Birdshot Retinochoroidopathy
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Disease
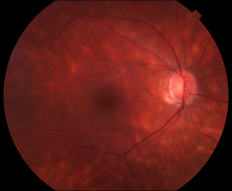
Birdshot Chorioretinopathy (BSCR)—also referred to as birdshot retinochoroidopathy, vitiliginous chorioretinitis, or birdshot uveitis—is a chronic, bilateral posterior uveitis characterized by multifocal, cream-colored choroidal lesions involving the posterior pole and mid-periphery.[1] These lesions are typically clustered around the optic nerve and posterior pole and radiate toward the periphery in a pattern resembling birdshot pellets from a shotgun. Individual lesions measure approximately one-quarter to one-half of an optic disc diameter and may be diffuse, macula-predominant, macula-sparing, or asymmetric. In the early stages, the disease is marked by retinal vascular leakage, followed by the development of the characteristic birdshot lesions. Advanced disease is associated with cystoid macular edema (CME)—reported in up to 84% of BSCR cases compared with approximately 30% in other forms of uveitis[2]—as well as vascular attenuation, retinal pigment epithelium (RPE) changes, optic nerve atrophy, and subretinal neovascularization.
BSCR was first described in 1949. The disease has been called:
- ‘la chorioretinite en tache de bougie’’ (Candle Wax Spot Chorioretinopathy) by Drs. Franceschetti and Bable in 1949
- Birdshot Retinochoroidopathy by Drs. Ryan and Maumenee in 1980
- Vitiliginious Chorioretinitis by Dr. Gass in 1981
- Salmon Patch Choroidopathy by Dr. Aaberg in 1981
- Rice Grain Chorioretinopathy by Dr. Amalric and Cuq in 1981
Risk Factors
BSCR predominantly affects individuals of Northern European descent between 40 and 60 years of age.[3] It has the strongest known association with a human class I major histocompatibility complex (MHC) allele of any disease, with 80–98% of affected patients testing positive for HLA-A29, compared with approximately 7% of the general population.[4] The presence of this allele confers a 50- to 224-fold increased relative risk of developing the disease.[5]
Pathophysiology
The pathophysiology of BSCR remains unclear, though it is widely regarded as a T-cell–mediated autoimmune disorder. One proposed mechanism involves an immune response directed against retinal S antigens; however, at least one study has demonstrated no significant difference in serum anti–S antigen titers between affected patients and controls.[6] An alternate theory suggests that an infectious trigger stimulates T lymphocytes to express self-peptides. Histopathologic examination of an HLA-A29–positive eye has revealed multiple foci of lymphocytic infiltration at various levels of the choroid, surrounding retinal blood vessels, and within the prelaminar optic nerve head.[7] Inflammatory exudates may infiltrate the choroidal cleavage plane, undergo fibrosis, fuse the choroidal interstitium, and result in atrophic lesions.
Diagnosis
History
The most common symptoms include decreased vision (68%), floaters (29%), nyctalopia (25%), dyschromatopsia (20%), glare (19%), and photopsia (17%).[2] Vision loss typically becomes more pronounced in the later stages of the disease. Although BSCR is primarily an ocular condition, reported systemic associations include hypertension, vitiligo, cutaneous malignancies, hearing loss, and mood disorders. Symptoms are often nonspecific, and patients may describe only a vague sense that “something is not right,” which can result in a substantial delay in diagnosis.[8]
Physical Examination
On examination, patients typically exhibit mild or absent anterior chamber inflammation with absent to moderate vitritis. In early disease, retinal vascular leakage may be the only detectable abnormality. As the disease progresses, the characteristic multifocal, cream-colored to yellow-orange, oval or round choroidal lesions emerge, often originating around the optic nerve. Advanced stages of BSCR may be accompanied by CME, vascular attenuation, subretinal neovascularization, and optic nerve atrophy.
