Ocular Adverse Reactions after Receiving COVID-19 Vaccine
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Introduction
The coronavirus disease 2019 (COVID-19) pandemic is an ongoing global health crisis that is caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). As of July 31, 2022, there have been over 570 million confirmed cases of COVID-19 and over 6 million deaths worldwide[1]. COVID-19 vaccinations are highly effective at preventing severe, symptomatic disease[2][3][4]. Although modest reductions in vaccine effectiveness have been observed with the emergence of variants, booster doses can provide adequate levels of protection[5]. Despite the efficacy of COVID-19 vaccines, vaccine hesitancy remains high with a global vaccine acceptance rate ranging from 54% to 86%[6]. Many of the concerns stem from perceived harms of receiving the vaccination. Indeed, uncommon but potentially fatal complications such as immune thrombotic thrombocytopenia and cerebral venous sinus thrombosis have been reported following COVID-19 vaccination[7]. Ocular adverse events following COVID-19 vaccines, although exceedingly rare, have also been documented[8][9]. The immune response induced by the vaccine is considered a pathophysiological mechanism of ocular adverse reaction following the COVID-19 vaccination. [10] In this article, we summarize the various ocular adverse events following COVID-19 vaccination reported in the peer-reviewed literature.
Ocular Inflammatory Diseases
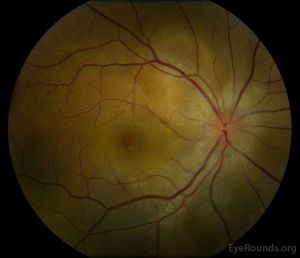
Multiple case reports have documented anterior uveitis following both the first and second doses of COVID-19 vaccines[8][9]. Although some patients had a previous history of uveitis-related diseases that may have predisposed them to this phenomenon, many notably did not[11]. Cases of scleritis, episcleritis, and multiple evanescent white dot syndrome, following COVID-19 vaccination have also been reported[9].. Most of these cases resolved with topical, intravitreal, and/or systemic corticosteroid therapy with complete recovery to baseline best corrected visual acuity (BCVA). Both recurrent and new onset cases of Vogt-Koyanagi-Harada disease following COVID-19 vaccination have also been reported[12][13]. These events resolved with oral and periocular steroids, respectively. Panuveitis[14], choroiditis[15], and central serous chorioretinopathy[16] impacting the choroid were also reported reactions associated with COVID-19 vaccination. Patients were treated with steroids and most were able to reach their baseline BCVA[14][15][16] . A case series of 3 cases with orbital inflammation following mRNA vaccines were described and all cases were successfully treated with oral prednisolone. [17] A case of dacryoadenitis following the COVID-19 vaccination was described with subsequent treatment with oral prednisolone and resolution of signs and symptoms during 2 months. [18]
Optic Neuropathy
Published literature to date demonstrates very few post-COVID-vaccination cases of optic neuritis[19][20][21][22] and ischemic optic neuropathy[23][24]. Optic neuritis cases typically resolved after several days of intravenous methylprednisolone therapy to near-baseline visual acuity. A case of arteritic anterior ischemic optic neuropathy (AAION)[23] and a case of non-arteritic anterior ischemic optic neuropathy (NA-AION)[24] were reported after the second and first mRNA vaccine doses, respectively. AAION was treated with high-dose oral corticosteroids and tocilizumab therapy[23] , while the NA-AION resolved without treatment[24]. It should be noted that there are no peer-reviewed cases of adverse COVID-19 vaccination effects in glaucoma patients[9].
