Anterior Segment Optical Coherence Tomography for Various Ocular Surface Lesions and Corneal Pathologies
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Billing:
Anterior segment optical coherence tomography: 2018-2019 Current Procedural Terminology (CPT): 92132 (scanning computerized ophthalmic diagnostic imaging, anterior segment with interpretation and report, unilateral or bilateral).
Payment is bilateral, meaning payment is made once, whether one or both eyes are tested. A laterality modifier is not needed for billing.
Introduction
Imaging technologies are instrumental adjuncts to the clinical examination for the diagnosis and treatment of various ocular pathologies. Imaging modalities for the ocular surface and anterior segment include corneal topography, Scheimpflug tomography, in vivo confocal microscopy, high-resolution ultrasound biomicroscopy and optical coherence tomography (OCT).[1][2]
While initially developed for imaging of the posterior segment, OCT has been gaining rising momentum for ocular surface and anterior segment imaging. This technology affords clinicians the ability to obtain dynamic, high and ultra-high resolution, cross-sectional images in a noninvasive and rapid manner, facilitating ease of use. Because they don't require touch, are user friendly, and can take images quickly, OCT devices are well-tolerated by patients and can be used by operators with varying levels of experience. Acquired images can range from 3 to 5 microns in resolution, allow for assessment of subtle nuances in tissue morphology and cellular architecture, and can be easily interpreted by novice as well as experienced clinicians.[1][3]
The gold standard for the diagnosis of several ocular surface conditions is the histopathological and cytologic examination of incisional or excisional conjunctival biopsy specimens. Biopsies are invasive diagnostic techniques that can sometimes provide a false negative if inadequate tissue is obtained. Anterior segment OCT (AS-OCT) facilitates obtaining an “optical biopsy” or in vivo imaging of various dystrophic, degenerative, and neoplastic ocular surface and corneal pathologies, allowing clinicians to diagnose diseases otherwise not visualized by traditional methods. [2]
How does AS-OCT work and what machine options are available?
The eventual transition from time-domain to spectral-domain OCT devices (also known as Fourier-domain OCT) has facilitated faster scanning speeds, greater tissue penetrance, and higher axial resolution images due to use of shorter wavelengths of light. Images with axial resolutions ranging from less than 5 microns (considered ultra-high resolution) to greater than 5 microns (considered high resolution) can now be obtained. Spectral domain OCT devices however have the disadvantage of having reduced scan depth compared to time domain OCT machines, due to shorter horizontal scan width. [4][5][6][7]
Swept-source OCT has also been developed, enabling simultaneous acquisition of numerous longitudinal and transverse scans to create 3-dimensional corneal, anterior segment, and gonioscopy images. [8] There are several high-quality commercially available AS-OCT machines. Some of the common spectral domain machines used include the Heidelberg Spectralis OCT, the Carl Zeiss Meditec Cirrus OCT, the Optovue RTVue and the Optovue Avanti. Common time domain machines include the Carl Zeiss Meditec Stratus OCT and Visante OCT. [1]
How does one read an AS-OCT of the ocular surface?
AS-OCT can systematically image the ocular surface from front to back (the tear film, conjunctiva, individual corneal layers, sclera, angle and lenticular structures).
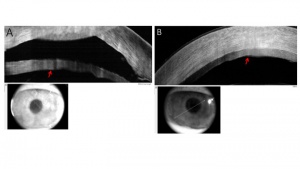
When assessing an AS-OCT through the normal corneal tissue, the tear film is the first, thin, hyperreflective layer. It overlies a thin band of hyporeflective tissue which is the normal corneal epithelium (normally ~50-70 microns in thickness). The underlying corneal stroma is thickened tissue with variable hyperreflectivity and the corneal endothelium is a thin band of hyperreflective tissue (Figure 1).
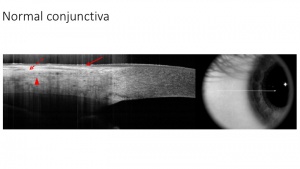
Similarly, an AS-OCT image through the normal conjunctiva and sclera typically shows a thin band of hyporeflective tissue which is the normal conjunctival epithelium that overlies a band of hyperreflectivity representing the subepithelial region. The sclera is then a thickened, well circumscribed band of hyperreflective tissue (Figure 2).
When obtaining an AS-OCT image through an ocular surface lesion, the clinician must first identify the band of normal epithelial tissue. Then, one must determine if the epithelium in the region of the lesion is normal, thin, or thickened and if the epithelium is hyperreflective or hyporeflective. Then, the location of the lesion in question needs to be identified. If the lesion is contained within the epithelium or the epithelium is abnormal in appearance, the lesion is epithelial in nature. If the lesion is below the band of epithelium, it is subepithelial. The differential of the imaged lesion can then be broadened based upon its epithelial or subepithelial location and reflectivity characteristics.
