle JJ, et al. A multicenter, retrospective pilot study of resource use and costs associated with severity of disease in glaucoma. Arch Ophthalmol. 2006 Jan;124(1):12–19. doi: 10.1001/archopht.124.1.12
- ↑ Radcliffe N. The case for standalone micro-invasive glaucoma surgery: rethinking the role of surgery in the glaucoma treatment paradigm. Curr Opin Ophthalmol. 2023;34(2):138-145. doi:10.1097/ICU.0000000000000927

