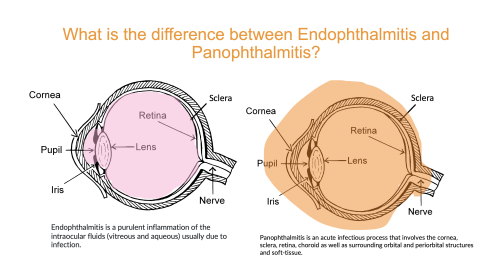
Panophthalmitis
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Summary
A comprehensive review of Panophthalmitis.

ICD-10 Code
| Diagnosis | ICD-10 Code |
| Panophthalmitis (acute), unspecified eye | H44.019 |
Disease Entity
Panophthalmitis is a rapidly progressing suppurative process encompassing all structures of the globe (choroid, retina, vitreous fluid, aqueous fluid, cornea, sclera, and conjunctiva) with extension into the orbital structures. Delayed or inadequate treatment can have devastating implications such as severe or permanent vision loss, removal of the eye (evisceration, enucleation or exenteration), cavernous sinus thrombosis, meningitis, encephalitis, sepsis, stroke, or death.
Disease
Panophthalmitis is an infectious process caused by a pyogenic organism that encompasses all structures of the globe (choroid, retina, vitreous fluid, aqueous fluid, cornea, lens, and sclera) along with surrounding orbital and periorbital structures. The etymology of the term panophthalmitis is from the latin root words of pan- meaning ‘all’, ophthalmo- meaning ‘of or relating to eyes’, and -itis meaning ‘inflammation.’ Simply, panophthalmitis is an “endophthalmitis” with orbital cellulitis.[1] Panophthalmitis is a devastating complication that can result in permanent vision loss, pthisis bulbi, or surgical removal of the eye (evisceration or enucleation). [2]
This disease process is typically aggressive and presents with orbital signs – extra-ocular movement restriction or complete ophthalmoplegia, pain, vision loss, afferent pupillary defect, and proptosis. Endogenous panophthalmitis can present with signs of systemic infection such as fever and leukocytosis. Depending on the virulence of the involved organism, patients can develop florid panophthalmitis in a span of 12-48 hours.
The source of panophthalmitis can be divided into endogenous versus exogenous infiltration. Exogenous infiltration is most commonly caused by a penetrating globe injury (organic material to eye such as wood, metallic foreign body such as a nail or knife), acute or chronic post-operative development (cataract extraction, glaucoma surgery), filtering bleb-associated infection, intravitreal injections, and corneal ulcers. Endogenous infiltration through hematogenous route with progressive breakdown of the blood-ocular barrier. This can occur from bacteremia from an extra-ocular nidus such as vegetations on heart valves (endocarditis), migration from abscess elsewhere in the body, bacterial or fungal Panuveitis, bacterial or fungal chorioretinitis with or without vitritis.[3] The most frequently implicated endogenous organ sources are lungs, bladder, liver, sinuses, skin, CNS (meningitis, brain abscess), and heart.
The treatment of panophthalmitis is multi-disciplinary and is predominated by patient presentation and history (medical, surgical, and social). Ophthalmology and Medicine services are involved in coordinating infectious disease work-up (blood cultures, vitreous or aqueous cultures, Trans-Thoracic Echocardiogram), and treatment with intravitreal and intravenous broad-spectrum antibiotics. Orbital imaging with thin cuts (1-3 mm) with and without contrast helps further guide the extent of orbital and periorbital involvement. Depending on culture speciation, an Infectious Disease specialist can help guide appropriate medication and extent of therapy. [3] Early intervention is critical to prognosis, and hospital admission with vigilant daily follow-up is necessary.
In extensive orbital and peri-orbital involvement with limited-to-no improvement with medical management, surgical evisceration or enucleation may be necessary to prevent further devastating complications.
Epidemiology
There is limited data available on the epidemiology of panophthalmitis, however there are certain critical social components that often correspond with the organism(s) involved. Increased incidence of panophthalmitis is found amongst the immunocompromised and intravenous drug abuse populations. Immunocompromised people remain easily susceptible to infections that the body has poor defense against. Intravenous drug users are at risk as they self-inoculate virulent organisms into their bloodstream and may often have an immunocompromised component due to additional medical pathology such as HIV. [4] There is no racial or gender predilection for panophthalmitis.[5]
In a 2018 study, the leading cause of endophthalmitis leading to panophthalmitis was post-traumatic injury to the orbit (39.9%), followed by post microbial keratitis (27.7%), endogenous source (21.21%), post cataract surgery (9.09%) and post penetrating keratoplasty (3.03%). [2]
Anatomy
Panophthalmitis can occur endogenously through orbital inoculation through hematogenous spread or exogenously through penetrating inoculation. Typically, endogenous endophthalmitis precedes panophthalmitis.