Diagnostic Procedures
In 2021, the Standardization of Uveitis Nomenclature (SUN) Working Group updated classification criteria for BSCR using a machine learning model based on 1,068 cases of posterior uveitis, including 207 cases of BSCR. [9]
Criteria 1- 3 or 4:
- Characteristic bilateral multifocal choroiditis on ophthalmoscopy: multifocal cream-colored or yellow-orange, oval or round choroidal lesions (“birdshot spots”)
- Absent to mild anterior chamber inflammation: absent to mild anterior chamber cells, no keratic precipitates, and no posterior synechiae
- Absent to moderate vitritis OR
- Multifocal choroiditis with Positive HLA-A29 test AND either of the following:
- Characteristic “birdshot” spots (multifocal cream-colored or yellow-orange, oval or round choroidal lesions) on ophthalmoscopy
- Characteristic indocyanine green angiogram (multifocal hypofluorescent spots) without characteristic “birdshot” spots on ophthalmoscopy
The following exclusions apply:
- Positive serologic test for syphilis using a treponemal test
- Evidence of sarcoidosis (either bilateral hilar adenopathy on chest imaging or tissue biopsy demonstrating non-caseating granulomata)
- Evidence of intraocular lymphoma on diagnostic vitrectomy or tissue biopsy
While BSCR is diagnosed clinically, diagnostic testing and multimodal imaging can play an important role in identifying patients who require further evaluation and monitoring of treatment. The Multimodal Imaging in Uveitis (MUV) Taskforce Report further highlights key diagnostic features, recommending fluorescein angiography (FA) for monitoring retinal vascular leakage and evaluating treatment response, as well as indocyanine green angiography (ICG) for detecting choroidal involvement.[10]
Laboratory Tests: Initially, common causes of uveitis should be considered and ruled out. Complete blood count (CBC), RPR/FTA-ABS, ACE, lysozyme chest X-ray, and tuberculin purified protein derivative (PPD) or QuantiFERON Gold testing may be indicated.
Ancillary Tests:
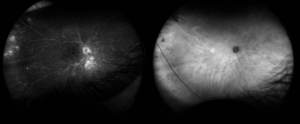
- Fundus Autofluorescence (FAF) can demonstrate peripapillary confluent hypo-autofluorescence which has been shown to be present in 73% of eyes and is associated with chronicity and severity of BSCR.[11] Linear hypo-autofluorescence streaks along retinal vessels and an arcuate pattern of hypo-autofluorescence at the posterior pole have also been reported.[11][12]
- Fluorescein Angiography (FA) may reveal macular edema and hyperfluorescent choroidal lesions. However, Indocyanine Green Angiography (ICG) is more sensitive compared to fundoscopy and FA in revealing the choroidal lesions, especially early in the course of disease .[13]
- Optical Coherence Tomography (OCT) can demonstrate the choroidal lesions and macular edema. It can be used to quantify retinal thinning and loss of the third highly reflective band (HRB), both of which have been associated with worse visual outcomes.[3] [14]
- OCT Angiography (OCTA) can identify abnormal flow signals, similar to those seen on ICGA.[15] Hypo-reflectivity in the macular and peripapillary regions on enhanced depth OCT (ED-OCT), which enables the analysis of deeper choroidal levels, is another common finding in BSCR, and it has been reported in up to 64% of patients.[16]
- Visual Field Testing: Patients can experience significant visual field defects despite well-preserved central VA, including diffuse constriction, paracentral scotomas, and blind spot enlargement.
