All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Superior orbital fissure syndrome (also known as Rochen-Duvigneaud syndrome)
Disease
Superior orbital fissure syndrome (also known as Rochen-Duvigneaud syndrome) is a collection of symptoms caused by compression of structures just anterior to the orbital apex [1]. The complex and crowded anatomy of the superior orbital fissure produces a characteristic pattern of cranial nerve, pupillary and extraocular findings. Importantly the optic nerve is spared which differentiates superior orbital fissure syndrome from orbital apex syndrome [2] . This constellation of findings was first described by Hirschfeld in 1858 and officially named by Rochen-Duvigneaud in 1896 [3].
Etiology
Superior orbital fissure syndrome is rare. When originally identified, the syndrome was thought to be secondary to a “syphilitic periostitis” [4] because it responded when treated for syphilis. The most frequent cause of superior orbital fissure syndrome is now known to be trauma, which is seen in 0.3-0.8% of patients [3][5] [6] . Common associations are motorcycle accidents, zygomatic fractures and orbital fractures [7] . Trauma-related superior orbital fissure syndrome often presents within 48 hours of a facial injury, but presentation can also be delayed by several days [8] . Other causes include neoplasms (particularly lymphoma and rhabdomyosarcoma) [9] or sometime metastasis[10], infections (such as meningitis) [11] , syphilis [12] , sinusitis [13] [14] , herpes zoster [15] [16] [17] , inflammation (such as Lupus, sarcoidosis, Tolosa-Hunt syndrome) [2] and vascular phenomena (such as carotid-cavernous fistulas, retro-orbital haematoma and carotid aneurysms), [5] [18] or idiopathic.[19] Rarely, superior orbital fissure syndrome has presented perioperatively in orbital hypertelorism surgery [20] . Presentation from trigeminocardiac reflex episodes during skull base surgery has also been reported [21].
Risk Factors
The only reported risk factor for superior orbital fissure syndrome is a pre-existing narrow superior orbital fissure [5].
Anatomy
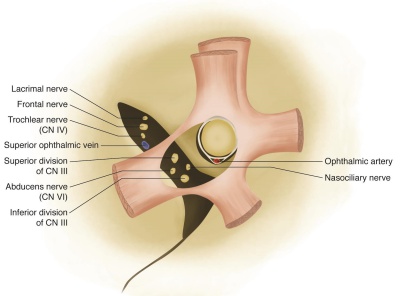
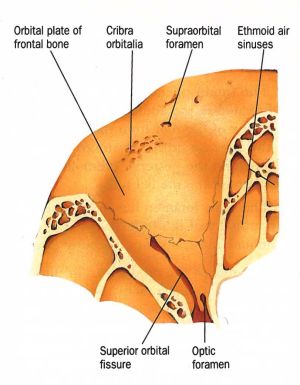
The superior orbital fissure is a bony cleft found at the orbital apex between the roof and lateral wall. It is a communication between the orbital cavity and middle cranial fossa and is bounded by the greater wing, lesser wing and body of sphenoid [22]. Its dimensions are 22mm in length and 2-8mm in width (narrowest and widest parts) and it is pear-shaped with a wide base nasally on the body of sphenoid and an apex directed superotemporally[5]. There is no significant variation of SOF size between gender or body type.[23]
Anteriorly the superior orbital fissure is related to the annulus of Zinn (common tendinous ring) on which the four rectus muscles attach. The annulus splits the superior orbital fissure into three sectors: lateral, central (oculomotor foramen) and inferior. The lateral sector contains the lacrimal, frontal and trochlear nerves and superior ophthalmic vein. The central sector, which is surrounded by the annulus of Zinn, contains the superior and inferior branches of oculomotor nerve, nasociliary and abducens nerves. The inferior sector contains the inferior ophthalmic vein[22][24] [25]. Older literature divided the superior orbital fissure into two (superior and inferior) divisions, rather than three, though the relative location of the contents inside superior orbital fissure has remained consistent[26].
Pathophysiology
Compression of structures in superior orbital fissure syndrome can be attributed to a bony fragment (such as with facial fractures) or mass effect. In all causes of superior orbital fissure syndrome, inflammation and compression of nearby nervous tissue is responsible for the majority of clinical symptoms.[19] Since the orbital muscle cone is of relatively fixed volume, being bounded by the intermuscular membrane and Tenon’s capsule, any edema or bleeding or masses in this area compromises delicate neural structure [27].