Blood supply of the eye
In endogenous endophthalmitis, a nidus will spread a virulent organism through the bloodstream. The most common route is through the posterior segment vasculature.
- The carotid artery bifurcates into the internal and external carotid artery.
- The first branch off the internal carotid artery is the ophthalmic artery, which is distal to the cavernous sinus.
- As the ophthalmic artery courses through the orbit, it gives off several branches that supply blood to the globe, extraocular muscles, orbit, and periorbital region. The most significant branch of the ophthalmic artery in the development of endogenous endophthalmitis is the short posterior ciliary artery.
- Short posterior ciliary artery
- The predominant source of infiltration of endogenous organisms into the eye
- There are approximately 20 short posterior ciliary arteries.
- Course: follows the path of the optic nerve to the posterior sclera and enters near the entrance of the optic nerve.
- Supplies the choroid (up to equator), ciliary processes, and is part of the anastomotic ring “circle of Zinn” to supply the optic disc.
- Long posterior ciliary artery
- There are 2 vessels in each eye, one nasally at 9:00 and one temporally at 3:00
- Combines with the anterior ciliary artery to form the major arterial circle of the iris.
- Supplies the iris, ciliary body, and choroid.
- Anterior ciliary artery
- There are 7 small arteries in each eye.
- Supplies the rectus muscles, conjunctiva, and sclera before joining the long posterior ciliary artery to form the supply the iris.
Pathophysiology
The pathophysiology of panophthalmitis is dependent on endogenous versus exogenous inoculation.
Endogenous pathophysiology
The leading pathophysiology for endogenous panophthalmitis is an organism released from a nidus in the body, that travels through the blood to gain entrance into the globe primarily through the short posterior ciliary arteries. This is the initial break in the blood-ocular barrier. As the infection proliferates, inflammation follows as an immune response, enters the choroid, and involves adjacent tissues such as the retina and vitreous with ultimate spill over into the anterior chamber. As the infection propagates, it involves all tunics of the globe and the surrounding tissue to become panophthalmitis.
Exogenous pathophysiology
Post-traumatic and post-operative panophthalmitis develops from the inoculation of organism into the eye through a physical break in the blood-ocular barrier, such as surgical incision, artificial implant, or globe laceration or perforation. Toxins from the intruding organism cause an inflammatory reaction that propagates from the vitreous fluid to all layers of the eye to become panophthalmitis. [6]
Notably, Bacillus cereus is a generally innocuous organism that most commonly gains access into the body through intravenous drug use, allowing bloodstream infiltration.
Distinction between Endophthalmitis vs. Panophthalmitis
Endophthalmitis is an inflammation involving vitreous humor that can have extension into the aqueous humor of the eye.[1] As the retina lines the vitreous cavity, purulent inflammation of the vitreous humor can extend into the retina and perpetuate inflammation in an outward fashion to involve adjacent layers.
Panophthalmitis is an extension to the surrounding orbital structures.[7] Patients with panophthalmitis present with eyelid edema and erythema, proptosis, conjunctival injection, and chemosis, often mimicking an orbital cellulitis presentation. [8]
Risk Factors
Predominant risk factors for development of panophthalmitis include conditions that lead to patients being more vulnerable to an orbital infection. This includes:
- Immunosuppressed (patients with organ transplant, or autoimmune diseases)
- Immunocompromised patients (Chronic disease such as diabetes, sickle cell disease, cancer patients, neonates, HIV/AIDS)
- Septic/Bacteremic Neonates
- Highest risk in those with candidemia, bacteremia, ROP and low birthweight.[9]
- History of penetrating injuries
- Post-operative surgery from intra-ocular surgery (cataract surgery, pars plana vitrectomy, filtering bleb)
- Intravenous drug abuse
- Indwelling catheters
- Intravenous Access
- Recent invasive medical procedure (Bowel surgery, Endoscopic surgery, Dental work)
- Endophthalmitis
- Bacteremia
- Endocarditis
- Corneal ulcer
- Chronic intravitreal injection patients (diabetic retinopathy patient, exudative macular degeneration patient)
- Panuveitis
- Advanced Age
Most Common Causative Organisms
Common Causative Organisms of Panophthalmitis: Bacteria
Bacillus Cereus
Bacillus Cereus is a gram positive, spore-forming, aerobic organism that is found throughout the environment in places such as the soil, water, and dust.[7] It is often found in cases of intravenous drug abuse or secondary to surgery or trauma. [10]
The pathogenicity of B. Cereus is hypothesized to be from lecithinase - a phospholipase toxin. Lecithinase is a virulent factor that targets the membrane of cells causing myonecrosis and hemolysis. [11]
Risk Factors
- Penetrating injuries
- Post-operative period
- Intravenous drug abuse
- Endophthalmitis
- Immunosuppression
Early intervention typically involves antibiotics and surgical intervention, with evisceration or enucleation reserved for end-stage florid disease.
