Leber Hereditary Optic Neuropathy
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Leber hereditary optic neuropathy (LHON) is the most common inherited mitochondrial disorder. It usually begins as a unilateral progressive optic neuropathy, with sequential involvement of the fellow eye months or years later.[1] Treatment options are limited, but include the use of antioxidant supplements. Gene therapy trials are currently underway.
Individuals eventually diagnosed with LHON may initially be asymptomatic or experience mild blurring of the central visual field of one eye. Males are more likely to be affected than females, with some reported ratios as high as 9:1. The age of onset is typically between 10 and 30 years, though onset in the seventh or eighth decades has been noted. Symptoms may progress from mild unilateral visual loss to severe bilateral visual loss, usually worse than 20/200.
Visual field testing typically shows a worsening central or cecocentral scotoma. The examination may initially reveal normal or pseudo-edematous optic nerves, with subsequent optic nerve pallor and atrophy. Patients may have concomitant conditions, including cardiac conduction abnormalities, dystonia, and multiple sclerosis–like illnesses. This is considered "LHON Plus" disease. Few patients recover their vision once it is lost, but visual loss rarely progresses to no light perception.
Etiology
LHON is classically associated with mitochondrial base pair mutations G11778A (guanine to adenine at position 11778), T14484C (tyrosine to cytosine), and G3460A. These mutations primarily affect respiratory chain complex I genes and the mitochondrial genes ND1, ND4, and ND6, among others.
The G11778A mutation is the most common, reported to account for 70% of cases in Northern European populations and 90% of cases in Asian populations. It is associated with more severe disease, with less chance of significant visual recovery. The T14484C mutation is associated with the best overall prognosis, with some visual recovery in 37%–58% of cases. This typically occurs after reaching a visual "nadir." This mutation variant has been seen in French Canadian populations with LHON.[2][3] The G3460A mutation usually portends a more intermediate course and has the worst prognosis.[2][3]
Many mitochondrial diseases display heteroplasmy. Each cell contains numerous mitochondria, and in 10%–15% of patients with LHON mitochondrial mutations only a certain percentage of these mitochondria may have mutant DNA. Those with a "mutation load" less than a certain threshold (60%–75%) may not exhibit a disease phenotype and may never lose vision. It has been posited that due to the heteroplasmy seen in LHON, the right and left eyes may have different amounts of affected mitochondrial DNA.[4]
Pathophysiology
Mitochondrial mutations cause defects in several NADH-ubiquinone oxidoreductase chains. This is thought to impair glutamate transport and increase reactive oxygen species production in retinal ganglion cells via apoptosis, though it is unclear why these cells are particularly vulnerable. Atrophy and demyelination are subsequently noted in the optic nerves, chiasm, and tracts.
In acute LHON, macular changes precede peripapillary retinal nerve fiber layer swelling. The acute thickening of these layers is then followed by long-term thinning. It has been suggested that this process is due to a state of pseudohypoxia, followed by a compensatory response. Optical coherence tomography angiography findings in patients with LHON have shown vascular dilation and tortuosity, which may also be seen clinically. [5]
Risk Factors
It is unknown why males are preferentially affected (up to 80% of patients); theories include the presence of a "susceptibility locus" on the Y chromosome. Otherwise, neither age nor lifestyle choices have been shown to increase LHON incidence. One study found that patients with late-onset LHON had significantly higher cumulative tobacco and alcohol consumption than unaffected LHON mutation carriers,[6] suggesting that these factors may in fact worsen disease severity once a person develops LHON.
Diagnosis
LHON must be distinguished from other causes of optic neuropathy, as the treatment course may vary significantly.
