Thiel-Behnke Corneal Dystrophy
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Disease
Thiel-Behnke corneal dystrophy (TBCD), also nicknamed “honeycomb” corneal dystrophy, is a type of epithelial-stromal transforming growth factor beta-induced (TGFβI) gene-linked dystrophy.[1] This is a progressive dystrophy that is bilateral and inherited. It can present with corneal opacities and erosions (often recurrent). Over time, the frequency of erosions decreases and the disease progresses to a gradual impairment of vision.[2][3]
The disease initially affects the central cornea in Bowman’s layer and progresses to involve the peripheral cornea and deep stroma later in life.[3][4]
Etiology
TBCD has been associated with chromosome 5q31 of the human TGFβI gene and is most commonly linked to a Arg555Gln mutation but also associated with other mutations such as Met502Val/Arg555Gln Gly623_His626del.[1][5][6][7]
A mutation on chromosome 10q24 was once thought to be associated with TBCD, though this was identified as a possible novel separate entity by the updated International Classification of Corneal Dystrophies.[3] TBCD is distinct from Reis-Bücklers Corneal Dystrophy, which, although localized to 5q31, contains a different mutation on the TGFβI gene (Arg124Leu).
Genetics
Autosomal dominant inheritance.[3]
Epidemiology
TBCD is an extremely rare condition with an unknown prevalence. The current literature is limited to case series and reports.
Pathophysiology
TGFβI is responsible for a variety of cell processes, namely cell migration, adhesion, differentiation, and growth. Although the exact mechanism is unclear, mutations in the TGFβI gene have been linked to dystrophic protein deposits in the cornea, specifically the superficial epithelium, Bowman membrane-anterior basement membrane complex, and anterior stroma, but have also been reported in the endothelium.[1] It has been hypothesized impaired autophagy leads to an accumulation of mutated TGFβI in corneal fibroblasts. Overall, this leads to a progressive and bilateral loss of corneal transparency and impaired visual acuity.[1]
Diagnosis
History
Patients typically present with bilateral recurrent painful corneal erosions in the first or second decade of life.[3][8] The disease course is slow and progressive, accompanied by gradual vision impairment as a result of corneal scarring with involvement of the visual axis.[3]
Signs & Symptoms
Slit lamp examination initially reveals diffuse irregularly shaped opacities appearing as solitary flecks which are followed by symmetrical subepithelial honeycomb-like opacities.[3][8][9] There is also irregular raising of the corneal surface.[8] As the disease progresses, opacities can affect the corneal periphery and deeper stroma.[3]
Differential Diagnosis
The challenge in identifying TBCD is in its similarity to the other epithelial-stromal TGFβI-associated corneal dystrophies, though genetic testing can aid in distinguishing them.
- Reis-Bücklers Corneal Dystrophy (RBCD): Presents similarly to TBCD and also affects Bowman’s layer, although RBCD is typically associated with a more aggressive disease course as well as irregular and well-demarcated opacities on slit lamp examination.[3] RBCD is associated with chromosone 5q31 mutation.
- Lattice Corneal Dystrophy, Type 1: Also presents with recurrent erosions and diffuse opacities. Involves a mutation on chromosome 5q21 and results in thick lattice patterned amyloid opacities along the central corneal stroma.[9]
- Granular Corneal Dystrophy: Associated with chromosome 5q21 mutation.
- Type 1: Recurrent erosions that progress to visual loss later in life, typically after 40 years. Hyaline deposits appear as distinct granules in the central cornea. In children, this appears as punctiform opacities in a vortex pattern and in adults, opacities have a snow-like appearance.
- Type 2: Presents similarly to Type 1 with less frequent erosions and slowly progresses after initial presentation in the second decade of life. Opacities result from amyloid deposits and appear as rings, granules, and lines in the superficial cornea.
Diagnostic Procedures
Light Microscopy
Thickening and thinning of the epithelium due to deformation of the stroma and epithelial basement membrane.[3] Bowman’s layer is replaced by the formation of a fibrocellular pannus.
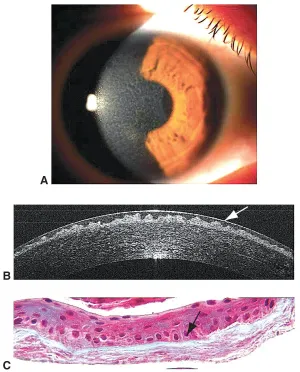
Optical Coherence Tomography
Demonstrates a moderate to hyperreflective material in a sawtooth pattern, corresponding in deposits in Bowman’s layer and epithelium.[3][10]
Electron Microscopy
Curly collagen fibers are pathognomonic for TBCD and can be seen on electron microscopy.[1][3][11]
Immunochemistry
Confirms the presence of TGFBI in curly fibers.[3][12]
Confocal Microscopy
Reveals irregular reflective deposits within Bowman’s layer and epithelium.[3] The reflective material in Bowman’s layer is irregular, while deposits in the basal epithelium are more homogeneously reflective with round edges and dark shadowing.[3]
Histology
An irregularly thick band containing hyaline, avascular and paucicellular connective tissue can be seen replacing Bowman’s layer.[10][11] This commonly presents in a wavy “sawtooth” pattern.[3][9]
Management
General Treatment
There is no consensus for the optimal treatment of TBCD. Treatment for recurrent epithelial erosions can include frequent lubrication, hypertonic saline drops, and bandage contact lens. Phototherapeutic keratectomy or superficial keratectomy (with or without femotosecond laser assistance) can treat corneal opacities and delay the need for keratoplasty.[4] Otherwise, patients have been reported to be treated with deep lamellar keratoplasty and penetrating keratoplasty.[4]
In recent studies, Bowman layer onlay transplantation has emerged as a promising surgical intervention for managing epithelial-stromal dystrophies such as Thiel-Behnke corneal dystrophy (TBCD)[13]. Oganesyan et al. demonstrated that the transplantation of a donor Bowman’s layer can restore the anatomical structure of the anterior cornea, providing better biomechanical stability and visual outcomes[13]. Unlike traditional lamellar keratoplasty, Bowman layer transplantation offers a less invasive approach, preserving more of the host corneal tissue while reducing the risk of recurrence and graft complications[13]. This technique represents an important advancement in the surgical management of TBCD, particularly for patients with significant visual impairment and anterior corneal opacification[13].