AS-OCT image characteristics for ocular surface lesions
Ocular surface squamous neoplasia (OSSN) - epithelial
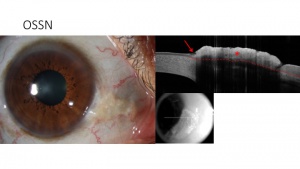
Ocular surface squamous neoplasia (OSSN) has proven to be distinctive on AS-OCT. Notably, OSSN is an epithelial lesion, both on the cornea and on the conjunctiva. Distinctive AS-OCT features including 1. thickened epithelium; 2. hyperreflective epithelial layer; and 3. an abrupt transition from normal to abnormal epithelium. AS-OCT features resolve completely with normalization of the epithelium after successful medical therapy or surgical intervention. Using a cut off of approximately 140 um, AS-OCT findings have a 94-100% sensitivity and 100% specificity for the diagnosis of OSSN. AS-OCT can also be used to detect subclinical disease that often is not appreciated on slit-lamp examination. As such, AS-OCT serves as a powerful tool for the non-invasive diagnosis of OSSN and can be used to determine need for treatment initiation as well as monitoring of the disease course. [1][5][9](Figure 3)
Conjunctival melanoma – subepithelial
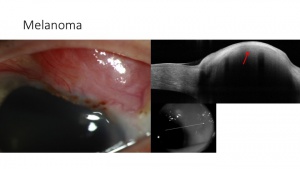
AS-OCT images of melanomas typically show a hyperreflective, subepithelial lesion. The epithelium is normal or slightly thickened with variable hyperreflectivity of the basal epithelium, which suggests involvement of the epithelium with atypical melanocytes. This imaging can help to rule in or rule out a pigmented OSSN versus melanoma.[9][10] (Figure 4).
Conjunctival lymphoma – subepithelial
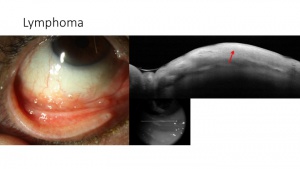
On AS-OCT, conjunctival lymphoma is characterized by a normal layer of epithelium overlying homogenous, dark, hyporeflective subepithelial lesions with smooth, discrete borders. The lesions can often contain monomorphic, stippled, dot like infiltrates that correspond to the infiltration of monoclonal lymphocytes.[9] (Figure 5).
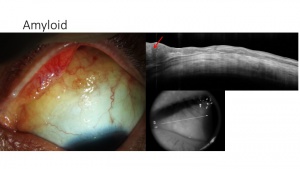
Conjunctival amyloidosis – subepithelial
Conjunctival amyloidosis can clinically appear similar to lymphoma. However, on AS-OCT, images show normal epithelium overlying heterogenous, dark lesions with irregular, diffuse borders, as compared with the homogenous and regular appearance of lymphomas. These subepithelial lesions often contain hyper-reflective linear infiltrates corresponding with the deposition of amyloid material.[9] (Figure 6).
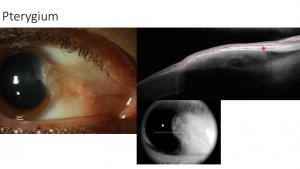
Pterygium – subepithelial
AS-OCT images of pterygia demonstrate a thin or normal layer of epithelium with varying levels of hyperreflectivity overlying a dense, hyperreflective, fibrillary subepithelial lesion that is between the corneal epithelium and Bowman’s layer. Studies have shown that high-resolution AS-OCT can reproducibly differentiate between pterygia and OSSN by identifying statistically significant differences in epithelial thickness and location of the primary lesion (epithelial location for OSSN and subepithelial location for pterygia).[10] [11](Figure 7).
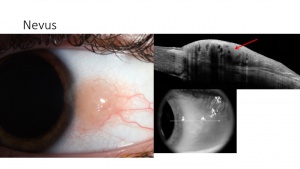
Conjunctival nevus – subepithelial
Conjunctival nevi often have normal thickness or slightly thickened epithelium overlying a well-circumscribed subepithelial lesion on AS-OCT. Unlike melanomas, nevi classically contain cystic spaces both clinically as well as on AS-OCT which is suggestive of chronicity. Imaging is particularly beneficial in amelanotic nevi seen in children where the cysts may not be clinically apparent, but the AS-OCT can easily allow them to be visualized to assist in the diagnosis. Compound nevi can, however, contain a portion of the lesion in the epithelium and substania propria in addition to the subepithelial space.[9] (Figure 8).
