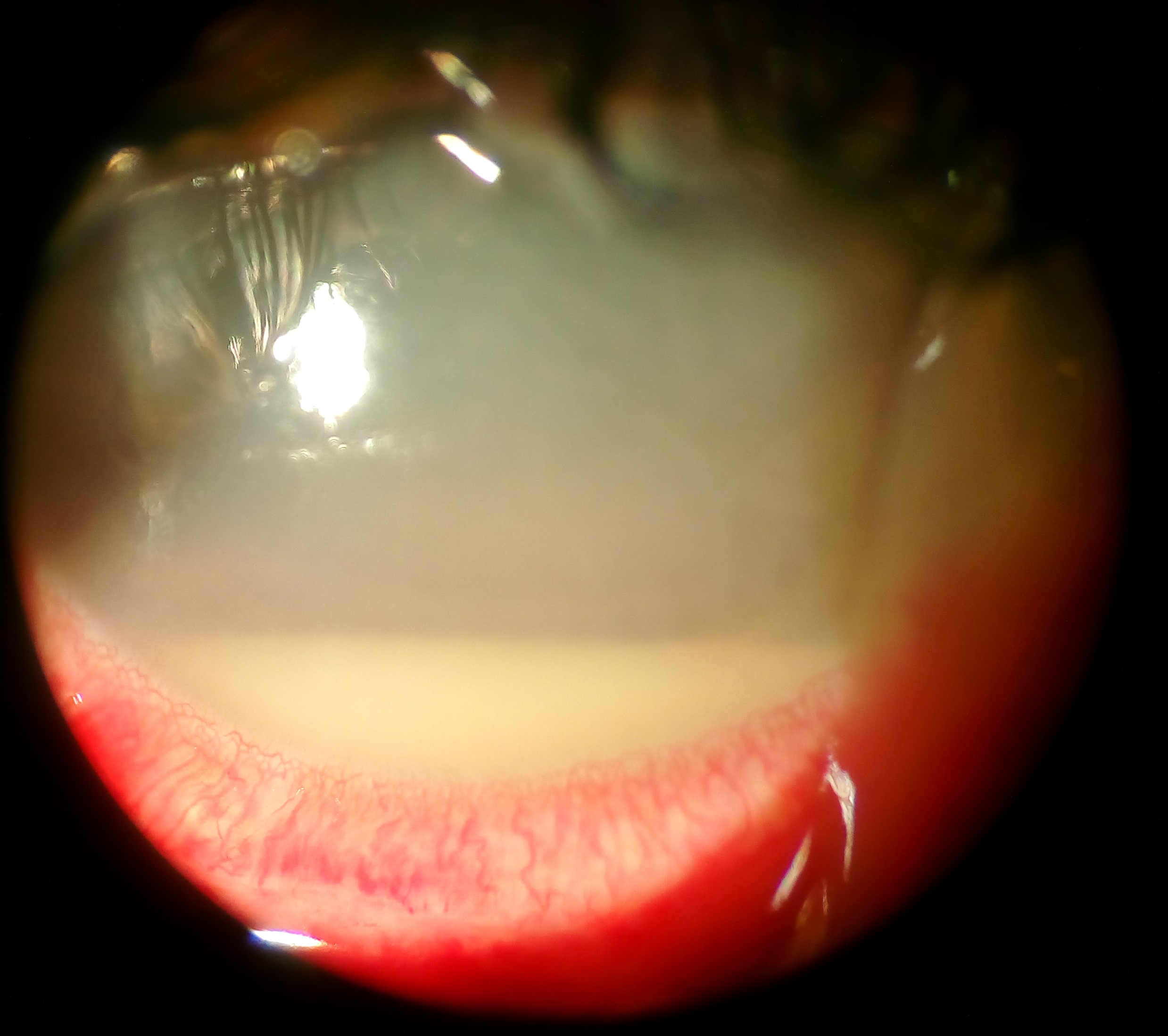
Endophthalmitis
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Definition
Endophthalmitis is a purulent inflammation of the intraocular fluids (vitreous and aqueous), usually due to infection.
