Optic Atrophy
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Optic atrophy refers to the death of the retinal ganglion cell axons that comprise the optic nerve, and manifests on fundoscopy as a pale optic nerve. Optic atrophy is the end stage of myriad causes of optic nerve damage anywhere along the path from the retina to the lateral geniculate. Since the optic nerve transmits retinal information to the brain, optic atrophy is associated with vision loss. However, the term "atrophy" is somewhat of a misnomer as it implies disuse; thus, this optic nerve damage is better termed "optic neuropathy."
Etiology
Anything that can compromise ganglion cell function can cause optic atrophy (and more broadly optic neuropathy) over time. Optic atrophy can occur due to damage within the eye (glaucoma, optic neuritis, papilledema, tumor, trauma, etc.) along the path of the optic nerve to the brain, or it can be congenital (Leber’s hereditary optic atrophy, autosomal dominant optic atrophy, etc.).
Risk Factors
Risk factors run the gamut from increased IOP (glaucoma), to ischemia, compression (tumors), inflammation, and infection.
General Pathology
The optic nerve is a bundle of 1.2 million axons of retinal ganglion cells that carries visual information from the retina to the brain. It is myelinated by oligodendrocytes that do not regenerate after damage. In optic nerve atrophy, there is a loss of axons and shrinkage of myelin leading to gliosis and widening of the optic cup.
Primary Prevention
Optic atrophy is the end stage of a process causing damage to the optic nerve. It is currently not possible to return function (regrow axons) to an atrophic optic nerve; the best option is to stabilize whatever function remains. The goal of primary prevention to prevent the loss of axons in the first place.
Diagnosis
Since the optic nerve is the conduit for information from the retina to the brain, a damaged optic nerve will result in vision loss. Subtle damage might not affect acuity but may lead to a loss of contrast or color vision. Severe damage may run the gamut from legal blindness to no light perception. Damage to a part of the optic nerve results in loss of vision in the corresponding visual field. Occasionally, if the process causing damage is removed before apoptosis occurs (e.g., removal of a pituitary tumor compressing the chiasm or reducing inflammation in a sarcoid), some improvement in visual function may be noted. A complete diagnosis is based on optic nerve appearance and tests of visual function (visual field, contrast, color, acuity), as well as identifying the causative factor of the damage and ruling out other causes for vision loss (such as retinal causes).
Certain disc appearances can help to determine the cause of the optic nerve damage. Sector disc pallor in an older individual could have been caused by nonarteritic ischemic optic neuropathy. Severe optic atrophy with gliosis could have been due to giant cell arteritis. Damage from papilledema may leave retinal folds and sometimes glistening bodies in the optic nerve head. Cupping is suggestive of glaucoma.
Optical coherence tomography has become a valuable tool for verifying the status of the nerve fiber layer/ganglion axons. Quantification of the nerve fiber layer height and comparison with normative data can document axon loss and differentiate between optic neuropathy and retinal disease as a cause of vision loss.
History
A full patient history is critical in the diagnosis of optic atrophy, since the physician needs to know how the eye arrived at this juncture. This medical history should include a list of all medications, time course of vision loss, and associated symptoms.
Physical Examination
A complete eye exam including visual field, assessing color and contrast vision, intraocular pressures, looking for afferent pupil defect, and fundoscopy should be done.
Signs
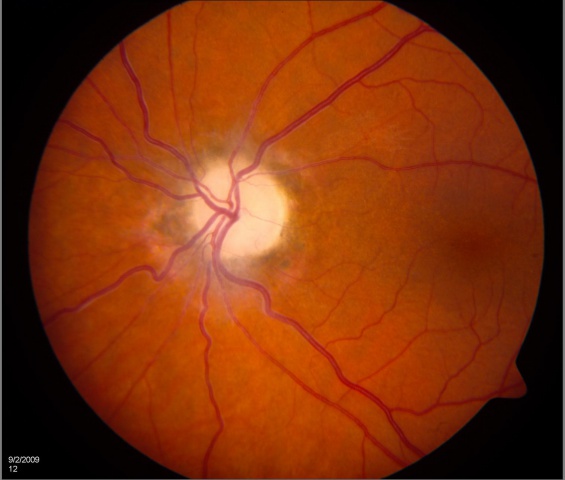
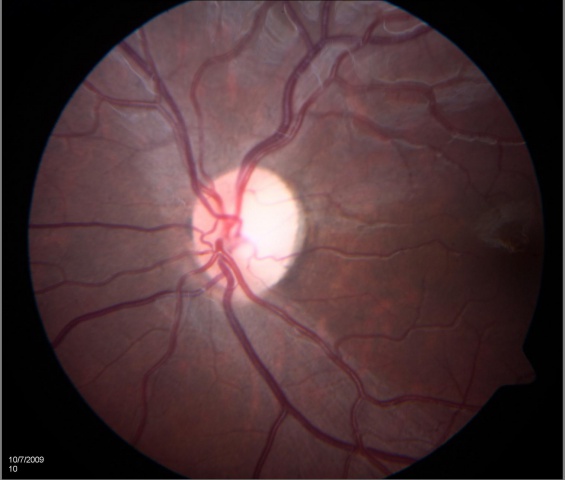
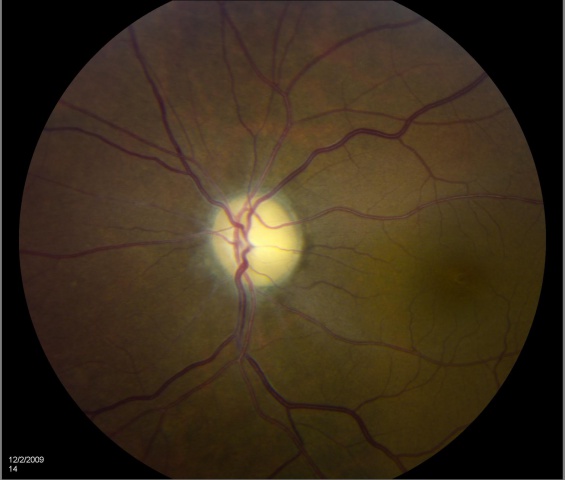
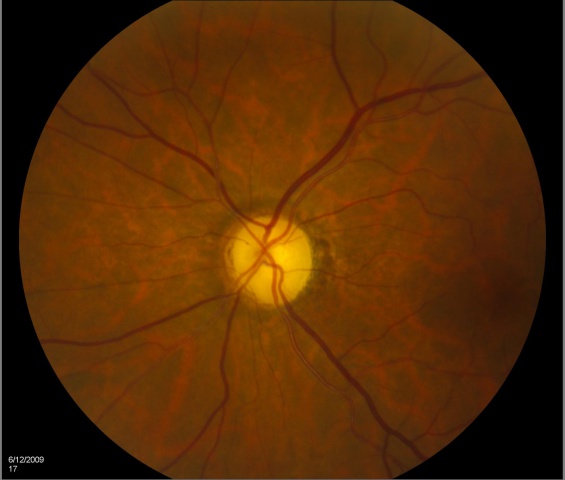
Optic atrophy is a sign and is typically noted as optic nerve pallor. This is the end stage of a process resulting in optic nerve damage. Because the optic nerve fiber layer is thinned or absent, the disc margins appear sharp and the disc is pale, probably reflecting absence of small vessels in the disc head.
