Lacrimal Gland Botulinum Toxin Injection for Epiphora Management
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Introduction
Epiphora (tearing) is the presence of an excessively watering eye[1]. It is a chronic condition that affects as many as 14% of the population over 40 years of age and is a commonly reported problem at ophthalmology clinic visits.[1] [2]
Epiphora is subdivided into two broad categories based on etiology: functional and non-functional epiphora. Functional epiphora refers to dysfunction of the lacrimal drainage system in the presence of anatomical patency resulting in hypersecretion from the lacrimal gland.[3] In contrast, non-functional epiphora refers to decreased tear outflow within the lacrimal drainage system due to anatomical obstruction or other causes of decreased outflow.[3] However, functional and non-functional epiphora are not mutually exclusive, and the disorder may arise from any alteration to the balance between tear inflow and outflow.
Current epiphora management includes medical therapy and surgical options. Medical management of epiphora includes systemic anticholinergics or observation with concurrent dry eye treatment, but these methods can have various systemic side effects or minimal reduction in symptoms, respectively.[1][3][4] Surgical management of epiphora aims to relieve canalicular obstruction in the lacrimal system and is dependent on location and etiology of the obstruction.[4] Surgical success rates are high but varied as these procedures are associated with a higher risk of postoperative complications and need for extensive follow-up.[4][5][6] In addition, some patients with epiphora may not be ideal surgical candidates. Thus, non-surgical options, such as the botulinum toxin injection of the lacrimal gland, are of interest and provide an alternative for management of epiphora.
Botulinum toxin description
Botulinum toxin refers to a group of neurotoxins produced by the bacteria Clostridium botulinum (C. botulinum).[7] Historically, C. botulinum was associated with causing fatal, paralytic disease from foodborne, wound, and infant botulism.[7][8] However, identification and purification of botulinum toxin has allowed its use to safely treat a variety of medical conditions.[7] Dr. Alan Scott, an ophthalmologist, helped pioneer the use of botulinum toxin as a medical treatment in the 1970s.[7][8] Since then, botulinum toxin use in ophthalmology has demonstrated treatment efficacy for strabismus, blepharospasm, hemifacial spasm, and other oculomotor disorders.[7] The use of botulinum toxin as a potential treatment for epiphora in humans was reported in 1998 by Boroojerdi, who injected botulinum toxin A into the lacrimal gland of two patients with hyperlacrimation due to aberrant facial nerve regeneration.[9] The treatment resulted in near complete recovery of symptoms with only transient focal side effects.[9]
Mechanism of action of botulinum toxin
There are seven serologically distinct toxins (A, B, C1-2, D, E, F, and G) produced by C. botulinum.[7] These serotypes appear to be structurally similar to each other and consist of a large (heavy, H) subunit and a small (light, L) subunit linked by at least one disulfide bridge.[7] However, botulinum toxin A is the most potent and widely used formulation for cosmetic and therapeutic treatment.[10]
The toxin acts by inhibiting the presynaptic release of the neurotransmitter acetylcholine at the neuromuscular junction, resulting in muscular weakening and paralysis.[7][10] In treating epiphora, botulinum toxin inactivates acetylcholine release from postganglionic parasympathetic secretomotor fibers necessary for tear secretion from the lacrimal gland, resulting in decreased tearing.[11][12]
Indications
Clinical trials using botulinum toxin have shown this agent is effective in treatment of:[3][12]
- Functional epiphora due to:
- Paroxysmal lacrimal hypersecretion
- Aberrant facial nerve regeneration
- Facial palsies
- Gustatory epiphora
- Submandibular gland transplant complicated by epiphora
- Non-functional epiphora due to:
- Canalicular obstruction
- Nasolacrimal duct stenosis
- Post-punctal cautery
However, there is a need to formulate clear guidelines of specific indications for the use of botulinum toxin in epiphora management.[12]
Contraindications
Although botulinum toxin has a strong safety profile, contraindications to its use include:[10][12]
- Neuromuscular disorders (e.g. ALS, Myasthenia Gravis, Eaton-Lambert Syndrome)
- Allergy to botulinum toxin
- Pregnancy
- Breastfeeding
Technique Description
Forms
There are different types of commercially available botulinum toxin used for epiphora management (Table 1).[3][10] Although there is no consensus as to which type of botulinum toxin A to use, most studies have reported using onabotulinum toxin A for treatment of epiphora.[3]
| Table 1: Botulinum Toxin-A formulations used for epiphora management | ||||
| Commercial Name | Molecule | Laboratory | Bioequivalence (U) | No. U per vial |
| Botox | Onabotulinum toxin A | Allergan | 1 | 100 U |
| Dysport | Abobotulinum toxin A | Ispen Ltd | 3-5 | 500 U |
| Xeomin | Incobotulinum toxin A | MERZ Pharma | 1 | 100 U |
Doses
There is no consensus regarding the dosing of botulinum toxin injection for epiphora management. The respective dose is quantified in units (U). Dosing of botulinum toxin injections for epiphora range from 1.25U to 15U per treatment. [3]The most frequently used doses to administer are between 2.5U to 5U.[3]
Reconstitution
Botulinum toxin is commercially available as a lyophilized powder in 50U or 100 U vials that must be reconstituted with balanced salt solution diluent for clinical use.[10][12] The concentration of botulinum toxin per 0.1 mL depends upon the volume of diluent used. Two mL of balanced salt solution are injected into a bottle containing 50-100U of botulinum toxin to obtain 5U per 0.1 mL. Reconstituted botulinum toxin should be transparent and free of particulate matter.[12] It should be stored in a refrigerator at 4 degrees Celsius and used within 6 hours after reconstitution.[12]
Anesthesia
The procedure is typically performed under topical anesthesia. Anesthetic eye drops are instilled in the eye and applied on sterile cotton swabs that are placed in the area designated for injection.