History
Individuals typically present with unilateral, slowly progressive, painless visual blurring in the second to fourth decade of life, though the peak age of onset for all cases is the second and third decades of life.[7] Studies have found the average age of onset to be 22–24 years in patients with the G11778A mutation and about 20 years in patients with the T14484C mutation.[8][9][10]
Signs and symptoms of LHON are often confused with those of optic neuritis, except for the lack of pain. Some patients may discover visual loss by incidentally covering the other eye, or by noting increasing difficulty with day-to-day tasks; other patients may present with LHON symptoms after the involvement of the second eye has begun (weeks to months later) and may thus complain of bilateral visual loss. The longest interval reported between first and second eye involvement is 18 years. Other patients may note color or light desaturation. History of concurrent, LHON Plus–related neurologic or cardiac dysfunction should be elicited, including dystonia, tremor, neuropathy, movement disorders, weakness, nonspecific myopathy, and arrhythmias. Leigh syndrome may also be associated with LHON.[10]
A history of worsening headaches, trauma, nutritional deficits (alcohol abuse), previous demyelinating disease, cancer, infiltrative orbital or lung disease, sexually transmitted disease, recent travel, or disease exposure may identify other causes of optic neuropathy. A family history of visual loss in maternal family members may be present.
Physical Examination
Visual acuity may be mildly reduced in the early stages of LHON, but can later progress to the counting fingers stage in one or both eyes. An afferent pupillary defect may be noted when disease activity is asymmetric, often in the interval between the presentation of unilateral visual loss and progression to the fellow eye. Color testing reveals decreased red-green discrimination. Extraocular motility is typically intact, and confrontation visual fields may show intact peripheral vision. However, central or cecocentral scotomata may be described and more plainly delineated with formal visual field testing (Humphrey or Goldmann). In patients presenting with unilateral symptoms, formal visual field testing may display subclinical visual deficits in the fellow, seemingly unaffected eye. Reduced contrast sensitivity and subnormal electroretinograms may also be present.
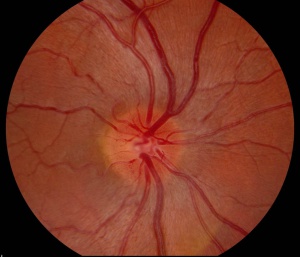
Slit lamp examination of the anterior segment is usually normal. Dilated fundus examination may be normal, or reveal hyperemic, pseudo-edematous optic nerves with peripapillary telangiectasias. Tortuosity of retinal arterioles may also be present (Figure) Though the optic nerve heads appear elevated and edematous, they do not display leakage on fluorescein angiogram, as opposed to those affected by inflammatory disease.
Clinical Diagnosis
Clinical suspicion for LHON may be heightened in the setting of a known family history of mitochondrial disease. The appearance of characteristic optic nerve changes, as described above, may lead to a tentative LHON diagnosis, but this can be confirmed with genetic testing. Late-phase LHON may display optic atrophy or nerve head pallor, which may be indistinguishable from other causes of optic neuropathy.
Diagnostic Procedures
A fluorescein angiogram may be performed to rule out true optic nerve edema. No dye leakage is noted along the borders of an otherwise swollen-appearing optic nerve head. Optical coherence tomography of the optic nerve may show elevation in the initial phases of the disease or atrophy in the later phases.
Laboratory Testing and Imaging
Based on patient history and physical examination, other causes of optic neuropathy should be excluded, especially in those with no family history of LHON. This may include laboratory serum testing of vitamin B12, folate, rapid plasma reagin, fluorescent treponemal antibody absorption, antinuclear antibody, Lyme titers, angiotensin-converting enzyme, and/or purified protein derivative.
Magnetic resonance imaging (MRI) is especially recommended to rule out demyelinating disease and compressive lesions. Ptients with LHON may have increased T2 signal in the optic nerves, chiasm, and tracts, so the orbital fat-suppressed contrast-enhanced MRI may show optic nerve enhancement similar to optic neuritis, as well as chiasmal enlargement and enhancement. Thus, optic nerve enhancement and enlargement of the optic chiasm may be detected on MRI in LHON. In appropriate clinical scenarios, MRI findings should not dissuade physicians from including LHON in the differential diagnosis of acute optic neuropathy.