Anterior Segment Conditions
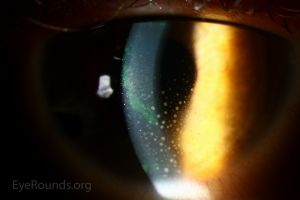
Cases of Herpes zoster ophthalmicus have been reported after both the first and second doses of COVID-19 vaccines[19][20]. Very few cases reported a previous history of herpes zoster ophthalmicus[21] and many documented a notable lack of vesicular rash accompanying ocular manifestations[22][23] . All cases resolved with steroid and antiviral treatment. Further, corneal graft rejections were also associated with COVID-19 vaccines[24][25]. Several patients notably had prior histories of Descemet membrane endothelial keratoplasty (DMEK)[26][27][28][29] or penetrating keratoplasty (PKP)[26][30][31]. Common findings consisted of corneal edema, corneal neovascularization, and keratic precipitates[26][27][32][33][34]. Steroid treatment was initiated for patients, with mixed results ranging from improved corneal graft clarity and visual acuity[33][35] to lasting corneal defects[34][35] . Keratitis has also been reported as an adverse event following COVID-19 vaccines[36][37][38]. One case of a patient with a previous history of PKP further described a reactivation of herpes simplex keratitis shortly after vaccination[36]. Antibiotics and corticosteroid treatment were used to resolve symptoms and visual acuity improved to near baseline levels[36][37]. One case of bilateral immune-mediated keratolysis was associated with vaccination, which was treated with tectonic penetrating keratoplasty and post-operative steroid antibiotics[39].
Retinal Conditions
Acute macular neuroretinopathy[40][41][42], central retinal artery occlusion[43], central retinal vein occlusion[44][45], central serous chorioretinopathy[46], paracentral acute middle maculopathy[47], and retinal hemorrhage[48] have all been reported in the peer-reviewed literature following COVID-19 vaccination. Disease courses ranged from complete resolution with return to BCVA to permanent vision loss, depending on the severity of disease. Many of these patients lacked medical comorbidities that may have predisposed them to these conditions, although hormone-based birth control was noted in a few patients[41][42]. However, population-based adverse event reporting systems show that vaccination-associated retinal conditions are exceedingly rare, making it difficult to ascertain the causative nature of COVID-19 vaccination on subsequent retinal pathologies[9].
Discussions and Conclusion
COVID-19 vaccines are widely recommended to the general public for prevention of severe disease courses. However, ophthalmologists should be aware of the rare side effects in the eye and should carefully monitor patients with pre-existing conditions that have been associated with the development of ophthalmologic side effects.
References
- ↑ WHO Coronavirus (COVID-19) Dashboard. Accessed March 12, 2022. https://covid19.who.int
- ↑ Baden LR, El Sahly HM, Essink B, et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N Engl J Med. Published online December 30, 2020:NEJMoa2035389. doi:10.1056/NEJMoa2035389
- ↑ Polack FP, Thomas SJ, Kitchin N, et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med. Published online December 10, 2020:NEJMoa2034577. doi:10.1056/NEJMoa2034577
- ↑ Voysey M, Clemens SAC, Madhi SA, et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet. 2021;397(10269):99-111. doi:10.1016/S0140-6736(20)32661-1
- ↑ Andrews N, Stowe J, Kirsebom F, et al. Covid-19 Vaccine Effectiveness against the Omicron (B.1.1.529) Variant. N Engl J Med. 2022;386(16):1532-1546. doi:10.1056/NEJMoa2119451
- ↑ Joshi A, Kaur M, Kaur R, Grover A, Nash D, El-Mohandes A. Predictors of COVID-19 Vaccine Acceptance, Intention, and Hesitancy: A Scoping Review. Front Public Health. 2021;9:698111. doi:10.3389/fpubh.2021.698111
- ↑ Sharifian-Dorche M, Bahmanyar M, Sharifian-Dorche A, Mohammadi P, Nomovi M, Mowla A. Vaccine-induced immune thrombotic thrombocytopenia and cerebral venous sinus thrombosis post COVID-19 vaccination; a systematic review. J Neurol Sci. 2021;428:117607. doi:10.1016/j.jns.2021.117607
- ↑ 8.0 8.1 Ng XL, Betzler BK, Testi I, et al. Ocular Adverse Events After COVID-19 Vaccination. Ocul Immunol Inflamm. 2021;29(6):1216-1224. doi:10.1080/09273948.2021.1976221
- ↑ 9.0 9.1 9.2 9.3 9.4 Wang MTM, Niederer RL, McGhee CNJ, Danesh-Meyer HV. COVID-19 Vaccination and The Eye. American Journal of Ophthalmology. 2022;240:79-98. doi:10.1016/j.ajo.2022.02.011
- ↑ Ng XL, Betzler BK, Ng S, Chee SP, Rajamani L, Singhal A, Rousselot A, Pavesio CE, Gupta V, de Smet MD, Agrawal R. The Eye of the Storm: COVID-19 Vaccination and the Eye. Ophthalmol Ther. 2022 Feb;11(1):81-100. doi: 10.1007/s40123-021-00415-5. Epub 2021 Dec 16. PMID: 34914035; PMCID: PMC8675299.