Prognosis
TBCD is a slowly progressive disease. Though recurrence of erosions subsides overtime, the resultant scarring affecting the central cornea impairs visual acuity which may necessitate corneal transplant.[2] Post-phototherapeutic keratectomy or penetrating keratoplasty, there is a high incidence of recurrence. Patients require continued monitoring for recurring symptoms and may need to have repeat procedures performed.
Additional Resources
- https://www.cornealdystrophyfoundation.org/
- https://rarediseases.info.nih.gov/diseases/9275/corneal-dystrophy-thiel-behnke-type
References
- ↑ 1.0 1.1 1.2 1.3 1.4 Wang L, Zhao C, Zheng T, et al. Torin 1 alleviates impairment of TFEB-mediated lysosomal biogenesis and autophagy in TGFBI (p.G623_H626del)-linked Thiel-Behnke corneal dystrophy. Autophagy. 2022;18(4):765-782. doi:10.1080/15548627.2021.1955469
- ↑ 2.0 2.1 Hieda O, Kawasaki S, Wakimasu K, Yamasaki K, Inatomi T, Kinoshita S. Clinical outcomes of phototherapeutic keratectomy in eyes with Thiel-Behnke corneal dystrophy. Am J Ophthalmol. 2013;155(1):66-72 e1. doi:10.1016/j.ajo.2012.06.022
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 Weiss JS, Moller HU, Aldave AJ, et al. IC3D classification of corneal dystrophies--edition 2. Cornea. 2015;34(2):117-59. doi:10.1097/ICO.0000000000000307
- ↑ 4.0 4.1 4.2 Abusayf MM, Tobaigy MF, Alfawaz A, Alkatan HM. Femtosecond laser assisted superficial lamellar keratectomy as a successful treatment of corneal opacity in a patient with Thiel Behnke corneal dystrophy. Int J Surg Case Rep. 2022;95:107257. doi:10.1016/j.ijscr.2022.107257
- ↑ Munier FL, Korvatska E, Djemai A, et al. Kerato-epithelin mutations in four 5q31-linked corneal dystrophies. Nat Genet. 1997;15(3):247-51. doi:10.1038/ng0397-247
- ↑ Okada M, Yamamoto S, Tsujikawa M, et al. Two distinct kerato-epithelin mutations in Reis-Bucklers corneal dystrophy. Am J Ophthalmol. 1998;126(4):535-42. doi:10.1016/s0002-9394(98)00135-4
- ↑ Niel-Butschi F, Kantelip B, Iwaszkiewicz J, et al. Genotype-phenotype correlations of TGFBI p.Leu509Pro, p.Leu509Arg, p.Val613Gly, and the allelic association of p.Met502Val-p.Arg555Gln mutations. Mol Vis. 2011;17:1192-202.
- ↑ 8.0 8.1 8.2 Moshirfar M, Bennett P, Ronquillo Y. Corneal Dystrophy. StatPearls. 2023. https://www.ncbi.nlm.nih.gov/books/NBK557865/
- ↑ 9.0 9.1 9.2 Lisch W, Weiss JS. Clinical and genetic update of corneal dystrophies. Exp Eye Res. 2019;186:107715. doi:10.1016/j.exer.2019.107715
- ↑ 10.0 10.1 Nishino T, Kobayashi A, Mori N, Yokogawa H, Sugiyama K. In vivo Imaging of Reis-Bucklers and Thiel-Behnke corneal dystrophies using anterior segment optical coherence tomography. Clin Ophthalmol. 2020;14:2601-2607. doi:10.2147/opth.S265136
- ↑ 11.0 11.1 Ridgway AE, Akhtar S, Munier FL, et al. Ultrastructural and molecular analysis of Bowman's layer corneal dystrophies: an epithelial origin? Invest Ophthalmol Vis Sci. 2000;41(11):3286-92.
- ↑ Streeten BW, Qi Y, Klintworth GK, Eagle RC, Jr., Strauss JA, Bennett K. Immunolocalization of beta ig-h3 protein in 5q31-linked corneal dystrophies and normal corneas. Arch Ophthalmol. 1999;117(1):67-75. doi:10.1001/archopht.117.1.67
- ↑ 13.0 13.1 13.2 13.3 Oganesyan OG, Ashikova PM, Ivanova AV, et al. Bowman layer onlay transplantation in the management of epithelial-stromal dystrophies of Reis–Bücklers and Thiel–Behnke. Russian Ophthalmology Journal. 2025; Available at: https://roj.igb.ru/jour/issue/download/45/31#page=57.