Anatomical structures within common tendinous ring (i.e. central sector) are the most vulnerable. In particular the abducens nerve is most commonly damaged [3] as it is within the central sector, lies close to the greater wing and has a relatively long intracranial course RIA. The trochlear nerve is the least commonly involved [3] as it exists above the common tendinous ring where it is well protected [5]. The anatomical proximity of the superior orbital fissure to the cavernous sinus and internal carotid artery permits vascular pathology such as carotid aneurysms and carotid-cavernous fistulas to cause compression [3].
Diagnosis
Clinical Presentation
The common clinical findings in superior orbital fissure syndrome include the following and their individual anatomical basis are explained [5][17]
- Ophthalmoplegia: due to compression or damage to oculomotor, trochlear and abducens nerves
- Ptosis: due to loss of oculomotor motor supply to the levator palpebrae superioris and loss of sympathetic input (third order postganglionic) to Muller’s muscle
- Proptosis: due to decreased tension in the extraocular muscles with loss of innervation
- Fixed dilated pupil: due to loss of parasympathetic supply to the pupil by the oculomotor nerve (please note, this is different than relative afferent pupillary defect(rAPD))
- Lacrimal hyposecretion and eyelid or forehead anaesthesia: due to damage to branches of the ophthalmic division of the trigeminal nerve
- Loss of corneal reflex: due to loss of afferent input from the ophthalmic division of the trigeminal nerve.
Other findings which may give insight into underlying etiology include:
- Chemosis and bruits in vascular causes: due to altered blood flow and vascular congestion [3]
- Facial trauma patients may concurrent subconjunctival hemorrhage, periorbital ecchymosis and soft tissue contusion [3]
- Extensive masses in the region of the superior orbital fissure may be marked by proptosis, eyelid swelling and chemosis [9]
It should also be noted a phenomena of partial superior orbital fissure syndrome has also been described where involvement is strictly confined to the central sector thus associated with isolated oculomotor, abducens and nasociliary involvement [5]
Diagnostic Procedures
The diagnosis of superior orbital fissure syndrome is suspected on clinical grounds. Imaging studies play a role in confirming the diagnosis, and more importantly, give insight into underlying etiology. Early use of plain film with superior orbital fissure views (20-25° head tilt) has largely been replaced by widespread availability of CT and MRI. The most common imaging modality is currently CT with 2mm fine cuts looking for bony fragments or compressive masses. Where a vascular cause is suspected, angiography is recommended to define any carotid-cavernous fistula or carotid aneurysm [5].
In cases managed conservatively or medically there is little consensus on need for follow-up imaging but Shama et al. (2012) suggests a repeat MRI at 6 months [1].
Differential Diagnosis
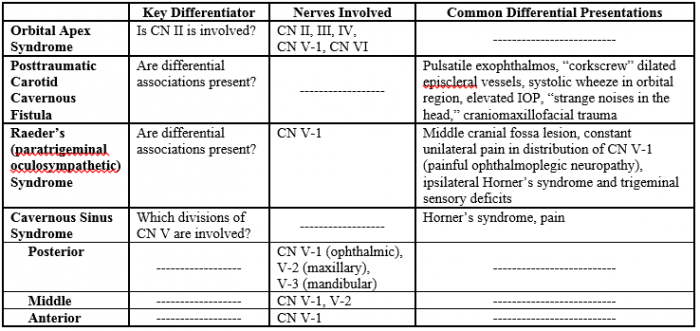
Important differentials to distinguish from superior orbital fissure syndrome are as follow (see Table 1):
- Orbital apex syndrome: where the main differentiator is optic nerve involvement [2], since both syndromes involve CN III, IV, VI, and the ophthalmic division of CN V (CNV-1) [28]. Superior orbital fissure syndrome accompanied by an ipsilateral posterior ischemic optic neuropathy from dissection and an occlusion of the internal carotid artery can be mistakenly diagnosed as orbital apex syndrome [29].
- Cavernous sinus syndrome: which can be classed into three subtypes. The posterior cavernous sinus syndrome includes whole trigeminal involvement. The middle cavernous sinus syndrome involves the first and second divisions of the trigeminal nerve (ophthalmic and maxillary). The anterior cavernous sinus syndrome only involves the ophthalmic nerve [30]. Furthermore, cavernous sinus syndrome may be associated with Horner’s syndrome and pain which is in contrast to superior orbital fissure syndrome [31].
- Posttraumatic carotid-cavernous fistula: which can be differentiated by pulsatile exophthalmos, “corkscrew dilated episcleral vessels, a clear systolic wheeze upon auscultation of the orbital region, elevated intraocular pressure, a complaint of “strange noises in the head,” and/or a history of craniomaxillofacial trauma [32].