- It is a serious intraocular inflammatory disorder resulting from infection of the vitreous cavity.
- Progressive vitritis is the hallmark of any form of endophthalmitis.
- Histologically, there is a massive infiltration of the vitreous cavity with inflammatory cells.
Types of Endophthalmitis[1]
Exogenous
- Acute postoperative
- Chronic postoperative
- Traumatic
- Filtering bleb-associated
- After intravitreal injections
- Corneal ulcer
Endogenous
- Bacterial or fungal endogenous chorioretinitis +/- vitritis
Acute Postoperative Endophthalmitis[2]
- The term refers to infectious endophthalmitis shortly after ocular surgery.
- It generally presents within 1-2 weeks, often 3-5 days after the surgery.
- Initial symptoms are rapidly progressive, including pain, red eye, ocular discharge, and worsening vision.
- Common signs are decreased visual acuity, lid swelling, conjunctival and corneal edema, anterior chamber cells + fibrin, hypopyon, vitreous inflammation, retinitis, retinal hemorrhages, and blunting of the red reflex.
- Retinal periphlebitis is the earliest sign.
Endophthalmitis Vitrectomy Study (EVS)[3]
The EVS evaluated only the patients with acute endophthalmitis that occurred within 6 weeks of cataract surgery or secondary intraocular lens implantation.
Symptoms
- 94.3% reported blurred vision
- 82.1% red eye
- 74% pain - It is important to note that over 25% of patients did not have pain.
- 34.5% swollen lid
- Epiphora
- Photophobia
Signs
- 85% hypopyon - 15% of patients may not have a hypopyon.
- 79% hazy media
- 26% light perception (LP) vision only
When there is uncertainty in the differential diagnosis between postoperative inflammation and endophthalmitis with severe haze in the ocular media or vitreous opacities clinically, the possibility of an infection should be prioritized, given the potentially rapid and devastating consequences of delaying treatment.
Epidemiology
- Acute postoperative endophthalmitis is the most common form of endophthalmitis.
- Incidence following cataract surgery is 0.08% - 0.68%.
- Rates are increasing since the advent of clear corneal incisions.
- The risk is highest after secondary IOL (0.2%-0.367%) implantation, and traditionally lowest after pars plana vitrectomy (0.03%-0.046%). However, recent studies in the microincisional vitrectomy surgery era suggest increased rates up to 0.11%-0.16%.[4][5]
Evaluation
- Suspect endophthalmitis in any eye with inflammation greater than the usual postoperative course
- Associations:
- wound leak or dehiscence, especially a leaking small incision cataract surgery (SICS) wound
- suture abscess
- vitreous incarceration in the wound
- eroding scleral sutures used to fixate intraocular lenses (IOLs)
- exposed tube shunt
Presenting Visual Acuity
- When measuring visual acuity, the technique of differentiating light perception (LP) vs. hand motion (HM) vision is most important.
- Pars plana vitrectomy recommended in patients with visual acuity of LP or worse.
- Tap and inject intravitreal antibiotics in patients with visual acuity of HM or better.
- EVS gave the standard criteria to evaluate visual acuity in endophthalmitis: 'If no letters could be read on the (ETDRS) chart at 4 m, then at 1 m, vision was tested for the ability to count fingers. If the patient was unable to count fingers, vision was tested for the ability to recognize hand motions. For this, the patient's opposite eye was occluded, and a light source, such as a lamp used for near vision, was directed from behind the patient to the examiner's hand that either was stationary or was moved at one motion per second in a horizontal or vertical direction at a distance of 60 cm from the eye. The patient was asked to identify whether the examiner's hand was still, moving sideways, or moving up and down. The presentation was repeated five times, and hand-motion visual acuity was considered present if the patient was able to identify the examiner's action on at least four of the presentations. If the examiner was not convinced that hand motions could be detected, LP was tested at 0.9 m with an indirect ophthalmoscope set at maximum intensity.'