Klebsiella pneumoniae
Klebsiella pneumoniae panophthalmitis is rare. It is associated with hepatobiliary, urinary tract, and nosocomial infections that leads to panophthalmitis through hematogenous spread. [12]
Risk Factors
- Southeast Asian population due to a genetic predisposition
- Diabetes
- Alcoholism
- Liver disease
- Systemic immunocompromised state
- History of recurrent antibiotic use
Treatment plans often include early systemic antibiotics in combination with intravitreal antibiotics. If the patient does not respond to this treatment method, a trans pars plana vitrectomy (TPPV) should be performed to reduce the infectious burden and remove toxic debris from the vitreous cavity. [12]
Mycobacterium Tuberculosis
Mycobacterium tuberculosis – most known to causes systemic illness from Tuberculosis – is also a rare cause of panophthalmitis. It is a weakly gram-positive, acid-fast, rod-shape, obligate anaerobe often residing and proliferating in the lungs. Panophthalmitis from M. tuberculosis is usually from a secondary infection from latent systemic disease with re-activation. It inoculates ocular tissue through the uveal tract and can result in granulomatous anterior uveitis, disseminated choroiditis with vitritis prior to endophthalmitis and subsequently, panophthalmitis.[13] It is also possible to have direct invasion into the eye from traumatic injury on the conjunctiva or cornea. [14]
Risk Factors
- Children
- Immunocompromised
- History of intravenous drug abuse
- History of Incarceration
- History of Homelessness
- Immigration from endemic region
It is important to consider tuberculosis as a source of infection, particularly in endemic regions, homeless patients, or patients with history of incarceration. If there is clinical suspicion, it is important to complete CXR or CT Chest and order a Quantiferon Gold Test (PPA).
Medical management has two phases: the initial phase includes RIPE therapy (Rifampin, Isoniazid, Pyrazinamide, Ethambutol, and Vitamin B6), and lasts for two months; the second phase is for four months with only Rifampin and Isoniazid. [15]
Clostridium perfringens
Clostridium perfringens is a species of gram-positive, anaerobic, bacillus bacteria that can infect the eye which ocular tissue is penetrated with soil and plant contaminated materials. Similar to B. cereus, C. perfringens also produces lecithinase that impacts cellular membrane. Infection by C. perfringens can be very aggressive with florid infection peak within 48 hours from the onset of injury/inoculation.
Risk Factors
- Immunocompromised patients
- Recent ocular injury with contamination from organic material
- Gastrointestinal perforation
Diagnostic microbial testing includes taking specimen from ocular tissue (corneal ulcer, aqueous humor, vitreous humor) and analyzing with gram stain, Nagler’s reaction, and reverse CAMP test.
Medical management of C. perfringens panophthalmitis requires a multidisciplinary team including infectious disease and primary care, and immediate initiation of intravenous antibiotics. [16]
Methicillin-Resistant Staphylococcus Aureus (MRSA)
Methicillin-resistant Staphylococcus aureus (MRSA) is an anaerobic, gram-positive, cocci bacteria that is predominant in hospital-acquired infections. MRSA induced panophthalmitis is a common post-surgical and can be very aggressive.
Risk Factors
- Prolonged hospital residence
- Recent surgery
- Skin trauma
- Medical device placement in the body (catheter, pacemaker, etc)
Studies have shown that patients with cataract surgery can be prone to MRSA infections, usually occurring within the post-operative 6-week period. A detailed history is also important because MRSA-associated panophthalmitis is more common in patients with a chronic disease state. Ophthalmologists can administer intravitreal injections of vancomycin with either ceftazidime or amikacin as a treatment plan, along with intravenous antibiotics. [17]
Common Causative Organisms of Panophthalmitis: Fungi
Aspergillus
Aspergillus associated panophthalmitis is associated with ocular trauma, particularly in immunocompromised individuals. [14] Despite typical fungal infections having a long latent period, ocular trauma destructs the integrity of the globe and high concentrations of Aspergillus can be inoculated and cause the patient to present within a week of exposure.