If other causes of optic neuropathy are comfortably excluded, gene testing can be performed via targeted mutation analysis. Ninety percent of patients with LHON patients harbor one of the three most common mutations, as listed above. Identification of the mutations may help predict disease prognosis.
Management
Management of this condition is primarily supportive, with an early introduction to visual aids and occupational therapy.
Medical Therapy
There is no established medical treatment for LHON, though there is a theoretical benefit from using antioxidants to help reduce the neurotoxic stress due to reactive oxygen species. Multiple supplements such as vitamins B12 and C, coenzyme-Q10, brimonidine, and lutein have shown minimal benefit but may be recommended. Patients are cautioned to avoid neurotoxins such as tobacco and alcohol, as these may increase oxidative stress.
Idebenone, a short-chain synthetic benzoquinone, has undergone clinical testing for the treatment of several neurologic diseases. The recently completed RHODOS (Rescue of Hereditary Optic Disease Outpatient Study) was a double-blind, randomized, placebo-controlled trial of idebenone for LHON. Eighty-five patients with one of the three primary LHON mutations and onset of symptoms within the last 5years were randomized to receive idebenone 300 mg 3 times daily or placebo for 24 weeks. Though the study did not reach its primary endpoint (best recovery in visual acuity), a post hoc analysis revealed improvement in visual acuity in patients with discordant vision between eyes. Those with the G11778A and G3460A mutations seemed to derive greater benefit from idebenone. The drug was also found to be well tolerated. The results have been deemed promising, but have limited utility.
EPI-743, another drug in development, may have more potency than idebenone, but as yet no clinical trials have been performed. Gene therapy techniques have advanced, including allotopic expression and nuclear transfer.
The introduction of exogenous DNA into the mitochondrial genome with mitochondrial targeting of viral vectors is also promising. Recently, researchers conducted a phase 1 trial using intravitreal injection to deliver genetically modified adeno-associated viral vectors with the ND4 gene to affected eyes in 15 patients with LHON with the G11778A mutation. This therapy appeared to be safe and well-tolerated, with mostly mild intraocular inflammation. Another study of 5 patients with the G11778A mutation demonstrated similar results with respect to safety as well as some improvement in certain patients. However, it should be noted that these are the preliminary results of ongoing trials.[5][11] [12]
Medical Follow-Up and Genetic Considerations
Patients may be followed with serial visual acuity, visual field, contrast sensitivity, and color testing. Low-vision aids should be provided early. Genetic counseling is recommended for affected individuals and maternal family members. Male patients may be reassured that they cannot transmit the disease to their offspring. The mother of an affected individual likely harbors the mitochondrial DNA mutation, but may or may not develop symptoms. Female patients will transmit the mutation to all offspring. However, those who exhibit heteroplasmy may transmit a low mutation load; therefore, offspring may have a low risk of disease. De novo mutations are rare but may be present in individuals whose maternal relatives do not have a history of visual loss.
Additional Resources
- International Foundation for Optic Nerve Disease. Accessed May 5, 2025. https://www.ifond.org.
- North American Neuro-Ophthalmology Society (NANOS). Leber hereditary optic neuropathy patient information brochure. Accessed May 5, 2025. https://www.nanosweb.org/LHON patient information brochure.
References
- Lam BL, Feuer WJ, Abukhalil F, et al. Leber hereditary optic neuropathy gene therapy clinical trial recruitment: year 1. Arch Ophthalmol. 2010 ;128(9):1129-1135.
- Klopstock T, Yu-Wai-Man P, Dimitriadis K, et al. A randomized placebo-controlled trial of idebenone in Leber's hereditary optic neuropathy. Brain. 2011;134(Pt 9):2677-2686.
- Newman NJ. Treatment of Leber hereditary optic neuropathy. Brain. 2011;134(Pt 9):2447-2450.
- Palace J. Multiple sclerosis associated with Leber's hereditary optic neuropathy. J Neurol Sci. 2009;286(1-2):24-27.