- ↑ Rabinovitch T, Ben-Arie-Weintrob Y, Hareuveni-Blum T, et al. UVEITIS AFTER THE BNT162b2 mRNA VACCINATION AGAINST SARS-CoV-2 INFECTION: A Possible Association. RETINA. 2021;41(12):2462-2471. doi:10.1097/IAE.0000000000003277
- ↑ Papasavvas I, Herbort CP. Reactivation of Vogt-Koyanagi-Harada disease under control for more than 6 years, following anti-SARS-CoV-2 vaccination. J Ophthalmic Inflamm Infect. 2021;11(1):21. doi:10.1186/s12348-021-00251-5
- ↑ Chen X, Wang B, Li X. Acute-onset Vogt-Koyanagi-Harada like uveitis following Covid-19 inactivated virus vaccination. Am J Ophthalmol Case Rep. 2022;26:101404. doi:10.1016/j.ajoc.2022.101404
- ↑ 14.0 14.1 Mudie LI, Zick JD, Dacey MS, Palestine AG. Panuveitis following Vaccination for COVID-19. Ocul Immunol Inflamm. 2021;29(4):741-742. doi:10.1080/09273948.2021.1949478
- ↑ 15.0 15.1 Pan L, Zhang Y, Cui Y, Wu X. Bilateral uveitis after inoculation with COVID-19 vaccine: A case report. Int J Infect Dis. 2021;113:116-118. doi:10.1016/j.ijid.2021.09.075
- ↑ 16.0 16.1 Fowler N, Mendez Martinez NR, Pallares BV, Maldonado RS. Acute-onset central serous retinopathy after immunization with COVID-19 mRNA vaccine. Am J Ophthalmol Case Rep. 2021;23:101136. doi:10.1016/j.ajoc.2021.101136
- ↑ Reshef ER, Freitag SK, Lee NG. Orbital Inflammation Following COVID-19 Vaccination. Ophthalmic Plast Reconstr Surg. 2022 May-Jun 01;38(3):e67-e70. doi: 10.1097/IOP.0000000000002161. Epub 2022 Mar 23. PMID: 35323144; PMCID: PMC9093224.
- ↑ Singh S, Gandhi A, Das S. Dacryoadenitis post COVID-19 infection and immunization. Indian J Ophthalmol. 2023 Aug;71(8):3100-3102. doi: 10.4103/IJO.IJO_30_23. PMID: 37530288; PMCID: PMC10538829.