Materials
The materials required for the procedure include:
- Botulinum toxin bottle
- Blepharostat (optional)
- 2 mL and 1 mL syringe
- 27 or 30 gauge needle
- Topical anesthesia
Injection Technique
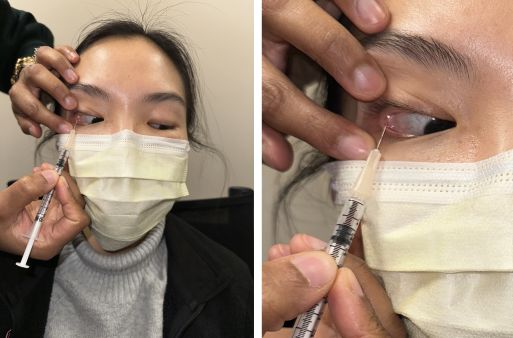
Injection of botulinum toxin into the lacrimal gland is an office-based procedure performed via the transconjunctival or transcutaneous approach (Figure 1).[13] The transconjunctival approach is most common and allows direct visualization of the palpebral lobe of the lacrimal gland, leading to a decreased risk of ptosis post-injection.
[13] Disadvantages of this approach include the need for eyelid retraction and/or eyelid inversion and an increased risk of subconjunctival hemorrhage.[3][13] The transcutaneous approach involves injecting the orbital lobe of the lacrimal gland through the upper lid skin, which does not allow direct visualization of the gland.[13]
The following steps demonstrate proper transconjunctival injection technique:
- Apply topical anesthesia to the ocular surface.
- Ensure asepsis of the surgical field.
- Retract and/or elevate the eyelid to expose the palpebral lobe of the lacrimal gland.
- Ask the patient to look down and away from the site of injection.
- Using a 30 (up to 33) gauge needle mounted on a 1 mL syringe, slowly inject into the palpebral lobe of the lacrimal gland
Post-injection care and follow-up
Post-injection care after is minimal as botulinum toxin injection is generally well-tolerated. Topical lubricants are advised for 2 weeks post-injection.
Patients and caregivers should be reminded of the potential adverse effects associated with botulinum toxin injection (e.g. ptosis, diplopia, etc.) and the need for long-term follow-up and repeat injections for continued management of epiphora.[3][10][11]
The effect of botulinum toxin is reversible and usually begins to subside after a few months because of the collateral sprouting of nerve endings in muscles.[13] Duration of effects for botulinum toxin injection ranges from three to six months.[3][10] Follow-up is recommended within a few weeks after the first injection to observe early treatment effects and after a few months to re-evaluate symptom control and need for re-injection.
Complications
Botulinum toxin injection is generally well-tolerated, but up to 21% of patients experience at least one side effect related to glandular botulinum toxin injections.[3] Adverse effects from botulinum toxin injection arise from inadvertent weakening of adjacent ocular muscles from toxin diffusion or injury to surrounding structures during the injection process .[3][10] These effects are dose-dependent and typically transient, with most resolving within eight weeks post-injection.[3] Patients and caregivers should be informed of potential complications prior to injection.
The most commonly reported adverse effects from botulinum toxin injection for epiphora include ptosis (13%) and diplopia (6%).[3][10][12] Less common side effects include dry eye syndrome (1-3%), eyelid or lacrimal gland hematoma (1-2%), subconjunctival hemorrhage, esotropia (3%), papillary conjunctivitis (1%), and bleeding.[3][12] Rarer complications from botulinum toxin injection may include infection, globe rupture, and allergic reaction to the toxin..[3][10]
Outcomes
Botulinum toxin injection is efficacious in treating both functional and non-functional epiphora. A recent 2022 review including 14 studies within the past 20 years demonstrated glandular botulinum toxin injection to be a safe and viable treatment option for both functional and non-functional epiphora, with subjective improvement ranging from 67 to 100%.[3] These studies show no significant differences in efficacy and side effects between patients with functional and non-functional epiphora treated with botulinum toxin injection.[3][14][15] There also appears to be no significant difference in efficacy between the transconjunctival and transcutaneous approaches of injection,[13] although a previous meta-analysis demonstrated the transconjunctival approach was associated with fewer complications as compared to the transcutaneous approach.[16]
Conclusions
Botulinum toxin injection presents a safe, minimally invasive, low-cost, and effective treatment option for epiphora. It is a viable alternative for patients who are unable to undergo surgery or who have failed symptom control with existing medical and surgical management. However, more extensive trials are needed to further evaluate the efficacy of botulinum toxin and establish standard indications and a routine treatment regimen for management of epiphora.