Differential Diagnosis of Acute Endophthalmitis
- Occult retention of lens cortex or nucleus
- Noninfectious hypopyon uveitis (Behçet or rifabutin)
- Infectious posterior uveitis (syphilitic, tuberculous, toxoplasmosis, toxocariasis in children)
- Noninfectious posterior uveitis (Vogt-Koyanagi-Harada disease, collagen vascular disorders, systemic vasculitides, idiopathic)
- Inflammatory reaction to intravitreal drug
- Blebitis
- Acute retinal necrosis
- Keratitis
- Toxic anterior segment syndrome (TASS)
- rapid onset (within 12-24 hours, often with limbus-to-limbus corneal edema)
- increased intraocular pressure
- absent vitreous inflammation
- iris damage
- response to steroid
- Intraocular lymphoma (masquerade syndrome)
Testing
Ultrasound Evaluation
- B-scan ultrasonography should be performed if significant vitritis or media opacity prevents an adequate view of the fundus.
- Findings consistent with endophthalmitis:
- dispersed vitreous opacities with vitritis
- chorioretinal thickening
- Assess for the presence of retinal or choroidal detachment, retained lens material, and retained foreign bodies.
- Retinal or choroidal detachment are poor prognostic factors.
Microbiological Characteristics
- Bacteria are the most common infecting agents.
- Causative organisms typically represent bacteria from the patient’s own periocular flora introduced during the procedure or gaining access later, usually in the early postprocedure period.
- In cases of intravitreal injection-associated endophthalmitis, additional causes include oral flora (from the treating physician or patient; it appears to occur at a higher rate compared to postoperative endophthalmitis) or drug contamination. Therefore, it is important for providers and patients to not speak during injections and consider wearing a mask or using a drape.
- In the EVS, 94.2% of culture-confirmed cases involved gram-positive bacteria.
- Seventy percent (70%) were gram-positive, coagulase-negative staphylococci (Staphylococcus epidermidis).
Obtaining Specimens for Microbiological Studies
- In the EVS, vitreous samples yielded positive cultures more often than aqueous samples.
- Aqueous samples yielded positive culture only in a small minority (4.2%) of eyes.
- Given this, vitreous sampling is most often the first choice. Some surgeons obtain both vitreous and aqueous, while others obtain an aqueous sample only if the vitreous tap is dry. A minority obtain only an aqueous sample.
- The eye should be prepped with 5% povidone-iodine. Lashes may be draped. An eyelid speculum may be used.
- Subconjunctival or periocular lidocaine anesthesia is usually required, as most patients are very uncomfortable with the procedure to begin with.
Vitreous Specimen
- Needle aspiration
- A 22G to 27G needle attached to a syringe is inserted into the vitreous cavity through the pars plana (pull the plunger back first to break the vacuum before inserting into the eye).
- Slowly aspirate. If no fluid vitreous can be obtained with a needle tap, an anterior chamber (AC) tap or vitreous biopsy must be performed instead to avoid aspirating formed vitreous.
- Vitrectomy
- After cannula insertion or conjunctival incision and sclerotomy (if 20 g), a vitrectomy probe attached to a syringe is inserted into the vitreous cavity.
- Approximately 0.2-0.5 mL of vitreous is removed from the anterior vitreous cavity by using the automated cutting mechanism of the probe and slow, manual aspiration. Often, the infusion may be kept clamped in order to obtain an undiluted sample, followed by opening of the infusion to also collect a diluted sample.
Anterior Chamber Specimen
- A 27G to 30G needle is inserted near the limbus into the AC.
- 0.05 cc to 0.1 cc of fluid is withdrawn.
- Care should be taken to avoid the lens if the patient is phakic by keeping the needle over the iris. The AC should not be flattened. Do not aspirate the hypopyon, as it is congealed and may not go through the needle.