Difficulties with AS-OCT images of subepithelial lesions
One drawback of spectral domain AS-OCT images is that thicker subepithelial lesions can exhibit shadowing which can obscure the internal details of subepithelial lesions. This is due to reduced scan depth of the spectral domain machines. While ultrasound biomicroscopy or time-domain OCT can be employed for improved depth visualization, the details of the lesions can concomitantly be lost. For conjunctival melanomas, lymphomas and amyloidosis, AS-OCT images do not always help arrive at a definitive diagnosis as they do for OSSN, but images can help rule out an epithelial malignancy and help guide the differential. Histopathologic analysis of biopsy tissue as well as flow cytometry and gene rearrangement is needed for final differentiation of these lesions.
| Pathology | Findings on AS-OCT |
|---|---|
| OSSN | Epithelial lesion with thickened, hyperreflective epithelium with abrupt transition from normal to abnormal epithelium |
| Conjunctival melanoma | Normal thickness epithelium with variable hyperreflectivity of the basal layer with subepithelial, hyperreflective lesion |
| Conjunctival lymphoma | Normal epithelium with homogenous, hyporeflective, subepithelial lesion with discrete and smooth borders and stippled monomorphic infiltrates |
| Conjunctival amyloidosis | Normal epithelium with homogenous, hyporeflective, subepithelial lesion with irregular diffuse borders and hyperreflective linear material |
| Pterygium | Normal epithelium with varying levels of hyperreflectivity with a dense, hyper-reflective, fibrillary subepithelial lesion |
| Conjunctival nevus | Normal epithelium with well-circumscribed subepithelial lesion, often containing cysts |
AS-OCT image characteristics for corneal pathologies
Keratoconus
AS-OCT images can be used to characterize corneal microarchitecture and regional epithelial thickness in patients with early keratoconus and post-operative corneal ectasia. AS-OCT can also be useful in evaluating the effects cross-linking by identifying corneal demarcation lines, which are identified as areas with changes in stromal reflectivity, to estimate the depth of penetration of different protocols of collagen crosslinking treatment. Finally, it can also be implemented to evaluate corneal changes and he position and depth of intracorneal ring segments.[12][13]
Corneal dystrophies
AS-OCT can also be used to image several dystrophic and degenerative conditions of the cornea. Salzmann’s nodular degeneration is characterized by localized areas of hyperreflective material deposited between Bowman’s layer and the anterior stroma. Band keratopathy can be visualized as hyperreflective material at the level of Bowman’s layer with some posterior shadowing. AS-OCT of anterior basement membrane dystrophy can show increased reflectivity of the epithelial basement membranes with areas of basement membrane duplication and intraepithelial hyporeflective cysts. Thiel Behnke dystrophy is characterized by hyperreflective material in a saw tooth configuration deposited on the surface of Bowman’s layer often extending into the epithelium on AS-OCT. Granular dystrophy is often found with hyperreflective material deposited in the anterior stroma with clear intervening spaces.[14]
Infectious keratitis
AS-OCT can also be used to image cases of microbial keratitis. Hyperreflectivity in the corneal stroma can signify corneal infiltration. In cases of Acanthamoeba keratitis, keratoneuritis can be seen as highly reflective lines in the anterior to mid stroma. Serial AS-OCT images can be used throughout the disease course to monitor corneal thickness, particularly areas of corneal thinning, and corneal scarring, which will appear as areas of subepithelial or stromal hyperreflectivity. AS-OCT can also be used to elucidate the depth of corneal opacities to assist surgeons in determining the optimal surgical procedure for visual rehabilitation, which can range from a phototherapeutic keratectomy (PTK), deep anterior lamellar keratoplasty (DALK) or penetrating keratoplasty (PK).
AS-OCT for anterior segment surgery planning
AS-OCT has proven to be a powerful tool to monitor the success and complications of several anterior segment surgical procedures, such as Descemet stripping automated endothelial keratoplasty (DSAEK), Descemet membrane endothelial keratoplasty (DMEK), laser-assisted in situ keratomileusis (LASIK) and even Boston Keratoprosthesis (KPro) implantation. Post-cataract surgery, it can be used to detect pathology, such as Descemet’s detachments in patients with persistent post-operative corneal edema. It can be using intraoperatively during lamellar keratoplasty to assess the effectiveness of Descemet’s membrane stripping, determine the presence of subclinical interface fluid between the host cornea, and even confirm graft adherence and centration.[15][16] Post-operatively, images can be used to assess long-term graft adherence, overall graft thickness and even epithelial remodeling (Figure 9). AS-OCT can also capture images of the KPro-cornea interface which can be difficult to assess by clinical examination alone.[17] AS-OCT can also be used to identify flap dislocations after laser-assisted in situ keratomileusis (LASIK) and also identify macrostriae, flap edema, epithelial hyperplasia and epithelial ingrowth in these cases.[18]
Conclusion
In conclusion, AS-OCT is a powerful addition to the clinical examination for the diagnosis of various ocular surface lesions and corneal pathologies. Its applications are numerous, and future studies can help elucidate its role for more challenging lesions.
