Iris Melanoma
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Iris melanoma is the most common primary neoplasm of the iris and account for approximately 5% of all uveal melanomas. The average age of involvement lies within the 5th decade. Although there is no gender predilection, iris melanomas occur more frequently in Caucasians with a light colored iris versus non-Caucasians. There is no consistent evidence linking iris melanoma with UV light or environmental exposure
Diagnosis
Most iris melanomas are asymptomatic and thus diagnosed at an ophthalmic routine examination. In addition to a circumscribed or diffuse iris mass, heterochromia, chronic uveitis, glaucoma, or spontaneous hyphema may occur.
Clinical Diagnosis
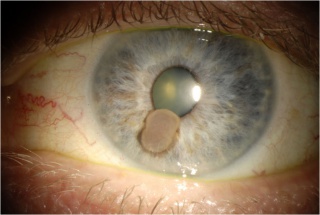
Clinically, circumscribed (Fig. 1A) and diffuse (Fig. 2A) iris melanomas can be distinguished.
There are several clinical variations including circumscribed, diffuse, and tapioca iris melanoma. Circumscribed iris melanomas appear as a variably pigmented well-defined mass in the iris stroma and is more commonly found in the inferior half of the iris. The degree of pigmentation can vary as well as the shape. Some are small and flat and others are elevated and dome shaped. Diffuse iris melanomas present with progressive iris discoloration with disappearance of iris crypts and accumulation of pigment in the anterior chamber. These can be misdiagnosed as pigmentary glaucoma or pigment dispersion syndrome. Tapioca melanoma is a name introduced by Reese and associates to describe a variant of iris melanoma with multiple amelanotic nodules on the surface giving the appearance of tapioca pudding.
In an analysis of 317 eyes with iris melanoma by Shields et al, clinical features included corectopia (45%), ectropion uveae (24%), glaucoma (35%), angle seeding (28%), hyphema (3%), and extraocular extension (3%). It is important to remember iris nevi can also be associated with ectropion uveae so the presence of this does not imply malignancy. Iris melanomas can cause secondary glaucoma from direct extension into the trabecular meshwork, neovascular glaucoma, or inflammatory posterior synechia. Obviously, a tube or filter would be contraindicated in these situations and therefore the correct diagnosis of iris melanoma is imperative.
Photo-documentation and ultrasound examination (Fig. 3A, B) should be obtained as a baseline value and on follow-up visits to document changes in size and shape.
Histologic diagnosis
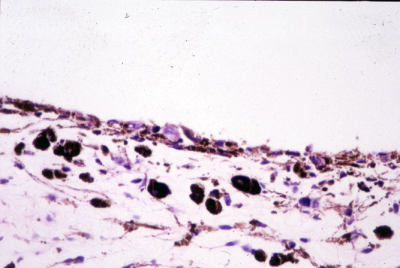
Most iris melanomas are composed of spindle cells (Fig. 1B, C) and are, therefore, regarded as low-grade tumors.
More aggressive iris melanomas (Fig. 2B) exhibit a varying amount of epithelioid cells. The amount of pigment also varies among lesions.
Differential diagnosis
- Iris freckle
- Iris nevus
- Iris melanocytoma
- Iris nevus syndrome
- Iris pigment epithelial cyst
- Peripheral anterior synechiae
- Iris foreign body
- Lisch nodules
- Fuch’s heterochromic iridocyclitis
- Latanoprost therapy
- Juvenile xanthogranuloma
- Iris metastases
- Leukemia
- Ciliary body melanoma with anterior extension
- Other iris tumors (e.g. leiomyoma, rhabdomyosarcoma)
Management
Treatment
The treatment strategy of iris melanomas depends on the clinical impression and behavior. Small non-growing lesions may be just periodically observed (“watchful waiting”) and thoroughly documented. If the clinical behavior is more worrisome with a lesion exhibiting documented growth, surface nodularity, height greater than 2mm, prominent vascularity, and associated hyphema. Different treatment options are available such as brachytherapy, excisional biopsy (iridectomy with anterior cyclectomy if needed), and enucleation. The latter procedure is reserved for a diffuse involvement of the iris and angle structures or the development of complications like glaucoma that are not amenable to non-invasive methods. A biopsy for gene expression profiling can be performed similarly for choroidal melanoma (see choroidal melanoma).
Follow up
Periodical follow-up is required in order to document the growth pattern of the mass or to evaluate the outcome after treatment. Many patients will develop glaucoma after brachytherapy.
Prognosis
Iris melanomas are more indolent and exhibit a lower metastasis rate then their ciliary body or choroidal counterparts. The rate of metastasis is approximately 3% at 5 years, 5% at 10 years, and 10% at 20 years. Many patients will develop glaucoma after treatment with brachytherapy and may need to be comanaged with a glaucoma specialist. Pressure lowering drops and CPC are recommended prior to any intraocular surgery (trabeculectomy and shunt tubes) after treatment of iris melanomas.
Additional Resources
- www.atlasophthalmology.com
- www.eyecancer.com
References
- Shields CL, Shields JA, Materin M, Gershenbaum E, Singh AD, Smith A. Iris melanoma: risk factors for metastasis in 169 consecutive patients. Ophthalmology. 2001;108(1):172-8.
- Spencer WH. Ophthalmic Pathology. An Atlas and Textbook (4th edition). Philadelphia: WB Saunders; 1996.
- Yanoff M, Sassani JW. Ocular Pathology (6th edition). Elsevier; 2009.
- Shields CL and Shields JA. Intraocular Tumors: an Atlas and Textbook. 2016
- Shields CL, Kaliki S et al. Iris melanomas: features and prognosis in 317 children and adults. J AAPOS 2012;16(1):10-16
- Reese AB, Mund ML et al. Tapioca melanoma of the iris mimicking a vascular tumor: a clinicopathological correlation. Clin Experiment Ophthalmol 2004;32;327-330.