Specimen Handling
- Culture inoculation by the surgeon or laboratory within minutes of obtaining specimens is ideal to maximize recovery of organisms.
- Inject the specimen deeply into liquid media to ensure the sample is within the media.
- Gram stain can help to determine the presence of infectious organisms and white cells acutely. Cultures (aerobic, anaerobic, and fungal) are useful for specific identification of organisms and sensitivity.
- Antibiotic sensitivities (although in the EVS, vancomycin and amikacin empirically covered 99.4% of all infecting organisms) should be identified.
- More recently, polymerase chain reaction (PCR) testing is being used more frequently to identify suspected pathogens in the sample. PCR testing requires the physician to specify the suspected organisms when requesting the test.
Treatment
Pars Plana Vitrectomy (PPV)
- Advantages: reduction of infecting organisms, toxins, inflammatory materials, and opacities. Allows a more robust collection of samples for culture.
- Disadvantages: Need for sophisticated equipment and operating room time/staff. Visualization may be poor due to corneal edema/opacification, posterior synechiae, and dense vitreous debris.
- Vitrectomized eyes may have more rapid clearance of intravitreal antibiotics.
- In inflamed, aphakic, vitrectomized rabbits, intravitreal vancomycin and ceftazidime half-lives are 7.5 and 5.1 hours, respectively, compared to 31.4 and 10.1 hours, respectively, in inflamed, phakic non-vitrectomized rabbits.
PPV – obtaining sample
- Standard 3-port setup
- A vitreous sample of 0.2-0.5 mL is first obtained without infusion using gentle manual aspiration into a syringe; this yields an undiluted vitreous specimen.
- Infusion is then initiated and core vitrectomy performed.
- The AC is cleared for visualization as necessary.
- In most cases, the IOL need not be removed initially.
- Aspiration of debris within the capsular bag, if it can be performed safely, is also helpful.
- In the EVS, attempts were made to clear 50% or more of the vitreous.
- Also in the EVS, no attempt was made to induce a posterior vitreous detachment if none was previously present. Since the visualization is often poor, creation of an iatrogenic break may go undetected and lead to postoperative retinal detachment and proliferative vitreoretinopathy.
Immediate PPV vs. Tap & Inject
- In the EVS, patients who presented with LP-only visual acuity had a significant, threefold improved chance of obtaining 20/40 vision after immediate vitrectomy (33%), compared to tap and inject (11%).
- 56% of eyes achieved 20/100 or better after immediate PPV compared to only 30% after tap.
- HM or better vision demonstrated no significant difference between the treatment groups in final visual acuity (consider earlier PPV for diabetics).
EVS Limitations
- Results may not apply to endophthalmitis after other ocular procedures (EVS only studied post cataract and IOL surgery).
- For example, post-intravitreal injection, bleb-related endophthalmitis or chronic postoperative endophthalmitis were not studied in the EVS.
- Patients with no light perception (NLP) or significant opacification of AC obscuring iris tissue were excluded from the study. Therefore, severe infections or virulent organisms were excluded.
- It was unknown whether PPV is superior in these circumstances.
Intravitreal Antibiotics
Current recommendations for empirical therapy (bacterial):
- Vancomycin 1.0 mg/0.1 mL
- Ceftazidime 2.25 mg/0.1 mL or Amikacin 400 μg/0.1 mL for penicillin (PCN)-allergic patients (although usually can still inject Ceftazidime since there is low cross-reactivity between cephalosporins and PCN; keep in mind that most PCN "allergies" may not really be an allergic reaction; also low systemic absorption from ocular injection)
- 4th generation fluoroquinolones – intravitreal moxifloxacin has been tried in some studies.
