Pleomorphic Adenoma (Benign Mixed Tumor) of the Lacrimal Gland
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Pleomorphic adenoma of the lacrimal gland (PALG), also known as benign mixed tumor, is a rare tumor of the lacrimal gland.[1] PALG typically presents with unilateral painless proptosis. Affected individuals may be unaware of the onset of proptosis because it is typically gradual in onset. It is important to differentiate PALG from adenoid cystic carcinoma (ACC) because of their vastly different prognosis and treatment. Diagnosis is bases on clinical characteristics and radiologic findings, and can usually be made prior to surgical intervention. Biopsy may be appropriate in cases of a diagnostic dilemma, although the biopsy track must also be excised if the diagnosis of PALG is made.[2] Definitive treatment of PALG is complete excision due to common recurrence and malignant transformation with incomplete excision.
Disease Entity
International Classification of Disease (ICD)
Definition
Pleomorphic adenoma is a neoplastic proliferation of epithelial cells that form characteristic ductal structures with surrounding myoepithelial cells.[3] These structures gradually trail out into myxomatous mesenchyme of the lacrimal gland.
Epidemiology
Lacrimal gland tumors represent approximately 9% of all orbital masses. Of those 9%, approximately 10% are PALG.[1]Among salivary glands, however, pleomorphic adenoma is the most common type of tumor, occurring most commonly in the parotid gland (80%)[4]
Risk Factors
The most common age of onset of PALG is in the 5th and 6th decades, with a mean age of diagnosis of 48 in one large case series.[1] PALG does not seem to have a male or female preponderance. Because of the rarity of this tumor, the risk factors have not been elucidated.
Pathology and Pathophysiology
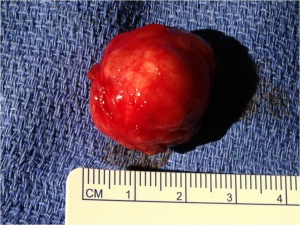
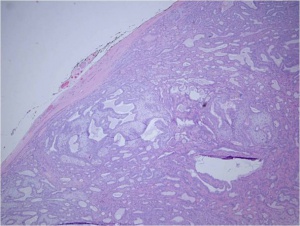
The gross tumor is typically has a pseudocapsule of surrounding tissue, without invasion. (see figure 1).Histologic sections reveal ductal epithelial cells with surrounding myoepithelial cells. The cells have benign cytology (see figures 2 and 3). The exact cause of PALG is unknown. Genetic rearrangements, such as chromosomal translocations involving 8q12 target the PLAG1 gene, may contribute to the pathophysiology of PALG. Alterations in this developmentally regulated zinc finger gene can lead to deregulation of target genes, including IGF2, and is likely to play a major role in the genesis of pleomorphic adenomas.[4]
Diagnosis
History
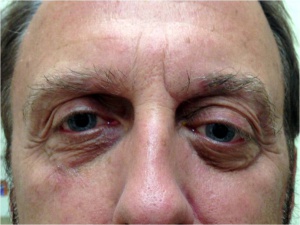
Patients usually present with unilateral gradual onset proptosis. It is often useful to look at old photos of the patient to observe changes in eye position. The location of PALG in the lacrimal fossa typically leads to an inferior and medial displacement of the globe. A full orbit may also be observed in comparison to the unaffected side (see figure 4). Pain may be present, but is more common in patient presenting with adenoid cystic carcinoma.