Treating panophthalmitis caused from Aspergillus infection can be done with antifungal medication such as Amphotericin B, Miconazole, and Ketoconazole. It is important for ophthalmologists to determine which organism is infecting their patients due to the different Minimal Inhibitory Concentrations (MIC).
Rhizopus
A Rhizopus infection progressing into a panophthalmitis is rare but has high morbidity and mortality. Host risk factors include diabetes mellitus, neutropenia, sustained immunosuppressive therapy, chronic prednisone use, iron chelation therapy, broad-spectrum antibiotic use, severe malnutrition, and primary breakdown in the integrity of the cutaneous barrier such as trauma, surgical wounds, needle sticks, or burns. Rhizopus infection is difficult to culture and a high clinical suspicion with evaluation of necrotic oral or nasal tissue is key to early diagnosis, debridement to reduce disease burden, and systemic treatment.
The most common antifungal drugs effective against Rhizopus are Amphotericin B, Posaconazole, or Isavuconazole, which are all administered intravenously. Oral drugs like Posaconazole and Isavuconazole could also be given. [18] Apart from surgical intervention, studies have shown that Amphotericin B is the most effective against Rhizopus.
Common Causative Organisms of Panophthalmitis: Virus
Herpetic Panophthalmitis (Herpes Simplex Virus and Varicella Zoster Virus)
Herpetic panophthalmitis is incredibly rare and difficult to diagnosis. Unique findings for HSV related panophthalmitis is granulomatous intraocular inflammation with elevated intraocular pressure. [19] Confirmation of HSV is typically done by a vitreous sample which can be analyzed by polymerase chain reaction. Treatment of Herpetic panophthalmitis is through intravitreal and intravenous antivirals along with steroids.
Dengue Virus
Dengue fever is caused by a Flavivirus with 4 known serotypes. Dengue panophthalmitis is most associated with serotype antibodies to NS1. The virus is spread by Aedes mosquitoes most commonly found in the Indian subcontinent therefore if there is any recent travel to an endemic region, there should be increased clinical suspicion.
Though treatment data is limited, early vitrectomy seems to have potential for vision and globe salvage in patients presenting in the early stages of the disease. [20]
Diagnosis
Patients who present with panophthalmitis will show signs of endophthalmitis (hypopyon, corneal edema, conjunctival and scleral injection, ocular pain) along with orbital cellulitis with orbital signs (vision loss, afferent pupillary defect, elevated intraocular pressure, extraocular movement pain, and restriction). [11]
History
Patient medical, surgical, and social histories are an important crux in the diagnosis of panophthalmitis. A thorough medical and surgical history can reveal predispositions to panophthalmitis such as recent intra-ocular surgery, GI surgery, organ transplant, autoimmune diseases, infections (pneumonia, diarrhea, bacteremia, abscess, infected joint, endocarditis, etc), or recent trauma. Social history can direct attention to more insidious or innocuous organisms related to intravenous drug abuse, homelessness, incarceration, or travel exposure to endemic regions.
Ophthalmic Exam
When an infectious etiology is suspected in the orbit, dilated fundus exam is deferred as it would interfere with identifying a new afferent pupillary defect, a critical orbital sign suggestive of progression. A Bright Scan Ultrasound (B-scan) can be used to visualize the posterior chamber of the eye to assess for vitritis (vitreous inflammation that present as vitreous echoes), exudative retinal detachment, scleritis (scleral thickening with classic “T-sign”), choroidal lesions (dome-shaped lesions), choroidal thickening, sub-tenon’s fluid, lens dislocation, lens edema, and shallow anterior chamber).
Differential Diagnosis
- Endophthalmitis, Endophthalmitis following Cataract Surgery
- Orbital Cellulitis
- Corneal Ulcer
- Orbital Abscess (sub-periosteal, scleral)
- Panuveitis (infectious and non-infectious
- Non-infectious uveitic processes such as Bechet’s or Sarcoidosis can mimic infection.
- Scleritis is most commonly caused by Rheumatoid Arthritis.
- Peripheral Ulcerative Keratitis is commonly associated with Rheumatoid Arthritis and can result in corneal melt. This allows easy access of organism into the eye which can predispose a patient to endophthalmitis.