- Visual Electrodiagnostic Testing: In earlier stages of BSCR, a disproportionate decrease in b-wave amplitude relative to a-wave amplitude may be present. With progression of disease changes to inner retinal function of cone and rod systems presents with a delayed cone mediated 30 Hz flicker on electroretinograms, which is the most sensitive method for the assessment and monitoring of BSCR using electrodiagnostic testing.[17]
Differential Diagnosis
Autoimmune (or Presumed Autoimmune)
- Sarcoidosis
- Acute Posterior Multifocal Placoid Pigment Epitheliopathy (APMPPE)
- Multiple Evanescent White Dot Syndrome (MEWDS)
- Multifocal Choroiditis (MFC)
- Punctate Inner Choroidopathy (PIC)
Infectious
Masquerade
- Primary CNS Lymphoma
Management
General Treatment
The mainstay treatment in BSCR is corticosteroids. Co-administering immunosuppressive therapy with steroids has been shown to be advantageous in preserving visual function and reducing the adverse effects associated with high doses of corticosteroids, when compared to corticosteroid therapy alone.[18] If left untreated, most patients will experience a progressive decline in visual function.
Steroids
- Oral steroids: less than 15% remained symptom-free on <20mg/d
- Local steroid injection: often used in those who have persistent macular edema despite systemic therapy or those who cannot tolerate or inadequately respond to systemic therapy. In BSCR particularly, steroid implants have been associated with a robust IOP response compared to other types of uveitis with up to 40% of patient requiring trabeculectomy surgery and all patient requiring cataract surgery. [19][20]
Immunomodulatory Therapy (IMT)
- Calcineurin inhibitors, which inhibit T-cell signaling, and anti-metabolites are most often the first IMTs considered.
- Mycophenolate mofetil has been shown to be more effective compared to other antimetabolites in treating posterior uveitis and panuveitis.[21] However, a randomized clinical trial of methotrexate and mycophenolate mofetil has revealed statistically insignificant higher treatment success in methotrexate for posterior and panuveitis in general.[22]
Biologics
If patients fail to respond to conventional IMT, escalation to treatment with biologics is indicated. Tumor necrosis factor (TNF)-alpha inhibitors, such as infliximab and adalimumab, have shown effectiveness in treating some forms of refractory uveitis, including BSCR.[23][24][25] A study of adalimumab demonstrated improvement of visual acuity and successful tapering of concomitant IMT; however, complete remission of disease is challenging with adalimumab alone.[25] Successful use of other classes of biologics, such as tocilizumab, have been reported, but current evidence for their use in BSCR is limited. [26]
Treatment Success
Treatment success with immunosuppression after one year of therapy is estimated to be 67% to 90%.[27] Single or dual agent immunosuppression has been successfully used to control BSCR while sparing steroid use to minimize systemic side effects. Steroid sparing, defined as successfully tapering Prednisone dose to < 7.5mg, is generally considered safe for long term use and is possible within 6 months. A recent study suggests that up to 90% of patients can achieve steroid sparing, and as many as 75% of patients can completely discontinue steroid use. [27] However, over 40% of patients can have reactivation of BSCR during tapering, requiring the use of a second IMT. Once steroid tapering is achieved, it may still be necessary to continue treatment with higher doses of IMT for at least 2 years before tapering these immunosuppressive agents to decrease the risk of relapse after remission induction.[27]
Prognosis
Studies have demonstrated a strong correlation between visual acuity at disease onset and long-term visual prognosis.[2] Approximately 97.5% of patients may have some visual symptom at baseline, with 44% having an abnormal visual field and 50-76% with abnormal EOG[3][28]. Without treatment, 16-22% of patients will developed VA ≤ 20/200 over 10 years (versus 4% in other types of uveitis). With the use of IMT, visual acuity remained stable or improved in 78.6-89.3% while visual fields improved from a loss of 56-107° per year to a gain of 30-53° per year.[18][29]
References
- ↑ Bousquet E, Duraffour P, Debillon L, Somisetty S, Monnet D, Brézin AP. Birdshot Chorioretinopathy: A Review. J Clin Med. 2022 Aug 16;11(16):4772. doi: 10.3390/jcm11164772.
- ↑ 2.0 2.1 2.2 Rothova A, Berendschot TT, Probst K, van Kooij B, Baarsma GS. Birdshot chorioretinopathy: long-term manifestations and visual prognosis. Ophthalmology. 2004 May;111(5):954-9. PubMed PMID: 15121374.