Physical Examination
A full eye examination should be performed to determine the extent of ocular involvement prior to any surgical intervention. Color vision and visual field testing assist in identifying any optic nerve involvement, which is rare in this condition. Additionally, a Schirmer test may be useful to document the production of the serous tear layer which is sure to decrease after removal of the lacrimal gland. Hertel exophthalmometry often reveals proptosis of >18mm or >2 mm difference between the two eyes. Often the enlarged lacrimal gland can be palpated under the superior orbital rim. Retropulsion of the globe will also be limited in comparison to the contralateral globe. Symptoms may include diplopia, visual changes, increased tear production, discomfort, or ptosis.[5]
Diagnostic Procedures
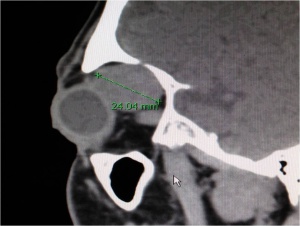
CT scan of the orbits with contrast enhancement is the radiologic study of choice for the diagnosis of PALG. Thin sections through the orbits highlight bony tissue which is critical in differentiating PALG from ACC. Features of PALG on CT include: solid (can be heterogenous), well defined, round or oval, occasional calcification, and bony remodeling (see figures 5-7). In contrast, ACC typically features: irregular margins (although can be well circumscribed), nodularity (with infiltration of adjacent tissue), calcification, and bony destruction.[6]
Differential diagnosis
- Lymphoproliferative neoplasm
- Non-specific orbital inflammation
- Dacryoadenitis
- Sarcoidosis
- Mikulich’s Disease
- Lacrimal Duct Cyst (Dacryops)
- Epithelial Mass
- Benign
- Pleomorphic adenoma with atypia
- myoepithelioma
- oncocytoma
- cystadenoma
- Malignant
- Adenoid cystic carcinoma
- carcinoma ex pleomorphic adenoma
- mucoepidermoid carcinoma
- adenocarcinoma
- undifferentiated carcinoma
- Benign
Management
Patients who are suspected to have a lacrimal gland mass such as a PALG or ACC should be referred to an Ophthalmic Plastic and Reconstructive Surgeon or other specialist who is familiar with the diagnosis and treatment of this tumor. If it is considered likely that the patient has a PALG based on clinical and radiologic evidence, complete excision of the mass is indicated. Several studies have demonstrated that incomplete excision can lead to recurrence or malignant transformation of adenoid cystic carcinoma.[7],[8],[9],[10],[11] Biopsy may be performed in cases where clinical and radiologic evidence do not clinch the diagnosis of PALG [2]
Medical therapy
At this time there is no medical therapy indicated for the treatment of pleomorphic adenoma.
Surgery
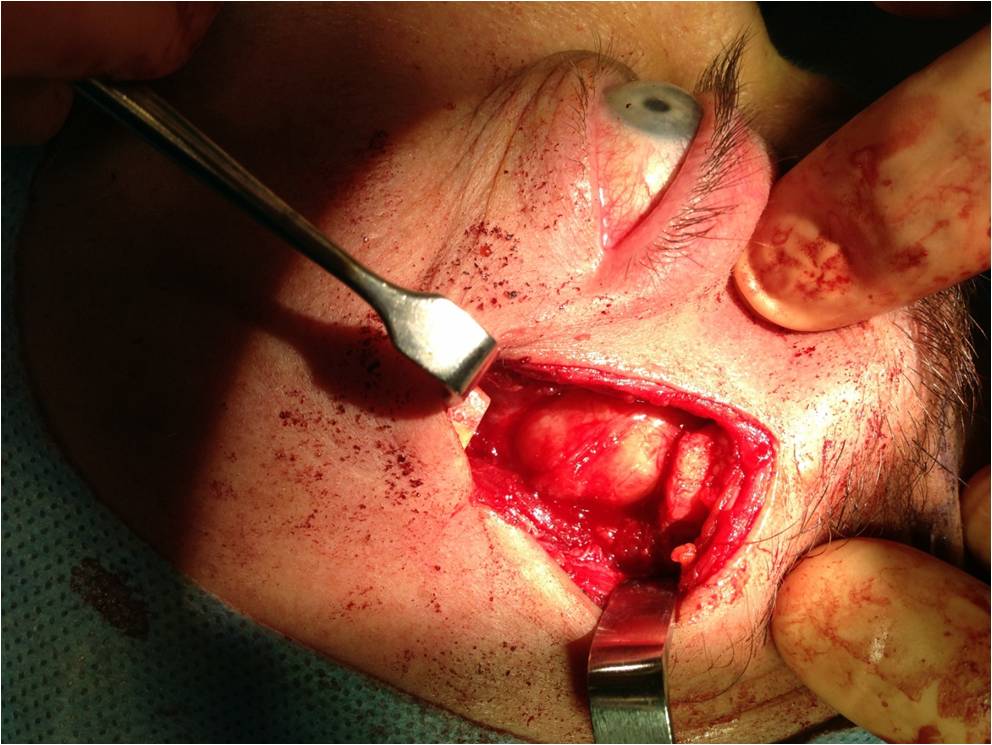
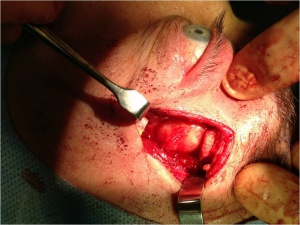
Curative treatment of PALG is complete excision with an intact pseudocapsule. In order to achieve complete excision of a lacrimal fossa tumor, a lateral orbitotomy approach is required. This surgery is performed under general anesthesia. Surgical techniques vary, but generally, the following steps are performed: a curvilinear incision (Stallard-Wright) is made over the lateral orbital wall (frontal and zygomatic bones). Temporalis fossa is exposed using periosteal elevators. The periorbital is elevated from the lateral wall and roof of the orbit with a malleable retractor. The lateral orbital rim is cut in two places using an oscillating bone saw. The lateral orbital rim is then outfractured using a large double-action Luer or Bull-nose rongeur. Using a combination of blunt and sharp dissection, the lacrimal gland is mobilized onto an island of free periorbita. Care is taken to avoid the levator and lateral rectus muscles. The gland should not be touched with forceps, always using digits or malleable retractors to mobilize it. Eventually the mass can be freed from the surrounding tissue with the pseudocapsule intact(see figure 8). [5],[12]
Surgical follow up
It may be prudent to observe the patient overnight for vision checks in cases where deep orbital dissection has been performed. The patient is typically evaluatated on postoperative day 1 to check vision, intraocular pressure, and pupils. Patient should avoid straining or applying pressure to the eye in the immediate postoperative period.