- Intra-ocular neoplasm (primary or metastatic)– typically more insidious presentation
- Neoplasm must be taken into consideration especially with the pediatric population when complicated with retinal or choroidal detachment & sub-retinal hemorrhage is involved. [21]
- Trauma
Clinical Presentation
Signs
- Decreased Visual Acuity
- Absent red reflex
- Partial or Full Ophthalmoplegia
- Chemosis
- Conjunctivitis
- Scleritis
- Afferent Pupillary Defect (APD)
- Eyelid Edema and Erythema
- Proptosis
- Corneal Edema
- Corneal Opacity
- Hypopyon
- Anterior Uveitis
- Posterior Uveitis
- Fever
- Leukocytosis
- Perforated Cornea or Perforated Globe
- Elevated or Reduced Intraocular Pressure
- Purulent discharge
Symptoms
- Ocular pain
- Pain with Extraocular movement
- Red eye
- Blurred Vision
- Photophobia
- Epiphora
- Headache
- Malaise
- Nausea/Vomiting
Diagnostic Tests
- Complete Blood Count with Differential
- Lactic Acid
- C-reactive protein
- Blood Culture from two different sources
- Urine Culture (if history warrants)
- Sputum Culture and Stain (if history warrants)
- Trans-thoracic Echo (TTE)
- CT Head, Abdomen/Pelvis, Extremities (as physical exam and history warrant)
Vitreous aspiration
Needle aspiration of vitreous fluid (vitreous tap) is typically performed to collect ocular fluid if the retina is attached on B-scan. Diagnostic and therapeutic vitrectomy is more invasive but provides a higher diagnostic yield compared to needle aspirate. If a retinal detachment is suspected, an aqueous tap is an alternate option.
Real-time polymerase chain reaction (RT-PCR) is a developing technique found to have excellent sensitivity and specificity for fungi in aqueous and vitreous samples. This emerging technique allows rapid results, better yield than cultures, and minimal to no fear of contamination. Limitation involves the inability to detect antibiotic sensitivities. In addition to vitreous fluid stain and culture, it is imperative to draw blood cultures from two different sources to identify bacteremia from an endogenous source.
- Aerobic (Blood Agar, Chocolate agar, Thioglycollate broth)
- Anaerobic (Blood Agar, Thioglycollate broth)
- Mycobacteria spp. and Nocardia spp. (Lowenstein-Jensen media, Acid Fast stain)
- Fungal (Sabouraud Dextrose agar)
- Gram Stain
- Giemsa Stain (Parasites, Bacteria, Fungi, Acanthomoeba spp., Chlamydia spp.)
- Viral transport
[Additional information regarding Microbial Stains]
Diagnostic Imaging
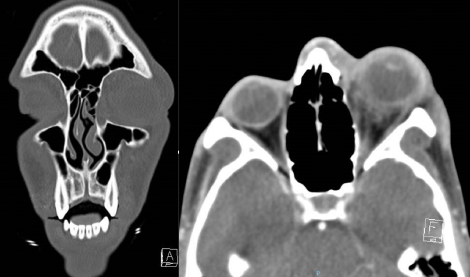
First-line diagnostic imaging is Computed Tomography (CT) of the Orbit and Maxillofacial region in thin cuts. Contrast is necessary if an abscess is suspected. Magnetic Resonance Imaging (MRI) of the Orbit is typically second-line unless there is a high suspicion of a progressive orbital sign without a source. MRI offers high resolution of soft tissue in the orbit and periorbital region. This allows improved visualization of an orbital or scleral abscess, cavernous thrombosis, meningeal involvement, or cranial involvement. [11]
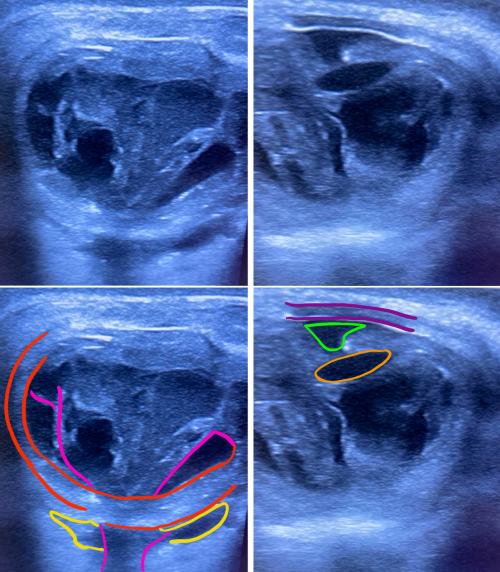
Bright-scan Ultrasonography (B-Scan)
B-Scan is a cost-effective, efficient modality to evaluate posterior segment lesions of the eye. It is particularly useful for assessment of the vitreous, posterior pole, sub-tenon’s space.
Computed Tomography (CT) of the Orbit and Maxillofacial Region
- Most often the primary imaging modality for panophthalmitis.
- Orbital cuts (1mm slices) allow the highest resolution of the orbit and periorbital region.
- Excellent for spatial resolution, trauma, and bony tumors.