- Fungal: Amphotericin B 5-10 μg/0.1 mL or voriconazole 50-100 μg/0.1 mL
Complications of Intravitreal Antibiotics
- Corneal opacification and retinal toxicity
- Retinal toxicity with aminoglycosides, most notably gentamicin (even as low as 0.1mg) may lead to macular infarction
- Less common with amikacin and tobramycin
- Amphotericin B can also cause retinal toxicity
- Hemorrhagic occlusive retinal vasculitis (HORV): rare but serious, associated with vancomycin injection[8][9]
Systemic Antibiotics
- Usually too slow to enter the eye in adequate concentrations so not sufficient as monotherapy. Some surgeons use adjunctive oral antibiotics (i.e., oral moxifloxacin) in addition to intravitreals
- In the EVS, there was no difference in VA or media clarity with or without intravenous antibiotics (amikacin plus ceftazidime or amikacin plus ciprofloxacin) when given in addition to intravitreal antibiotics
- However, newer 4th generation fluoroquinolones with better ocular penetration were not available at the time the EVS was conducted. Systemic ceftazidime, cefazolin, ciprofloxacin, gatifloxacin3 and moxifloxacin have been shown to penetrate the blood ocular barrier efficiently.
Corticosteroids
- Goal is to modulate the host inflammatory response to the infection and minimize ocular damage from this response. Avoid when fungal infection is suspected.
- Intravitreal corticosteroids (dexamethasone) is controversial
- 2 retrospective nonrandomized studies showed 20/400 or better vision was achieved more often in patients who received 400 μg adjuvant intravitreal dexamethasone
- Randomized, prospective trial of 63 patients showed in no difference in visual acuity at 12 weeks
- Another retrospective, nonrandomized trial of 57 patients showed worse visual acuity
Reinjection
A minority of patients will require further treatment, mainly for worsening ocular infection. It typically requires more than 24 hours to observe an improvement in the clinical appearance after initial treatment. In many cases, the treated eyes look somewhat worse 1 day after treatment before improving subsequently.
Considerations of reinjection
- Should be considered if the infection fails to stabilize or improve more than 48 hours after the first injection
- Based on consensus view, the EVS protocol recommended reinjection if the infection was worsening at 36-60 hours after initial injection
- Often, 36 hours after treatment, culture results are available
- Decision to reinject antibiotics should not be taken lightly, since repeat injection may increase risk of retinal toxicity
- 100% retinal toxicity rates with 3 intravitreal doses of 1mg vanco combined with 400 μg amikacin or 200 μg gentamicin in rabbit eyes
- Patients with no culture growth, equivocal growth, or coagulase-negative Staphylococci had a 5% rate of additional procedures, compared to 30% in patients with cultures that grew gram negative or other gram positive organisms
Outcomes and Risk Factors for Poor Outcomes
- The most common cause of visual loss in the EVS was attributed to macular abnormalities, such as ERM, macular edema, pigmentary degeneration, and ischemia
- Strongest predictor of poor visual outcome was light perception only vision
- Other risk factors: older age, diabetes mellitus, corneal infiltrate or ring ulcer, compromised posterior capsule, low or high intraocular pressure, afferent pupillary defect, rubeosis, and absent red reflex
Virulence
Eyes with coagulase-negative Staphylococci were most likely to obtain 20/100 final visual acuity or better (84%). More virulent organisms, such as Staphylococcus aureus, Streptococcal species, and gram negative (Pseudomonas) organisms were associated with worse visual outcomes
Chronic Postoperative Endophthalmitis (> 6 weeks)
History
- Usually manifest several weeks or months after surgery
- Less common than acute variety
- Organisms isolated are less virulent bacteria and fungi
- 63% Propionibacterium acnes, 16% Staphylococcus epidermidis, 16% Candida parapsilosis
Clinical features
- Pain or discomfort may not be present
- Inflammation can be initially steroid responsive but recurrent after steroid taper
- Fungal infections demonstrate paradoxical worsening with steroids
- Frank hypopyon is often absent
- Granulomatous uveitis with large precipitates on cornea or IOL
- White intracapsular plaque with P. acnes
- Stringy white infiltrates and “fluff balls” or “pearls-on-a-string” near the capsular remnant are characteristic but not pathognomonic for fungal infection
Treatment for Chronic Cases
- Obtain AC and vitreous samples
- Identification of infectious organism is key in management of these infections
- Intravitreal injection
- However, vancomycin is often inadequate for P. acnes
- Often need PPV and removal of capsule and/or IOL
Filtering Bleb-Associated Endophthalmitis
- Occurs as a result of pathogenic organisms gaining entry to intraocular tissue through the conjunctival filtering bleb
- Mean time between surgery and endophthalmitis is 19.1 months (range 3-9 years)
- 0.2 – 9.6% of glaucoma filtering procedures
- Increased incidence with use of antifibrotic agents
- Thin, cystic, avascular conjunctiva
- Blebitis – if no uveitis or vitritis
Post-Trabeculectomy Endophthalmitis Treatment
- If only blebitis is present, provide conservative treatment with systemic and topical fortified antibiotic therapy.