- Raeder’s syndrome: which is characterized by constant, unilateral pain in the distribution of the CN V-1 (ophthalmic), but may also include spread to the CN V-2 (maxillary). Ipsilateral Horner’s syndrome, ipsilateral trigeminal sensory deficits, and an absence of anhidrosis secondary to the interruption of the post ganglionic pathway are usually present. Syndrome pain worsens with eye movement and may originate from periarterial inflammation [33][34]. Raeder’s syndrome, also known as paratrigeminal oculosympathetic syndrome, is a rare disorder with very few epidemiological and case series studies available. [35]
Management
The management of superior orbital fissure syndrome depends on its etiology. If orbitocranial trauma is the origin, then the sooner the treatment the best the prognosis.
Reported treatment options include medical and surgical intervention, as well as observation alone [36]. Indications for urgent intervention are vascular causes (requiring embolization) and bony fractures with displaced fragments or severe foraminal narrowing [3], particularly lateral orbital wall fractures [37]. Cases involving neoplasms, physical impingement, infections, or retrobulbar hemorrhage warrant the use of surgical exploration.[19] In certain cases, a combination of treatment options may be appropriate, such as a megadose corticosteroid treatment combined with surgical intervention [37].
Fractures
If the superior orbital fissure is compressed or narrowed by a facture, surgical decompression may be beneficial [36][37]. Surgical management in the non-urgent setting is performed at an average of 10.7 days due to concerns that early intervention is associated with increased swelling and ocular pressure [3]. Both orbital and cranial extradural approaches have been described including extranasal intraorbital to access the lateral wall, extranasal transethmoidal to access the medial wall, a modified extranasal intraorbital route, transtemporal route in causes of infectious collections and combined orbital and cranial for deep decompressions [38][39][40]. An endoscopic transmaxillary transMuller’s muscle route for decompression may also be effective, though sample size is limited [41]. A concomitant facial fracture can be surgically reduced [36]. An additional surical approach is that of simultaneous endoscopic endonasal simultaneous decompression of optic nerve canal, SOF and orbital apex[42].
Historical treatment of restricted movement due to edema has focused on conservative management and careful observation because spontaneous improvement had been documented in the past. However, if there is no evidence of bone dislocation, and thus no need for urgent surgical intervention, conservative medical treatment involving megadoses of corticosteroids has proven effective [36][37]. The most common regimen mirrors that of spinal cord/nerve injury protocols and involves intravenous IV methylprednisolone as a 30mg/kg bolus followed by a 5.4mg/kg/h on a 48 hour infusion, followed by oral prednisone taper over 2 weeks [4][43].
Vascular
In cases of retroorbital hematoma, hemorrhage generally resorbs spontaneously after 3-12 weeks. However, some authors recommend intervention if the hematoma is associated with fractures, such as aspiration, open reduction, or intravenous steroids [36]. Cases of carotid-cavernous sinus fistula can be confirmed with carotid angiography and treated via detachable balloon or coil embolization [36].
Conservative Treatment
Conservative treatment through observation alone has been suggested due to the operative difficulty and risk of further injury from surgical exploration. Spontaneous recovery, both complete and partial, of motor and sensory function has been reported in cases of traumatic etiology [44]. A recent literature review documented nineteen cases of superior orbital fissure syndrome secondary to craniofacial trauma treated with observation alone, with complete spontaneous recovery in eight of the cases [36].
Complications and Prognosis
No adverse effects from medical therapy with high dose steroids in this setting have been described [35]. Complete recovery of all nerves has been reported in 24-40% of patients receiving steroid treatment compared to 21.4% in those without. Recovery is usually extended over a period of months with progress plateauing at 6 months. The abducens nerve which is most commonly damaged shows the best recovery.
Possible sequelae from superior orbital fissure syndrome may include remaining deficit requiring further strabismus or ptosis surgery[3] [5]
References
- ↑ 1.0 1.1 Shama SA, Gheida U. Superior orbital fissure syndrome and its mimics: What the radiologist should know? The Egyptian Journal of Radiology and Nuclear Medicine. 2012;43(4):589-94.
- ↑ 2.0 2.1 2.2 Yeh S, Foroozan R. Orbital apex syndrome. Current opinion in ophthalmology. 2004;15(6):490-8.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 3.9 Chen C-T, Wang TY, Tsay P-K, Huang F, Lai J-P, Chen Y-R. Traumatic superior orbital fissure syndrome: assessment of cranial nerve recovery in 33 cases. Plastic and reconstructive surgery. 2010;126(1):205-12.
- ↑ 4.0 4.1 Walsh F, Hoyt W. Superior Orbital Fissure Syndrome. Walsh and Hoyt's clinical neuro-ophthalmology. Vol 1. 2th ed. Baltimore MD, USA.: Lippincott Williams & Wilkins;1969:398-399.