- Contrast enhances visualization of high vascular lesions or lesions with abnormal vascular permeability.
- No contrast is adequate to assess for orbital cellulitis components for fat stranding.
- Contrast is recommended to assess for abscess (subperiosteal).
Magnetic Resonance Imaging (MRI) of the Orbit
- Most often the secondary imaging modality for panophthalmitis.
- Excellent resolution for soft tissue such as irregular margins in retinal detachment, globe deformities, scleral abscess, orbital apex, periorbital structures, and the cavernous sinus.
- Contrast enhances visualization of an abscess.
Optical Coherence Tomography (OCT)
- Noninvasive, high-resolution optical imaging
- Produces a real time cross-sectional image of the macula or posterior pole with layers of the chorioretinal interface. This allows visualization of any posterior pole lesions and inter- or sub-retinal fluid.

Management
Medical Management
Medical management is focused on exhausting all resources in order to preserve the eye. Treatment of panophthalmitis begins with the attempt to reduce infectious burden through intravitreal and intravenous antibiotics. While it is important to identify the underlying source of infection, the severity of panophthalmitis requires empiric treatment until cultured results are obtained and/or a source is identified.
Bacterial
Empiric treatment of panophthalmitis includes intravitreal and periocular injections of vancomycin and ceftazidime.[3]
- Vancomycin: most effective against gram positive organisms.
- Administer 0.1 ml of 1 mg/0.1ml composition.
- Ceftazidime: most effective against gram negative organisms.
- Administer 0.1 ml of 2 mg/0.1ml composition.
Fungal
- Mild cases (predominantly outside of the blood-retinal barrier) - consider systemic antifungals therapies such as fluconazole.
- Severe case with involvement of the retina or vitreous humor - combined intraocular and systemic antifungal therapies should be initiated.[22]
- Voriconazole (common choice for intravitreal injections)
- Administer 0.1 ml of 50-100 micrograms/0.1mL composition.
- Amphotericin B
- Administer 0.1 ml of a 5 micrograms/0.1mL composition.
Viral
Viral Panophthalmitis is usually treated with combined intravitreal and intravenous anti-viral agents.
- Ganciclovir
- Administer 0.05 ml of 4 mg/0.1 ml composition.
- Foscarnet
- Administer 0.05 ml of 2.4mg/0.1mL composition.
- Intravenous: 40mg every 8-12 intravenously. [23]
- Acyclovir is usually given intravenously and orally in doses of 13 mg/kg/dose divided every 8 hours for 7 days, followed by 800 mg five times daily orally for 3–4 months.[24]
Steroids
While panophthalmitis is initiated from an infectious etiology, there is also a very strong inflammatory cascade involved. If a patient continues to have severe inflammation after starting treatment of the infectious organism and/or there are positive cultures, steroids can be cautiously administered. Options include:
- Intravitreal Dexamethasone
- Administer 0.10 ml of 0.04mg/0.1ml composition.
- Oral steroids taper dosed at 0.5-2.0 mg/kg/day orally for up to 6-8 weeks can help reduce excess inflammation in the eye and help with recovery. [25]
Complications to monitor are systemic side effects and elevated intraocular pressure from a steroid response.
Surgical Management
When all medical options have been exhausted, the only remaining option for intractable panophthalmitis is to remove the eye through surgical management involving evisceration or enucleation. [3]
| Surgical Procedure | CPT Code |
|---|---|
| Evisceration | 65093 |
| Enucleation | 65105 |
| Pars Plana Vitrectomy | 67036 |
Surgical Options
Evisceration
Evisceration is currently the more preferred surgical intervention for panophthalmitis due to short operating time with efficient and significant reduction in disease burden. The intact scleral shell allows for lower risk of orbital implant extrusion, good prosthetic mobility and cosmesis. However, as panophthalmitis is an infection beyond the globe, careful radiological assessment for scleral abscess should be made prior to planning for an evisceration.
There is some theoretical concern for residual infection/inflammation to propagate into orbital abscess, cavernous venous thrombosis, meningitis, or encephalitis, and for the development of sympathetic ophthalmia, but there is limited data supporting this.
Enucleation
Enucleation is a more labor-intensive surgery but ensures complete removal of the globe and the proximal optic nerve. The benefit of an enucleation is completely removing the infected globe and associated tissue, and reduced risk of sympathetic ophthalmia. Complications include risk of orbital implant exposure and extrusion as it is only covered by the remaining tenon’s fascia and conjunctiva.
Pars Plana Vitrectomy
Pars Plana Vitrectomy can be an alternate option to reduce disease burden with possibility to preserve vision.