- ↑ 19.0 19.1 Lee C, Park KA, Ham DI, et al. Neuroretinitis after the second injection of a SARS-CoV-2-vaccine: A case report. Am J Ophthalmol Case Rep. 2022;27:101592. doi:10.1016/j.ajoc.2022.101592
- ↑ 20.0 20.1 Roy M, Chandra A, Roy S, Shrotriya C. Optic neuritis following COVID-19 vaccination: Coincidence or side-effect? - A case series. Indian J Ophthalmol. 2022;70(2):679-683. doi:10.4103/ijo.IJO_2374_21
- ↑ 21.0 21.1 Leber HM, Sant’Ana L, Konichi da Silva NR, et al. Acute Thyroiditis and Bilateral Optic Neuritis following SARS-CoV-2 Vaccination with CoronaVac: A Case Report. Ocular Immunology and Inflammation. 2021;29(6):1200-1206. doi:10.1080/09273948.2021.1961815
- ↑ 22.0 22.1 Helmchen C, Buttler GM, Markewitz R, Hummel K, Wiendl H, Boppel T. Acute bilateral optic/chiasm neuritis with longitudinal extensive transverse myelitis in longstanding stable multiple sclerosis following vector-based vaccination against the SARS-CoV-2. J Neurol. 2022;269(1):49-54. doi:10.1007/s00415-021-10647-x
- ↑ 23.0 23.1 23.2 23.3 Maleki A, Look-Why S, Manhapra A, Foster CS. COVID-19 Recombinant mRNA Vaccines and Serious Ocular Inflammatory Side Effects: Real or Coincidence? J Ophthalmic Vis Res. 2021;16(3):490-501. doi:10.18502/jovr.v16i3.9443
- ↑ 24.0 24.1 24.2 24.3 Tsukii R, Kasuya Y, Makino S. Nonarteritic Anterior Ischemic Optic Neuropathy following COVID-19 Vaccination: Consequence or Coincidence. Case Rep Ophthalmol Med. 2021;2021:5126254. doi:10.1155/2021/5126254
- ↑ Lee YK, Huang YH. Ocular Manifestations after Receiving COVID-19 Vaccine: A Systematic Review. Vaccines (Basel). 2021;9(12):1404. doi:10.3390/vaccines9121404
- ↑ 26.0 26.1 26.2 Shah AP, Dzhaber D, Kenyon KR, Riaz KM, Ouano DP, Koo EH. Acute Corneal Transplant Rejection After COVID-19 Vaccination. Cornea. 2022;41(1):121-124. doi:10.1097/ICO.0000000000002878
- ↑ 27.0 27.1 Crnej A, Khoueir Z, Cherfan G, Saad A. Acute corneal endothelial graft rejection following COVID-19 vaccination. J Fr Ophtalmol. 2021;44(8):e445-e447. doi:10.1016/j.jfo.2021.06.001
- ↑ Phylactou M, Li JPO, Larkin DFP. Characteristics of endothelial corneal transplant rejection following immunisation with SARS-CoV-2 messenger RNA vaccine. Br J Ophthalmol. Published online April 2021:bjophthalmol-2021-319338. doi:10.1136/bjophthalmol-2021-319338
- ↑ Abousy M, Bohm K, Prescott C, Bonsack JM, Rowhani-Farid A, Eghrari AO. Bilateral EK Rejection After COVID-19 Vaccine. Eye Contact Lens. 2021;47(11):625-628. doi:10.1097/ICL.0000000000000840
- ↑ Wasser LM, Roditi E, Zadok D, Berkowitz L, Weill Y. Keratoplasty Rejection After the BNT162b2 messenger RNA Vaccine. Cornea. 2021;40(8):1070-1072. doi:10.1097/ICO.0000000000002761
- ↑ Molero-Senosiain M, Houben I, Savant S, Savant V. Five Cases of Corneal Graft Rejection After Recent COVID-19 Vaccinations and a Review of the Literature. Cornea. 2022;41(5):669-672. doi:10.1097/ICO.0000000000002980
- ↑ Ryu KJ, Kim DH. Recurrence of Varicella-Zoster Virus Keratitis After SARS-CoV-2 Vaccination. Cornea. 2022;41(5):649-650. doi:10.1097/ICO.0000000000002999