- ↑ 1.0 1.1 1.2 Patel J, Levin A, Patel BC. Epiphora. [Updated 2022 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK557449/
- ↑ Dalgleish R. Idiopathic acquired lacrimal drainage obstruction. Br J Ophthalmol. 1967 Jul;51(7):463-8. doi: 10.1136/bjo.51.7.463. PMID: 6029232; PMCID: PMC506426.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 3.16 3.17 3.18 Jeffers J, Lucarelli K, Akella S, Setabutr P, Wojno TH, Aakalu V. Lacrimal gland botulinum toxin injection for epiphora management. Orbit. 2022 Apr;41(2):150-161. doi: 10.1080/01676830.2021.1966810. Epub 2021 Aug 15. PMID: 34396904; PMCID: PMC8844302.
- ↑ 4.0 4.1 4.2 Kanski's Synopsis of Clinical Ophthalmology, 4th edition Chapter 3 Lacrimal Drainage System, 49-57
- ↑ Ullrich K, Malhotra R, Patel BC. Dacryocystorhinostomy. [Updated 2022 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK557851/
- ↑ Eisenbach N, Karni O, Sela E, Nemet A, Dror A, Levy E, Kassif Y, Ovadya R, Ronen O, Marshak T. Conjunctivodacryocystorhinostomy (CDCR) success rates and complications in endoscopic vs non-endoscopic approaches: a systematic review. Int Forum Allergy Rhinol. 2021 Feb;11(2):174-194. doi: 10.1002/alr.22668. Epub 2020 Aug 6. PMID: 32761875.
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 Melling, J., Hambleton, P. & Shone, C. Clostridium botulinum toxins: nature and preparation for clinical use. Eye 2, 16–23 (1988). https://doi.org/10.1038/eye.1988.5
- ↑ 8.0 8.1 Erbguth FJ. Historical notes on botulism, Clostridium botulinum, botulinum toxin, and the idea of the therapeutic use of the toxin. Mov Disord. 2004 Mar;19 Suppl 8:S2-6. doi: 10.1002/mds.20003. PMID: 15027048.
- ↑ 9.0 9.1 Boroojerdi B, Ferbert A, Schwarz M, Herath H, Noth J. Botulinum toxin treatment of synkinesia and hyperlacrimation after facial palsy. J Neurol Neurosurg Psychiatry. 1998 Jul;65(1):111-4. doi: 10.1136/jnnp.65.1.111. PMID: 9667571; PMCID: PMC2170154.
- ↑ 10.0 10.1 10.2 10.3 10.4 10.5 10.6 10.7 10.8 10.9 Padda IS, Tadi P. Botulinum Toxin. [Updated 2022 Nov 30]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK557387/
- ↑ 11.0 11.1 Whittaker KW, Matthews BN, Fitt AW, Sandramouli S. The use of botulinum toxin A in the treatment of functional epiphora. Orbit. 2003 Sep;22(3):193-8. doi: 10.1076/orbi.22.3.193.15622. PMID: 12868028.
- ↑ 12.0 12.1 12.2 12.3 12.4 12.5 12.6 12.7 12.8 Singh S, Ali MJ, Paulsen F. A review on use of botulinum toxin for intractable lacrimal drainage disorders. Int Ophthalmol. 2018 Oct;38(5):2233-2238. doi: 10.1007/s10792-017-0661-9. Epub 2017 Aug 1. PMID: 28766277.
- ↑ 13.0 13.1 13.2 13.3 13.4 13.5 Lee AG, Lee SH, Jang M, Lee SJ, Shin HJ. Transconjunctival versus Transcutaneous Injection of Botulinum Toxin into the Lacrimal Gland to Reduce Lacrimal Production: A Randomized Controlled Trial. Toxins (Basel). 2021 Jan 21;13(2):77. doi: 10.3390/toxins13020077. PMID: 33494380; PMCID: PMC7911887.
- ↑ Singh S, Nair AG, Alam MS, Mukherjee B. Outcomes of lacrimal gland injection of botulinum toxin in functional versus nonfunctional epiphora. Oman J Ophthalmol. 2019 May-Aug;12(2):104-107. doi: 10.4103/ojo.OJO_52_2018. PMID: 31198296; PMCID: PMC6561039.
- ↑ Wojno TH. Results of lacrimal gland botulinum toxin injection for epiphora in lacrimal obstruction and gustatory tearing. Ophthalmic Plast Reconstr Surg. 2011 Mar-Apr;27(2):119-21. doi: 10.1097/IOP.0b013e318201d1d3. PMID: 21224749.
- ↑ Falzon K, Galea M, Cunniffe G, Logan P. Transconjunctival botulinum toxin offers an effective, safe and repeatable method to treat gustatory lacrimation. Br J Ophthalmol. 2010 Mar;94(3):379-80. doi: 10.1136/bjo.2008.155887. PMID: 20215376.