- Peragallo JH, Newman NJ. Is there treatment for Leber hereditary optic neuropathy? Curr Opin Ophthalmol. 2015;26(6):450-457.
- Van Westen D, Hammar B, Bynke G. Magnetic resonance findings in the pregeniculate visual pathways in Leber hereditary optic neuropathy. J Neuroophthalmol. 2011;31(1):48-51.
- Vaphiades MS. Magnetic resonance findings in the pregeniculate visual pathways in Leber hereditary optic neuropathy. J Neuroophthalmol. 2011;31(2):194.
- Phillips PH, Vaphiades M, Glasier CM, et al. Chiasmal enlargement and optic nerve enhancement on magnetic resonance imaging in Leber hereditary optic neuropathy. Arch Ophthalmol. 2003;121(4):577-579.
- Vaphiades MS, Phillips PH, Turbin RE. Optic nerve and chiasmal enhancement in Leber hereditary optic neuropathy. J Neuroophthalmol. 2003;23(1):104-105.
- Vaphiades MS, Newman NJ. Optic nerve enhancement in Leber hereditary optic neuropathy: four years later. J Neuroophthalmol. 2002;22(1):66-67.
- ↑ Yen MT, Lam BL. Update on hereditary optic neuropathy. Semin Ophthalmol. 1999;14(2):74-80.
- ↑ 2.0 2.1 Mackey DA, Oostra RJ, Rosenberg T, et al. Primary pathogenic mtDNA mutations in multigeneration pedigrees with Leber hereditary optic neuropathy. Am J Hum Genet. 1996;59(2):481-485.
- ↑ 3.0 3.1 Jia X, Li S, Xiao X, et al. Molecular epidemiology of mtDNA mutations in 903 Chinese families suspected with Leber hereditary optic neuropathy. J Hum Genet. 2006;51(10):851-856.
- ↑ Ohden KL, Tang PH, Lilley CC, et al. Atypical Leber hereditary optic neuropathy: 18 year interval between eyes. J Neuroophthalmol. 2016;36(3):304.
- ↑ 5.0 5.1 Jurkute N, Yu-Wai-Man P. Leber hereditary optic neuropathy: bridging the translational gap. Curr Opin Ophthalmol. 2017;28(5):403-409.
- ↑ Dimitriadis K, Leonhardt M, Yu-Wai-Man P, et al. Leber's hereditary optic neuropathy with late disease onset: clinical and molecular characteristics of 20 patients. Orphanet J Rare Dis. 2014;9:158.
- ↑ Yu-Wai-Man P, Chinnery PF. Leber Hereditary Optic Neuropathy. GeneReviews; 2007. Updated March 11, 2021. Accessed May 6, 2025. http://www.ncbi.nlm.nih.gov/books/NBK1174.
- ↑ Harding AE, Sweeney MG, Govan GG, et al. Pedigree analysis in Leber hereditary optic neuropathy families with a pathogenic mtDNA mutation. Am J Hum Genet. 1995;57(1):77-86.
- ↑ Macmillan C, Kirkham T, Fu K, et al. Pedigree analysis of French Canadian families with T14484C Leber's hereditary optic neuropathy. Neurology. 1998;50(2):417-422.
- ↑ 10.0 10.1 Yu-Wai-Man P, Griffiths PG, Brown DT. The epidemiology of Leber hereditary optic neuropathy in the North East of England. Am J Hum Genet. 2003;72(2):333-339.
- ↑ Feuer WJ, Schiffman JC, Davis JL, et al. Gene therapy for Leber hereditary optic neuropathy: initial results. Ophthalmology. 2016;123(3):558-570.
- ↑ Bouquet C, Vignal Clermont C, Galy A, et al. Immune response and intraocular inflammation in patients with Leber hereditary optic neuropathy treated with intravitreal injection of recombinant adeno-associated virus 2 carrying the ND4 gene: a secondary analysis of a phase 1/2 clinical trial. JAMA Ophthalmol. 2019;137(4):399-406.