- If the vitreous is clear, examine these patients frequently, treating for endophthalmitis if vitritis or acute hypopyon develops.
- Needle aspiration of the bleb is not advised.
- Studies suggest possible benefit from PPV.
Endophthalmitis after Intravitreal Injection
- Intravitreal injections have increased in volume worldwide, leading to increased prevalence of endophthalmitis
- Coagulase-negative Staphylococci is the most common organism, similar to acute postop endophthalmitis.
- In many cases, no infectious agent is identified.
- Triamcinolone acetonide crystals can migrate into the AC and mimic hypopyon.
- Up to 1.4% after intravitreal triamcinolone injections
- 0.008% - 0.092% after anti-vascular endothelial growth factor injections
- Intravitreal triamcinolone may play a role in endophthalmitis potentiation.
Intraocular Inflammation after Triamcinolone Injection or Intravitreal Anti-VEGF Injection
- Triamcinolone crystals in AC or vitreous cavity can be difficult to distinguish from inflammatory reaction.
- Use gravity-induced shifting of layered material and absence of AC flare or fibrin as a sign of a noninflammatory cause.
- Triamcinolone crystals may shift from the inferior anterior chamber angle when the patient is placed on his or her side, a phenomenon that does not occur with sticky, fibrin-laden material in a true inflammatory hypopyon.
- Intraocular inflammation from triamcinolone often presents with a white eye and little pain or discomfort.
- Reported cases of noninfectious intraocular inflammation after anti-VEGF injections[10]
- Cases can be difficult to distinguish from true endophthalmitis. Therefore, there is a low threshold for immediate treatment with tap and inject.
Management options
- Close observation, topical steroids alone, oral fluoroquinolones alone
Traumatic Endophthalmitis
Etiology
- Approximately 25% of endophthalmitis cases are traumatic.
- After open-globe injury, the chance of developing endophthalmitis is approximately 7%.
- Injuries including intraocular foreign body (IOFB) have higher rates.
Risk Factors
- Other risk factors: dirty wound, lens capsule rupture, age >50 years, delayed presentation of more than 24 hours after injury
- Bacillus and Staphylococcus species are common in penetrating trauma with IOFB.
Workup
- Primary repair and removal of IOFB as soon as possible
- Exclude the possibility of occult, retained IOFB.
- CT scan with thin 1 mm cuts.
- Obtain cultures.
- Intravitreal vancomycin and ceftazidime
- Some advocate gentamicin + clindamycin for synergistic effect against Bacillus and Staphylococcus.
Treatment considerations
- Application of EVS may not be appropriate, due to different organisms. In severe cases, vitrectomy should be strongly considered.
- Some authors advocate therapeutic vitrectomy in all cases of traumatic endophthalmitis.
- Others suggest performing vitrectomy only if patients do not respond after 48 hours or rapidly deteriorate in the first 24 hours following intravitreal antibiotics.