Follow-up
Patient's with Panophthalmitis are nearly always hospitalized with close observation and treated with a multidisciplinary approach. Without surgical intervention, if a patient is discharged, close follow-up is warranted and may required outpatient intravenous antibiotics through a PICC line. Recurrence has been reported within 24 months in the setting of primary liver abscesses and the sole use of medical therapy. [3]
Surgeon preference often dictates post-operative follow-up, however patients will be evaluated post-operatively in the hospital to ensure improvement prior to discharge. Outpatient antibiotic course is decided with the assistance of the medicine and infectious disease services. The first post-operative visit is typically at one to three weeks after intervention to assess healing and symptoms. At approximately five to eight weeks, patients are evaluated to assess whether a patient is adequately improved to book for a orbital implant placement .
Prognosis
Due to the severity of the disease, visual prognosis is typically poor even when the disease is treated in a timely manner. Virulence of the pathogen, medical history, time to presentation all play a significant role in the morbidity of the disease. However, mortality is rarely reported from Panophthalmitis (exceptions include the virulence of the pathogen and patient disease state), due to advancements available in detection and intervention for the disease process.
Case References
Case 1: Panophthalmitis and Orbital Cellulitis
Chief Complaint: 89-year-old female with a history of gradual redness and proptosis OD over 3 weeks.[26]
Case 2: Spontaneous rupture of a descemetocele with resulting Panophthalmitis
Case 3: Post-cataract surgery Panophthalmitis
Additional Resources
| Panophthalmitis Research Articles by Species | |
|---|---|
| Type of Organism | Species |
| Bacteria | |
| Gram Negative | |
| Bacillus cereus | |
| Serratia marcesans | |
| Clostridium perfringens | |
| Klebsiella pneumoniae | |
| Treponema pallidum | |
| Pseudomonas aeruginosa | |
| Citrobacter koseri | |
| Aeromonas hydrophila | |
| Gram Positive | |
| Staphylococcus aureus | |
| Streptococcus dysgalactiae | |
| Streptococcus mitis | |
| Nocardia spp. | |
| Mycobacterium spp. | |
| Fungi | |
| Rhizopus spp. | |
| Acrimonious faciforme | |
| Aspergillus fumigatus | |
| Candida auris | |
| Viral | |
| Dengue | |
| Herpes Simplex | |
| Human Immunodeficiency Virus | |
| Neoplasm | |
| Melanoma [Choroidal 1] [Choroidal 2] [Uveal] | |
| Retinoblastoma | |
| Secondary Source | |
| Acute Dacryocystitis | |
| Post-Cataract Surgery | |
| Penetrating Keratoplasty | |
- ↑ 1.0 1.1 Bennett, John E. “Endophthalmitis .” Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases, Ninth Edition, Ninth ed., vol. 2, Elsevier, 2019, pp. 1523–1530.
- ↑ 2.0 2.1 Pappuru RR, Dave VP, Pathengay A, Gangakhedkar S, Sharma S, Narayanan R, Tyagi M, Grzybowski A, Das T. Endophthalmitis Progressing to Panophthalmitis: Clinical Features, Demographic Profile, and Factors Predicting Outcome. Semin Ophthalmol. 2018;33(5):671-674. doi: 10.1080/08820538.2017.1416411. Epub 2017 Dec 19. PMID: 29256717.
- ↑ 3.0 3.1 3.2 3.3 3.4 Chen KJ, Chen YP, Chao AN, Wang NK, Wu WC, Lai CC, Chen TL. Prevention of Evisceration or Enucleation in Endogenous Bacterial Panophthalmitis with No Light Perception and Scleral Abscess. PLoS One. 2017 Jan 5;12(1):e0169603. doi: 10.1371/journal.pone.0169603. PMID: 28056067; PMCID: PMC5215906.
- ↑ Ihde, D. C., Armstrong, D. Clinical spectrum of infection due to Bacillus species. Am. J. Med. 55: 839-845.
- ↑ Weller, P. F., Nicholson, A., Braslow, N. Spectrum of bacillus bacteremias in heroin addicts. Arch. Intern. Med. 139:293-294, 1979.
- ↑ Oude Lashof AM, Rothova A, Sobel JD, Ruhnke M, Pappas PG, Viscoli C, Schlamm HT, Oborska IT, Rex JH, Kullberg BJ (2011) Ocular manifestations of candidemia. Clin Infect Dis : an official publication of the Infectious Diseases Society of America 53(3):262–268. doi:10.1093/cid/cir355
- ↑ 7.0 7.1 Shamsuddin D, Tuazon CU, Levy C, Curtin J. Bacillus cereus panophthalmitis: source of the organism. Rev Infect Dis. 1982 Jan-Feb;4(1):97-103. doi: 10.1093/clinids/4.1.97. PMID: 6803328.