Complications
Possible complications are dependent on surgical technique, and may include: Infection, hemorrhage, diplopia or strabismus, vision loss, eyelid malposition, sensory hypoesthesia, dry eye, pupillary or accomidative paresis, cerebrospinal fluid leakage, orbital compartment syndrome. [12]
Prognosis
With complete excision, long term risk of recurrence is negligible. Follow-up may be on an as-needed basis rather than regular checkups.[13]
References
- ↑ 1.0 1.1 1.2 Shields JA, Shields CL, Rcartozzi R. Survey of 1264 Patients with Orbital Tumors and Simulating Lesions. Ophthalmology 2004;111:997-1008
- ↑ 2.0 2.1 Lai T, Prabhakaran VC, Malhortra R, Selva D. Pleomorphic adenoma of the lacrimal gland: is there a role for biopsy? Eye. 2009;23,2-6
- ↑ Weis E, Rootman J, Joly TJ, et al. Epithelial Lacrimal Gland Tumors. Arch Ophthalmol. 2009;127(8):1016-1028.
- ↑ 4.0 4.1 Eveson JW, Kusafuka K, Stenman G, Nagao T. “Pleomorphic Adenoma” Pathology and Genetics of Head and Neck Tumors. 2005;253-258
- ↑ 5.0 5.1 Rose GE, Wright JE. Pleomorphic adenoma of the lacrimal gland. British Journal of Ophthalmology. 1992;76,395-400
- ↑ Vaidhyanath R, Kirke R, Brown L, Sampath R. Lacrimal Fossa Lesions: Pictorial Review of CT and MRI Features. Orbit, 2008; 27:410-418
- ↑ Font RL, Gamel JW. Epithelial tumors of the lacrimal gland: an analysis of 265 cases. In: Jakobiec FA, ed. Ocular and Adnexal Tumors. Birmingham, AL: Aesclulapius Publishers Inc; 1978:787-805
- ↑ Ni C, Cheng SC, Dryja TP, Cheng TY. Lacrimal gland tumors: a clinicopathological analysis of 160 cases. Int Ophthalmol Clin. 1982;22(1):99-120
- ↑ Wright JE, Steward WB, Krohel GB. Clinical presentation and management of lacrimal gland tumors. Br J Ophthalmol. 1979;63(9):600-606.
- ↑ Wright JE. Factors affecting the survival of patients with lacrimal gland tumors. Can J Ophthalmol. 1982;17(1):3-9
- ↑ Mensink HW, Mooy CM, Paridaens D. In situ adenocarcinoma ex pleomorphic adenoma of the lacrimal gland. Clin Experiment Ophthalmol. 2005;33(6):669-671.
- ↑ 12.0 12.1 Turbin RE. Lateral Orbitotomy. In: Dunn JP and Langer PD ed. Basic Techniques of Ophthalmic Surgery. 2009. Chapter 72:434-443
- ↑ Currie ZI, Rose GE. Long-term risk of recurrence after intact excision of Pleomorphic Adenomas of the Lacrimal Gland. Arch Ophthalmol. 2007;125(12):1643-1646