- ↑ 33.0 33.1 Nahata H, Nagaraja H, Shetty R. A case of acute endothelial corneal transplant rejection following immunization with ChAdOx1 nCoV-19 coronavirus vaccine. Indian J Ophthalmol. 2022;70(5):1817-1818. doi:10.4103/ijo.IJO_66_22
- ↑ 34.0 34.1 Yu S, Ritterband DC, Mehta I. Acute Corneal Transplant Rejection After Severe Acute Respiratory Syndrome Coronavirus 2 mRNA-1273 Vaccination. Cornea. 2022;41(2):257-259. doi:10.1097/ICO.0000000000002886
- ↑ 35.0 35.1 Simão MF, Kwitko S. Corneal Graft Rejection After Inactivated SARS-CoV-2 Vaccine: Case Report. Cornea. Published online 2022:502-504
- ↑ 36.0 36.1 36.2 Al-Dwairi RA, Aleshawi A, Adi S, Abu-Zreig L. Reactivation of Herpes Simplex Keratitis on a Corneal Graft Following SARS-CoV-2 mRNA Vaccination. Med Arch. 2022;76(2):146-148. doi:10.5455/medarh.2022.76.146-148
- ↑ 37.0 37.1 Farrell DA, Deacon S, Mauger T. “Marginal keratitis following COVID 19 vaccination.” IDCases. 2022;29:e01536. doi:10.1016/j.idcr.2022.e01536
- ↑ Richardson-May J, Rothwell A, Rashid M. Reactivation of herpes simplex keratitis following vaccination for COVID-19. BMJ Case Rep. 2021;14(9):e245792. doi:10.1136/bcr-2021-245792
- ↑ Khan TA, Sidhu N, Khan L, et al. Bilateral Immune-Mediated Keratolysis After Immunization With SARS-CoV-2 Recombinant Viral Vector Vaccine. Cornea. 2021;40(12):1629-1632. doi:10.1097/ICO.0000000000002844
- ↑ Patel SN, Yonekawa Y. ACUTE MACULAR NEURORETINOPATHY AFTER SARS-COV-2 VACCINATION. Retin Cases Brief Rep. 2022;16(1):5-8. doi:10.1097/ICB.0000000000001195
- ↑ 41.0 41.1 Book BAJ, Schmidt B, Foerster AMH. Bilateral Acute Macular Neuroretinopathy After Vaccination Against SARS-CoV-2. JAMA Ophthalmology. 2021;139(7):e212471. doi:10.1001/jamaophthalmol.2021.2471
- ↑ 42.0 42.1 Valenzuela DA, Groth S, Taubenslag KJ, Gangaputra S. Acute macular neuroretinopathy following Pfizer-BioNTech COVID-19 vaccination. American Journal of Ophthalmology Case Reports. 2021;24:101200. doi:10.1016/j.ajoc.2021.101200
- ↑ Lee S, Sankhala KK, Bose S, Gallemore RP. Combined Central Retinal Artery and Vein Occlusion with Ischemic Optic Neuropathy After COVID-19 Vaccination. Int Med Case Rep J. 2022;15:7-14. doi:10.2147/IMCRJ.S328931
- ↑ Endo B, Bahamon S, Martínez-Pulgarín DF. Central retinal vein occlusion after mRNA SARS-CoV-2 vaccination: A case report. Indian J Ophthalmol. 2021;69(10):2865-2866. doi:10.4103/ijo.IJO_1477_21
- ↑ Sonawane NJ, Yadav D, Kota AR, Singh HV. Central retinal vein occlusion post-COVID-19 vaccination. Indian J Ophthalmol. 2022;70(1):308-309. doi:10.4103/ijo.IJO_1757_21
- ↑ Hanhart J, Roditi E, Wasser LM, Barhoum W, Zadok D, Brosh K. Central serous chorioretinopathy following the BNT162b2 mRNA vaccine. J Fr Ophtalmol. 2022;45(6):597-602. doi:10.1016/j.jfo.2022.01.006
- ↑ Malerbi FK, Schoeps VA, T F Matos K. Paracentral acute middle maculopathy in Susac syndrome after dual exposure to SARS-CoV-2 antigen. BMJ Case Rep. 2022;15(5):e247159. doi:10.1136/bcr-2021-247159
- ↑ Amin MA, Nahin S, Dola TA, Afrin S, Hawlader MDH. Retinal hemorrhage of late post-COVID-19 and post-vaccine-related pathogenic mechanisms: A new challenge for ophthalmologist in COVID era. Clin Case Rep. 2022;10(2):e05471. doi:10.1002/ccr3.5471