- Post-traumatic endophthalmitis has a worse visual prognosis than other categories.
Prophylaxis after Penetrating Trauma
- Clinical evidence not established.
- Some authors advocate intravitreal antibiotic administration in all cases of penetrating trauma, especially when associated with an IOFB.
- Others recommend administration in the presence of risk factors (2 of 3: dirty wound, lens breach, or delay in closure over 24 hours).
- Systemic antibiotics: intravenous cefazolin (1 g every 6 hours for 48-72 hours), followed by an oral agent such as gatifloxacin or moxifloxacin for 7 days.
Endogenous Bacterial Endophthalmitis
For endogenous fungal endophthalmitis, please refer to the article on Fungal Endophthalmitis.
Etiology
- Originates from sources within the body.
- Infectious extraocular focus can be found in 90% of the cases, such as tooth abscess, pneumonia, endocarditis, liver abscess, urinary tract infection, and bacterial meningitis.
- Bacterial causes are responsible for half of endogenous endophthalmitis, while fungal origins are responsible for the other half.
- Gram-positive organisms:
- Streptococcus species: primary infection is often endocarditis, which accounts for 40% of endogenous bacterial endophthalmitis in North America.
- Staphylococcus aureus often comes from a skin infection.
- Bacillus species are mostly seen in IV drugs users.
- Nocardia species are mostly seen in immunocompromised patients.
- Gram-negative organisms:
- Escherichia coli from urinary tract infection is the second leading cause and is responsible for about a third of cases in North America.
- Neisseria meningitidis and Haemophilus influenzae can both be associated with meningitis.
- Klebsiella species are often associated with liver abscess and are the leading cause of endogenous bacterial endophthalmitis in Asian populations.
Other Risk Factors [11]
- Impaired immune status: HIV/AIDS, chronic disease (eg, diabetes mellitus, systemic malignancy, sickle cell anemia, systemic lupus erythematous), immunosuppressive treatment. Also, neonatal immune systems are vulnerable to endogenous fungal endophthalmitis.
- Recent invasive medical procedures: extensive gastrointestinal surgery, endoscopy, dental procedures
- Intravenous access lines and catheters
Diagnosis
- Anterior chamber paracentesis with vitrectomy: vitreous and aqueous cultures (eg, aerobic, anaerobic, fungal) with stains (eg. gram, giemsa)
- Identification of the causative organism is made by systemic culture of blood, urine, and/or cerebrospinal fluid in 75%-80% of cases.
Treatment[12]
- In contrast to postoperative endophthalmitis, systemic antibiotics are central to the treatment of endogenous endophthalmitis.
- Intravitreal antibiotics administered rapidly
- Initial empiric treatment with vancomycin 1 mg/0.1 mL with ceftazidime 2.25 mg/0.1 mL or amphotericin B 5-10 μg/0.1 mL in addition to systemic therapy
- Focal chorioretinitis and associated mild vitritis can respond to systemic therapy alone.
- A late vitrectomy is preferred to additional antibiotics when the disease progresses under antibiotic therapy.
- Additional antifungal therapy if there is doubt on the etiology
- Corticosteroids are controversial, but they may help to reduce enucleation and evisceration.
Conclusions
- Early diagnosis and treatment with antimicrobial therapy are critical.
- Usually, diagnosis must be made on clinical grounds.
- Treat with empirical therapy, usually vancomycin + ceftazidime if bacterial.
- EVS showed that PPV was of benefit in patients with endophthalmitis following cataract surgery or secondary IOL placement who presented with LP-only visual acuity.
- Although the role of vitrectomy in other forms of endophthalmitis is less clear, it remains an accepted adjunct to intravitreal antibiotics for treatment of moderate to severe cases.
References
- ↑ Lemley et al. Endophthalmitis, a review of current evaluation and management. Retina. 2007; 27:662-680.