- ↑ Vinay Kumar MBBS, MD, FRCPath, inRobbins & Cotran Pathologic Basis of Disease, 2021
- ↑ Moshfeghi AA, Charalel RA, Hernandez-Boussard T, Morton JM, Moshfeghi DM (2011) Declining incidence of neonatal endophthalmitis in the United States. Am J Ophthalmol 151(1):59–65. doi:10.1016/j.ajo.2010.07.008, e51
- ↑ Bouza, E., Grant, S., Jordan, M. C., York, R. H., Sulit, H. L., Bacillus cereus endogenous panophthalmitis. Arch. Ophthalmol. 97:498-499, 1979.
- ↑ 11.0 11.1 11.2 Davenport R, Smith C. Panophthalmitis due to an organism of the Bacillus subtilis group. Br J Ophthalmol. 1952;36(7):389-392. doi:10.1136/bjo.36.7.389
- ↑ 12.0 12.1 Chai HX, Samsudin A, Jayaraman K, Chong MF. The Noxious Intruder of the Eye: Endogenous Klebsiella Panophthalmitis. Cureus. 2021 Oct 12;13(10):e18725. doi: 10.7759/cureus.18725. PMID: 34790480; PMCID: PMC8585515.
- ↑ Tuberculosis Uveitis. American Academy of Ophthalmology, EyeWiki. November 2, 2021.
- ↑ 14.0 14.1 Gupta P, Singh R, Gupta S, Kumar A, Kakkar N. Tuberculosis presenting as posttraumatic panophthalmitis. Oman J Ophthalmol. 2016 Jan-Apr;9(1):52-4. doi: 10.4103/0974-620X.176102. PMID: 27013830; PMCID: PMC4785710.
- ↑ Yang, Boyu. “Mycobacterium Tuberculosis Review.” Microbewiki, https://microbewiki.kenyon.edu/index.php/Mycobacterium_tuberculosis_review.
- ↑ Hsu HY, Lee SF, Hartstein ME, Harocopos GJ. Clostridium perfringens Keratitis Leading to Blinding Panophthalmitis. Cornea. 2008 Dec;27(10):1200-3. doi: 10.1097/ICO.0b013e318180e5a1. PMID: 19034143.
- ↑ Deramo VA, Lai JC, Winokur J, Luchs J, Udell IJ. Visual outcome and bacterial sensitivity after methicillin-resistant Staphylococcus aureus-associated acute endophthalmitis. Am J Ophthalmol. 2008 Mar;145(3):413-417. doi: 10.1016/j.ajo.2007.10.020. Epub 2008 Jan 11. PMID: 18191097.
- ↑ Samanta TK, Biswas J, Gopal L, AnandA, Kumarasamy N, Solomon S. Panophthalmitis due to Rhizopus in an AIDS Patient: A clinicopathological study. Indian J Ophthalmol 2001;49:49-5
- ↑ Hassman LM, Chung MM, Gonzalez M, Bessette AP. Herpetic Panophthalmitis: A Diagnostic Dilemma. Ocul Immunol Inflamm. 2020;28(1):116-118. doi: 10.1080/09273948.2018.1546404. Epub 2018 Nov 16. PMID: 30444434.
- ↑ Center for Disease Control and Prevention (CDC) “About Dengue: What You Need to Know.” Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, 23 Sept. 2021, https://www.cdc.gov/dengue/about/index.html.
- ↑ 21.0 21.1 Radhakrishnan R, Cornelius R, Cunnane MB, Golnik K, Morales H. MR imaging findings of endophthalmitis. The Neuroradiology Journal. 2016;29(2):122-129. doi:10.1177/1971400916633480
- ↑ Ly V, Sallam A. Fungal Endophthalmitis. [Updated 2022 Jun 27]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK559257/
- ↑ Schachat, Andrew P, et al. “Foscarnet.” Foscarnet - an Overview | ScienceDirect Topics, https://www.sciencedirect.com/topics/neuroscience/foscarnet.
- ↑ Blumenkranz MS, Culbertson WW, Clarkson JG, et al. Treatment of the acute retinal necrosis syndrome with intravenous acyclovir. Ophthalmology 1986, 93: 296–300.
- ↑ Emerson GG, Smith JR, Wilson DJ, et al. Primary treatment of acute retinal necrosis with oral antiviral therapy. Ophthalmology 2006, 113: 2259–2261.
- ↑ https://webeye.ophth.uiowa.edu/eyeforum/cases/case16.htm