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 5.7 5.8 Chen C-T, Chen Y-R. Traumatic superior orbital fissure syndrome: current management. Craniomaxillofacial Trauma and Reconstruction. 2010;3(01):009-16.
- ↑ Skoch J, Ansay TL, Lemole GM. Injury to the Temporal Lobe via Medial Transorbital Entry of a Toothbrush. Journal of Neurological Surgery Reports. 2013;74(1):23-28.
- ↑ Kurzer A, Patel MP. Superior orbital fissure syndrome associated with fractures of the zygoma and orbit. Plastic and reconstructive surgery. 1979;64(5):715-9.
- ↑ Kim YJ, Choi, WK. Delayed Superior Orbital Fissure Syndrome After Reconstruction of Blowout Fracture. Journal of Craniofacial Surgery. 2016;27(1):e8-e10.
- ↑ 9.0 9.1 Bone I, Hadley D. Syndromes of the orbital fissure, cavernous sinus, cerebello-pontine angle, and skull base. Journal of Neurology, Neurosurgery & Psychiatry.
- ↑ https://webeye.ophth.uiowa.edu/eyeforum/cases/355-Superior-Orbital-Fissure-Syndrome.htm#gsc.tab=0
- ↑ Lakke J. Superior orbital fissure syndrome: Report of a case caused by local pachymeningitis. Archives of neurology. 1962;7(4):289-300.
- ↑ Currie JN, Coppeto JR, Lessell S. Chronic Syphilitic Meningitis Resulting in Superior Orbital Fissure Syndrome and Posterior Fossa Gumma: A Report of Two Cases followed for 20 Years. Journal of Neuro-Ophthalmology. 1988;8(3):145-56.
- ↑ Bikhazi NB, Sloan SH. Superior orbital fissure syndrome caused by indolent Aspergillus sphenoid sinusitis. Otolaryngology--Head and Neck Surgery. 1998;118(1):102-4.
- ↑ Matsuda M, Hanazono A, Kamada S, Okawa S, Kawasaki Y, Honda K, Ishikawa K, Sugawara M, Ohnishi, H. Superior orbital fissure syndrome caused by aspergillus infection from maxillary sinusitis. Neurology Clinical Neuroscience. 2015;3(4):134–136.
- ↑ Yong V, Yip C, Yong V. Herpes zoster ophthalmicus and the superior orbital fissure syndrome. Singapore medical journal. 2001;42(10):485-6.
- ↑ Jensen H, Thomsen ST, Hansen SS, Munksgaard SB, Lindelof M. Superior Orbital Fissure Syndrome and Ophthalmoplegia Caused by Varicella Zoster Virus with No Skin Eruption in a Patient Treated with Tumor Necrosis Alpha Inhibitor. Case Reports in Neurology. 2015;7:221-226.
- ↑ 17.0 17.1 Lavaju P, Badhu BP, Shah S. Herpes zoster ophthalmicus presenting as orbital abscess along with superior orbital fissure syndrome: A case report. Indian Journal of Ophthalmology. 2015;63(9):733-735.
- ↑ Postma MP, Seldomridge GW, Vines FS. Superior orbital fissure syndrome and bilateral internal carotid pseudoaneurysms. Journal of Oral and Maxillofacial Surgery. 1990;48(5):503-8.
- ↑ 19.0 19.1 19.2 Evans HH, Wurth BA, Penna KJ. Superior Orbital Fissure Syndrome: A Case Report. Craniomaxillofacial Trauma & Reconstruction. 2012;5(2):115-120. doi:10.1055/s-0032-1313363.
- ↑ Glass G, Hon K, Schweibert K, Bowman R, Jones B, Dunaway D, Britto J. Ocular Morbidity in the Correction of Orbital Hypertelorism and Dystopia: A 15-Year Experience. Plastic & Reconstructive Surgery. 2017;139(4):967-975.
- ↑ Haldar R, Gyanesh P, Srivastava A, Bettaswamy GP. Trigeminocardiac reflex preceding development of postoperative superior orbital fissure syndrome. Asian Journal of Neurosurgery. 2017;12(1):116-119.
- ↑ 22.0 22.1 Natori Y, Rhoton Jr AL. Microsurgical anatomy of the superior orbital fissure. Neurosurgery. 1995;36(4):762-75.
- ↑ Reymond J, Kwiatkowski J, Wysocki J. Clinical anatomy of the superior orbital fissure and the orbital apex. J Craniomaxillofac Surg. 2008 Sep;36(6):346-53. doi: 10.1016/j.jcms.2008.02.004. Epub 2008 May 1. PMID: 18450458.