- ↑ 3.0 3.1 3.2 Monnet D, Brézin AP, Holland GN, et al. Longitudinal Cohort Study of Patients With Birdshot Chorioretinopathy. I. Baseline Clinical Characteristics. Am J Ophthalmol. 2006;141(1):135-142. doi:10.1016/j.ajo.2005.08.067
- ↑ Kiss S, Anzaar F, Stephen Foster C. Birdshot retinochoroidopathy. Int Ophthalmol Clin. 2006 Spring;46(2):39-55.
- ↑ American Academy of Ophthalmology. "Birdshot Retinochoroidopathy." Section 9: Intraocular Inflammation and Uveitis. Singapore, 2011-2012. 152-155.
- ↑ LeHoang P, Cassoux N, George F, Kullmann N, Kazatchkine MD. Intravenous immunoglobulin (IVIg) for the treatment of birdshot retinochoroidopathy. Ocul Immunol Inflamm. 2000 Mar;8(1):49-57. PubMed PMID: 10806434.
- ↑ Gaudio PA, Kaye DB, Crawford JB. Histopathology of birdshot retinochoroidopathy.Br J Ophthalmol. 2002 Dec;86(12):1439-41.
- ↑ Taylor S, Menezo V. Birdshot uveitis: current and emerging treatment options. Clin Ophthalmol. Published online December 2013:73. doi:10.2147/OPTH.S54832
- ↑ Standardization of Uveitis Nomenclature (SUN) Working Group. Development of classification criteria for birdshot chorioretinitis. Ophthalmology. 2021;128(11):1624-1630. doi:10.1016/j.ophtha.2021.05.028.
- ↑ Pichi F, Miserocchi E, Grewal DS, Sharma S, Brézin AP, Bodaghi B, Agarwal A, Jabs DA, Fawzi A, Sarraf D, Gupta V; MULTIMODAL IMAGING IN UVEITIS (MUV) TASKFORCE. Evidence and Consensus-based Imaging Guidelines in Birdshot Chorioretinopathy: Multimodal Imaging in Uveitis (MUV) Taskforce Report 8. Am J Ophthalmol. 2025 Oct;278:271-281.
- ↑ 11.0 11.1 Böni C, Thorne JE, Spaide RF, et al. Fundus Autofluorescence Findings in Eyes With Birdshot Chorioretinitis. Investig Opthalmology Vis Sci. 2017;58(10):4015. doi:10.1167/iovs.17-21897
- ↑ Koizumi H, Pozzoni MC, Spaide RF. Fundus Autofluorescence in Birdshot Chorioretinopathy. Ophthalmology. 2008;115(5):e15-e20. doi:10.1016/j.ophtha.2008.01.025
- ↑ Fardeau C, Herbort CP, Kullmann N, Quentel G, LeHoang P. Indocyanine green angiography in birdshot chorioretinopathy. Ophthalmology. 1999;106(10):1928-1934. doi:10.1016/S0161-6420(99)90403-7
- ↑ Shields CL, Materin MA, Walker C, Marr BP, Shields JA. Photoreceptor Loss Overlying Congenital Hypertrophy of the Retinal Pigment Epithelium by Optical Coherence Tomography. Ophthalmology. 2006;113(4):661-665. doi:10.1016/j.ophtha.2005.10.057
- ↑ Pepple KL, Chu Z, Weinstein J, Munk MR, Van Gelder RN, Wang RK. Use of En Face Swept-Source Optical Coherence Tomography Angiography in Identifying Choroidal Flow Voids in 3 Patients With Birdshot Chorioretinopathy. JAMA Ophthalmol. 2018;136(11):1288-1292.