EXAMPLES OF OPTIC ATROPHY (NEUROPATHY):
Symptoms
The main symptom of optic atrophy is vision loss. Any other symptoms are attributable to the underlying process that caused the disc damage (such as pain with angle-closure glaucoma).
Clinical Diagnosis
Optic atrophy is usually not difficult to diagnose due to its characteristic pale optic disc, but the cause may be difficult to ascertain. Sometimes it may be hard to differentiate between subtle optic neuropathy and disease of the retina (or both). Electrophysiology and OCT can be helpful for to assessing the thickness of the nerve fiber layers.
Characteristic visual field patterns include papillomacular defect (cecocentral scotoma), arcuate defect (include altitudinal), and/or temporal wedge defect (nasal fibers) for pre=chiasmal lesions, bitemporal (superior) field defects for chiasmal lesions, and hemianopsia for post-chiasmal lesions.
The following workup should be considered for patients presenting with unexplained optic atrophy:
- Check for afferent pupil
- Visual fields 30-2, color vision
- MRI of brain and orbit with contrast
- CT with contrast (check bony disease, sinuses)
- Blood pressure and cardiovascular health (carotids, etc.)
- Serum glucose
Diagnostic Procedures
- Visual field testing (Humphrey 30-2, Tangent Screen) to help localize the location of the lesion
- Optical coherence tomography to assess the thickness of peripapillary retinal nerve fiber layers and/or ganglion cell layers
- Electroretinography to rule out retinal disease.
- Neuroimaging (MRI, CT) to assess for tumors, bone growth, sinusitis, fractures, multiple sclerosis, and infections
Laboratory Tests
If history or examination are suggestive, it may be useful to screen for the following: heavy metals, vitamin B12, folate, VDRL (syphilis), antinuclear antibodies, homocysteine, angiotensin-converting enzymes, and antiphospholipid antibodies. [1] If the patient is or could be pregnant, a TORCH panel could be considered as well.
Differential Diagnosis
Optic atrophy is not usually difficult to diagnose, but it might be confused with optic nerve hypoplasia, myelinated nerve fibers, myopic or scleral crescent, or tilted disc.
The causes for optic atrophy include:
- Compressive – secondary to papilledema, tumor, bony growth (fibrous dysplasia, osteopetrosis), thyroid eye disease, chiasmal (pituitary, etc), optic nerve sheath meningioma, disc drusen, increased IOP (glaucoma)
- Vascular – arteritic and nonarteritic ischemic optic neuropathy, diabetes
- Inflammatory – sarcoid, systemic lupus, Behcet’s, demyelination (multiple sclerosis)
- Infectious – herpes, tuberculosis, bartonella, etc.
- Toxic & nutritional – medications (e.g., ethambutol, amiodarone, methanol), vitamin deficiency
- Metabolic – diabetes
- Neoplastic – lymphoma, leukemia, tumor, glioma
- Genetic – autosomal dominant optic atrophy (OPA1), Leber hereditary optic atrophy, Leber hereditary optic neuropathy, late complications of retinal degeneration.
- Radiation – optic neuropathy
- Traumatic – optic neuropathy
Management
The management goal is to intervene before optic atrophy is noted or to save remaining visual function. How the goal is achieved will depend on the underlying cause of the optic nerve damage.
Prognosis
Studies in patients with glaucoma (based on OCT nerve fiber layer measurements and other methods) have shown that the optic nerve has some reserve (axons) before vision loss is appreciated. After that reserve is depleted, small changes in nerve fiber loss lead to significant decrease in vision. Early detection is key, since we cannot replace dead axons.
Additional Resources
- The Neuro-Ophthalmology Virtual Education Library: library.med.utah.edu/NOVEL/
- The North American Neuro-Ophthalmolgy Society (NANOS): www.nanosweb.org
Reference
- ↑ Lee AG, Chau FY, Golnik KC, et akl. The diagnostic yield of the evaluation for isolated unexplained optic atrophy. Ophthalmology. 2005;112(5):757-759.