- ↑ Das T, Sharma S, Hyderabad Endophthalmitis Research Group. Current management strategies of acute post-operative endophthalmitis. Semin Ophthalmol. 2003;18(3):109-115.
- ↑ Endophthalmitis Vitrectomy Study Group. Results of the Endophthalmitis Vitrectomy Study. A randomized trial of immediate vitrectomy and of intravenous antibiotics for the treatment of postoperative bacterial endophthalmitis. Arch Ophthalmol. 1995;113:1479–1496.
- ↑ Chen G, Tzekov R, Li W, Jiang F, Mao S, Tong Y. Incidence of endophthalmitis after vitrectomy: a systematic review and meta-analysis. Retina. 2019;39:844-852.
- ↑ Baudin F, Benzenine E, Mariet AS, Ben Ghezala I, Bron AM, Daien V, Korobelnik JF, Quantin C, Creuzot-Garcher C. Epidemiology of Acute Endophthalmitis after Intraocular Procedures: A National Database Study. Ophthalmol Retina. 2022 Jun;6(6):442-449. doi: 10.1016/j.oret.2022.01.022. Epub 2022 Feb 5. PMID: 35134544.
- ↑ Samuelson AG, Patel SN, Kommareddy K, Momenaei B, Yu-Chuan Kang E, Chaudhary V, Hsu J, Dunn JP, Vander JF, Garg SJ. Outcomes of Postcataract Surgery Endophthalmitis Managed Without Microbial Cultures. Ophthalmol Retina. 2024 Jul 20:S2468-6530(24)00338-5. doi: 10.1016/j.oret.2024.07.015. Epub ahead of print. PMID: 39038541.
- ↑ Patel SN, Cai LZ, Mahmoudzadeh R, Salabati M, Magan T, Obeid A, Soares RR, Hinkle JW, Hsu J, Dunn JP, Vander JF, Garg SJ. Endophthalmitis After Intravitreal Anti-Vascular Endothelial Factor Injections: Outcomes of Eyes Managed Without Microbiologic Cultures. Am J Ophthalmol. 2022 Mar;235:1-6. doi: 10.1016/j.ajo.2021.08.012. Epub 2021 Aug 27. PMID: 34461083.
- ↑ Andreanos K, Petrou P, Kymionis G, Papaconstantinou D, Georgalas I. Early anti-VEGF treatment for hemorrhagic occlusive retinal vasculitis as a complication of cataract surgery. BMC Ophthalmol. 2017 Dec 6;17(1):238. doi:10.1186/s12886-017-0632-y. PubMed PMID:29212481; PubMed Central PMCID:PMC5719895.
- ↑ Witkin AJ, Chang DF, Jumper JM, Charles S, Eliott D, Hoffman RS, Mamalis N, Miller KM, Wykoff CC. Vancomycin-Associated Hemorrhagic Occlusive Retinal Vasculitis: Clinical Characteristics of 36 Eyes. Ophthalmology. 2017 May;124(5):583-595. doi: 10.1016/j.ophtha.2016.11.042. Epub 2017 Jan 19. PubMed PMID: 28110950.
- ↑ Grewal DS, Schwartz T, Fekrat S. Sequential Sterile Intraocular Inflammation Associated With Consecutive Intravitreal Injections of Aflibercept and Ranibizumab. Ophthalmic Surg Lasers Imaging Retina. 2017 May 1;48(5):428-431. doi: 10.3928/23258160-20170428-10. PubMed PMID: 28499055.
- ↑ Jackson TL, Paraskevopoulos T, Georgalas I. Systematic review of 342 cases of endogenous bacterial endophthalmitis. Surv Ophthalmol. 2014;59(6):627–635.
- ↑ Spelta S, Di Zazzo A, Antonini M, Bonini S, Coassin M. Does Endogenous Endophthalmitis Need a More Aggressive Treatment? Ocul Immunol Inflamm. 2021 Jul 4;29(5):937–43.