References
- ↑ 1.0 1.1 1.2 1.3 Thomas BJ, Galor A, Nanji AA, et al. Ultra high-resolution anterior segment optical coherence tomography in the diagnosis and management of ocular surface squamous neoplasia. The ocular surface. 2014;12(1):46-58.
- ↑ 2.0 2.1 Karp CL. Evolving Technologies for Lid and Ocular Surface Neoplasias: Is Optical Biopsy a Reality? JAMA ophthalmology. 2017;135(8):852-853.
- ↑ Yim M, Galor A, Nanji A, et al. Ability of novice clinicians to interpret high-resolution optical coherence tomography for ocular surface lesions. Canadian Journal of Ophthalmology. 2017.
- ↑ Han SB, Liu YC, Noriega KM, Mehta JS. Applications of Anterior Segment Optical Coherence Tomography in Cornea and Ocular Surface Diseases. Journal of ophthalmology. 2016;2016:4971572.
- ↑ 5.0 5.1 Wang J, Abou Shousha M, Perez VL, et al. Ultra-high resolution optical coherence tomography for imaging the anterior segment of the eye. Ophthalmic surgery, lasers & imaging : the official journal of the International Society for Imaging in the Eye. 2011;42 Suppl:S15-27.
- ↑ Kanellopoulos AJ, Asimellis G.Anterior-segment optical coherence tomography investigation of corneal deturgescence and epithelial remodeling after DSAEK. Cornea. 2014;33(4):340-348.
- ↑ Ramos JL, Li Y, Huang D. Clinical and research applications of anterior segment optical coherence tomography - a review. Clin Exp Ophthalmol. 2009;37(1):81-89.
- ↑ Nowinska AK, Teper SJ, Janiszewska DA, et al. Comparative Study of Anterior Eye Segment Measurements with Spectral Swept-Source and Time-Domain Optical Coherence Tomography in Eyes with Corneal Dystrophies. BioMed research international. 2015;2015:805367.
- ↑ 9.0 9.1 9.2 9.3 9.4 Shousha MA, Karp CL, Canto AP, et al. Diagnosis of ocular surface lesions using ultra-high-resolution optical coherence tomography. Ophthalmology. 2013;120(5):883-891.
- ↑ 10.0 10.1 Wong JR, Nanji AA, Galor A, Karp CL. Management of conjunctival malignant melanoma: a review and update. Expert review of ophthalmology. 2014;9(3):185-204.
- ↑ Nanji AA, Sayyad FE, Galor A, Dubovy S, Karp CL. High-Resolution Optical Coherence Tomography as an Adjunctive Tool in the Diagnosis of Corneal and Conjunctival Pathology. The ocular surface. 2015;13(3):226-235.
- ↑ Temstet C, Sandali O, Bouheraoua N, et al. Corneal epithelial thickness mapping using Fourier-domain optical coherence tomography for detection of form fruste keratoconus. Journal of cataract and refractive surgery. 2015;41(4):812-820.
- ↑ Kymionis GD, Tsoulnaras KI, Grentzelos MA, et al. Corneal stroma demarcation line after standard and high-intensity collagen crosslinking determined with anterior segment optical coherence tomography. Journal of cataract and refractive surgery. 2014;40(5):736-740.
- ↑ Vajzovic LM, Karp CL, Haft P, et al. Ultra high-resolution anterior segment optical coherence tomography in the evaluation of anterior corneal dystrophies and degenerations. Ophthalmology. 2011;118(7):1291-1296.
- ↑ Knecht PB, Kaufmann C, Menke MN, Watson SL, Bosch MM. Use of intraoperative fourier-domain anterior segment optical coherence tomography during descemet stripping endothelial keratoplasty. American journal of ophthalmology. 2010;150(3):360-365.e362.
- ↑ Mencucci R, Favuzza E, Tartaro R, Busin M, Virgili G. Descemet stripping automated endothelial keratoplasty in Fuchs' corneal endothelial dystrophy: anterior segment optical coherence tomography and in vivo confocal microscopy analysis. BMC ophthalmology. 2015;15:99.
- ↑ Zarei-Ghanavati S, Betancurt C, Mas AM, Wang J, Perez VL. Ultra high resolution optical coherence tomography in Boston type I keratoprosthesis. Journal of ophthalmic & vision research. 2015;10(1):26-32.
- ↑ Iovieno A, Sharma DP, Wilkins MR. OCT visualization of corneal structural changes in traumatic dislocation of LASIK flap. International ophthalmology. 2012;32(5):459-460.