- ↑ Shi X, Han H, Zhao J, Zhou C. Microsurgical anatomy of the superior orbital fissure. Clinical Anatomy. 2007;20(4):362-6.
- ↑ Morard M, Tcherekayev V, de Tribolet N. The superior orbital fissure: a microanatomical study. Neurosurgery. 1994;35(6):1087-93.
- ↑ Reymond J, Kwiatkowski J, Wysocki J. Clinical anatomy of the superior orbital fissure and the orbital apex. Journal of Cranio-Maxillofacial Surgery. 2008;36(6):346-353.
- ↑ Zachariades N, Vairaktaris E, Papavassiliou D, Papademetriou I, Mezitis M, Triantafyllou D. The superior orbital fissure syndrome. Journal of maxillofacial surgery. 1985;13:125-8.
- ↑ Aryasit O, Preechawai P, Aui-Aree N. Clinical presentation, aetiology and prognosis of orbital apex syndrome. Orbit. 2013;32(2):91-94.
- ↑ Anders U, Taylor E, Martel J, Martel, James. Pseudo-Orbital Apex Syndrome in the Acute Trauma Setting Due to Ipsilateral Dissection of Internal Carotid Artery. 2016;32(4):e96-e97.
- ↑ Bowerman JE. The superior orbital fissure syndrome complicating fractures of the facial skeleton. British Journal of Oral Surgery. 1969;7(1):1-6.
- ↑ Lee JH, Lee HK, Park JK, Choi CG, Suh DC. Cavernous sinus syndrome: clinical features and differential diagnosis with MR imaging. American Journal of Roentgenology. 2003;181(2):583-90.
- ↑ Lazaridou M, Bourlidou E, Kontos K, Mangoudi D. Carotid-Cavernous Fistula as a Complication of Facial Trauma: A Case Report. Craniomaxillofacial Trauma & Reconstruction. 2015;8(3):239-245.
- ↑ Nagel MA, Bert RJ, Gilden D. Raeder syndrome produced by extension of chronic inflammation to the internal carotid artery. Neurology. 2012;79(12):1296-1297.
- ↑ Solomon S. Raeder Syndrome. Arch Neurol. 2001;58(4):661–662.
- ↑ 35.0 35.1 Evers S. Facial pain: overlapping syndromes. Cephalagia. 2017;37(7):705-713.
- ↑ 36.0 36.1 36.2 36.3 36.4 36.5 36.6 Chen CT, Chen YR. Traumatic superior orbital fissure syndrome: current management. Craniomaxillofacial Trauma & Reconstruction. 2010;3(1):9-16.
- ↑ 37.0 37.1 37.2 37.3 Caldarelli C, Benech R, Iaquinta C. Superior Orbital Fissure Syndrome in Lateral Orbital Wall Fracture: Management and Classification Update. Craniomaxillofacial Trauma & Reconstruction. 2016;9(4):277-283.
- ↑ Murakami I. Decompression of the superior orbital fissure. American journal of ophthalmology. 1965;59(5):803-8.
- ↑ Antonyshyn O, Gruss JS, Kassel E. Blow-in fractures of the orbit. Plastic and reconstructive surgery. 1989;84(1):10.
- ↑ Al-Hasmi A, Al-Saadi N. Surgical versus medical management of traumatic superior orbital fissure syndrome: a report of two cases. International Journal of Oral and Maxillofacial Surgery. 2015;44(s1):e24.
- ↑ Wang X, Li Y, Huang C, Liu H, Li Q, Yu M, Hou L. Endoscopic transmaxillary transMüller's muscle approach for decompression of superior orbital fissure: A cadaveric study with illustrative case. Journal of cranial-maxillofacial surgery. 2014;42(2):132-140.
- ↑ Liu J, Zhao J, Wang Y, et al. Simultaneous Endoscopic Endonasal Decompression of the Optic Canal, Superior Orbital Fissure, and Proper Orbital Apex for Traumatic Orbital Apex Syndrome: Surgical Anatomy and Technical Note. Front Surg. 2022;8:811706. Published 2022 Jan 21. doi:10.3389/fsurg.2021.811706
- ↑ Acartürk S, Seküçoglu T, Kesiktäs E. Mega dose corticosteroid treatment for traumatic superior orbital fissure and orbital apex syndromes. Annals of plastic surgery. 2004;53(1):60-4.
- ↑ Shanker V, Nigam V, Misra N. Successful Conservative Management of a Case of Traumatic Superior Orbital Fissure Syndrome. Indian Journal of Neurotrauma. 2016;13(1):31-34.