- ↑ Böni C, Thorne JE, Spaide RF, et al. Choroidal Findings in Eyes With Birdshot Chorioretinitis Using Enhanced-Depth Optical Coherence Tomography. Investig Opthalmology Vis Sci. 2016;57(9):OCT591. doi:10.1167/iovs.15-18832
- ↑ Sobrin L, Lam BL, Liu M, Feuer WJ, Davis JL. Electroretinographic Monitoring in Birdshot Chorioretinopathy. Am J Ophthalmol. 2005;140(1):52.e1-52.e18. doi:10.1016/j.ajo.2005.01.053
- ↑ 18.0 18.1 Kiss S, Ahmed M, Letko E, Foster C. Long-term Follow-up of Patients with Birdshot Retinochoroidopathy Treated with Corticosteroid-Sparing Systemic Immunomodulatory Therapy. Ophthalmology. 2005;112(6):1066-1071.e2. doi:10.1016/j.ophtha.2004.12.036
- ↑ Burkholder BM, Wang J, Dunn JP, Nguyen QD, Thorne JE. POSTOPERATIVE OUTCOMES AFTER FLUOCINOLONE ACETONIDE IMPLANT SURGERY IN PATIENTS WITH BIRDSHOT CHORIORETINITIS AND OTHER TYPES OF POSTERIOR AND PANUVEITIS. Retina. 2013;33(8):1684-1693. doi:10.1097/IAE.0b013e31828396cf
- ↑ Jaffe GJ, Martin D, Callanan D, Pearson PA, Levy B, Comstock T. Fluocinolone Acetonide Implant (Retisert) for Noninfectious Posterior Uveitis. Ophthalmology. 2006;113(6):1020-1027. doi:10.1016/j.ophtha.2006.02.021
- ↑ Galor A, Jabs DA, Leder HA, et al. Comparison of Antimetabolite Drugs as Corticosteroid-Sparing Therapy for Noninfectious Ocular Inflammation. Ophthalmology. 2008;115(10):1826-1832. doi:10.1016/j.ophtha.2008.04.026
- ↑ Rathinam SR, Babu M, Thundikandy R, et al. A Randomized Clinical Trial Comparing Methotrexate and Mycophenolate Mofetil for Noninfectious Uveitis. Ophthalmology. 2014;121(10):1863-1870. doi:10.1016/j.ophtha.2014.04.023
- ↑ Artornsombudh P, Gevorgyan O, Payal A, Siddique SS, Foster CS. Infliximab Treatment of Patients with Birdshot Retinochoroidopathy. Ophthalmology. 2013;120(3):588-592. doi:10.1016/j.ophtha.2012.05.048
- ↑ Suhler EB. A Prospective Trial of Infliximab Therapy for Refractory Uveitis: Preliminary Safety and Efficacy Outcomes. Arch Ophthalmol. 2005;123(7):903. doi:10.1001/archopht.123.7.903
- ↑ 25.0 25.1 Huis in het Veld PI, van Asten F, Kuijpers RWAM, Rothova A, de Jong EK, Hoyng CB. ADALIMUMAB THERAPY FOR REFRACTORY BIRDSHOT CHORIORETINOPATHY. Retina. 2019;39(11):2189-2197. doi:10.1097/IAE.0000000000002281
- ↑ Muselier A, Bielefeld P, Bidot S, Vinit J, Besancenot JF, Bron A. Efficacy of Tocilizumab in Two Patients with anti-TNF-alpha Refractory Uveitis. Ocul Immunol Inflamm. 2011;19(5):382-383. doi:10.3109/09273948.2011.606593
- ↑ 27.0 27.1 27.2 Crowell EL, France R, Majmudar P, Jabs DA, Thorne JE. Treatment Outcomes in Birdshot Chorioretinitis. Ophthalmol Retina. 2022;6(7):620-627. doi:10.1016/j.oret.2022.03.003
- ↑ Gasch AT, Smith JA, Whitcup SM. Birdshot retinochoroidopathy. Br J Ophthalmol. 1999 Feb;83(2):241-9.
- ↑ Thorne JE, Jabs DA, Kedhar SR, Peters GB, Dunn JP. Loss of visual field among patients with birdshot chorioretinopathy. Am Jo Ophthalmol. 2008 Jan;145(1):23-28. Epub 2007 Nov 12