Papilledema
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Papilledema is a term that is exclusively used when a disc swelling is secondary to increased intracranial pressure (ICP). It must be distinguished from optic disc swelling owing to other causes, which is simply termed "optic disc edema." Papilledema must also be distinguished from pseudopapilledema, such as optic disc drusen. Since the root cause of papilledema is increased intracranial pressure (ICP), this is an alarming sign which may presage such entities as brain tumor, central nervous system (CNS) inflammation, cerebral venous thrombosis, or idiopathic intracranial hypertension (IIH).
Etiology
Papilledema is due to increased intracranial pressure from any cause. Normal ICP is typically less than 250 mm of water in adults when measured with a manometer and is less than 280 mm of water in children. Papilledema results from orthograde axoplasmic flow stasis at the optic nerve head, leading to edema of the nerve from the increased intracranial pressure on the nerve behind the eye. Continued pressure can result in loss of axons and eventual optic atrophy secondary to intraneuronal ischemia, ultimately causing vision loss.[1] With optic atrophy there is little or no edema seen, even with continuing increased intracranial pressure, since dead fibers do not swell.
Increased intracranial pressure can be caused by 5 patho-mechanisms:
- When the skull is too small for the brain (eg, craniosynostosis)
- When the brain volume becomes too large for the skull, as with a space-occupying lesion (eg, tumor, hemorrhage), or brain edema (eg, trauma)
- When there is obstruction of the cerebrospinal fluid (CSF) flow (eg, colloid cyst obstructing the foramen of Monroe)
- When there is increased production of CSF (eg, choroid plexus papilloma)
- When there is reduced absorption of CSF (eg, meningitis, cerebral venous thrombosis)
The time course for development of papilledema may be weeks if there is only a slow and mild rise in intracranial pressure, but severe and rapid changes in pressure can cause papilledema to present within a few hours to a day. The mechanism of Idiopathic intracranial hypertension is not clearly understood; however, proposed mechanisms include increased venous sinus pressure from venous sinus stenosis, and reduced absorption, to name a few. [2]
Risk factors
Risk factors for papilledema are those that raise intracranial pressure as mentioned above. This includes space-occupying lesions such as tumor or subarachnoid hemorrhage, decreased absorption of cerebrospinal fluid, change in the dynamics of cerebrospinal flow through the ventricles (for example by obstruction of the 4th ventricle), or rarely by increased production of cerebrospinal fluid.
Risk factors for idiopathic intracranial hypertension include recent weight gain, and underlying associated conditions such as obesity, polycystic ovarian syndrome, anemia, thyroid disease, other endocrinological disease, some medications, and obstructive and sleep apnea, to name a few. [2]
Diagnosis
Fundus examination noting optic disc swelling (usually bilateral, unless 1 disc is atrophic) in the presence of high increased intracranial pressure is the key to diagnosis. Other diagnostic tools to exclude pseudo disc swelling include ultrasonography (B-scan), fluorescein angiography, fundus autofluorescence, and, more recently, optical coherence tomography with enhanced depth imaging of the optic nerve head. When presented with the possibility of papilledema, the first approach is to check the blood pressure to rule out malignant hypertension, and to send the patient for urgent imaging by CT or MRI to rule out a space-occupying lesion and a venogram to rule out cerebral venous thrombosis. The optimal imaging modality is MRI and an accompanying MRV with contrast to rule out a venous sinus thrombosis. A lumbar puncture with opening pressure and fluid sent for CSF composition is done to confirm the diagnosis of high ICP and to assess for other possible underlying causes of the high ICP.
The diagnosis of IIH as a cause of papilledema is one of exclusion, the modified Dandy criteria help in diagnosing patients with IIH in order to safely exclude other causes and initiate the treatment in a timely manner to prevent visual function loss.
| The modified Dandy Criteria to Diagnose IIH[3] |
|---|
Probable IIH: if criteria 1–4 are met with bilateral papilledema present, and the measured CSF pressure is lower than the specified for a definite diagnosis. |
Disc appearance
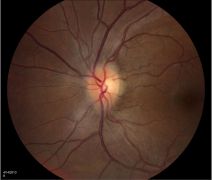
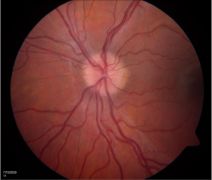
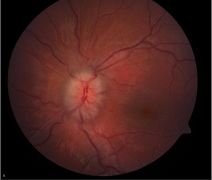
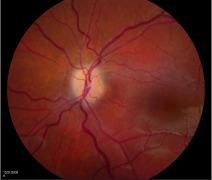
When performing a fundus examination in a patient with papilledema, variable signs can be found, depending on the severity of the condition. Looking at the fundus, the ophthalmologist must pay attention to the following:
- the optic nerve head : retinal nerve fiber opacification, elevation of the margins, hyperemia, and obliteration of the cup
- vascular features related to congestion: venous dilation, vascular tortuosity, hemorrhages, cotton wool spots, and exudates (which can extend to the macula, causing a macular star appearance). If spontaneous venous pulsations (SVPs) are present, this suggests that ICP is probably not high at this time; however. the ICP may have been high before simply due to diurnal variations in intracranial pressure. The absence of SVP on exam is not always suggestive of high ICP, because it can be absent in 10%-20% of the normal population.[4]
- mechanical features: retinal folds ( eg, peripapillary circumferential water marks called Paton's lines) , and choroidal folds caused by posterior globe deformation
Chronic papilledema may show all the aforementioned signs, in addition to optic disc pallor, along with decreasing swelling due to loss of axons, gliosis (greying of the retinal fibers due to scarring), optociliary shunts, and refractile bodies.
The Frisén scale is a grading system that is used for papilledema.
It is important to note that the grade of papilledema does not reflect the severity of the intracranial pathology present; in other words, mild papilledema of grade 1 may still harbor a large intracranial tumor. However, grading of the papilledema is important, as it is a prognostic indicator for visual outcome, which means severe papilledema corroborates with a worse visual prognosis.
| Papilledema Grading System (Frisén Scale)[5] |
|---|
| Stage 0 - Normal Optic Disc Blurring of nasal, superior, and inferior poles in inverse proportion to disc diameter. Radial nerve fiber layer (NFL) without NFL tortuosity.
Rare obscuration of a major blood vessel, usually on the upper pole. |
| Stage 1 -Very Early Papilledema Obscuration of the nasal border of the disc. No elevation of the disc borders. Disruption of the normal radial NFL arrangement with
grayish opacity accentuating nerve fiber layer bundles. Normal temporal disc margin. Subtle grayish halo with temporal gap (best seen with indirect ophthalmoscopy). Concentric or radial retrochoroidal folds. |
| Stage 2 - Early Papilledema: Obscuration of all borders. Elevation of the nasal border. Complete peripapillary halo. |
| Stage 3 - Moderate Papilledema: Obscurations of all borders. Increased diameter of optic nerve head. Obscuration of 1 or more segments of major blood vessels
leaving the disc. Peripapillary halo-irregular outer fringe with finger-like extensions. |
| Stage 4 - Marked Papilledema: Elevation of the entire nerve head. Obscuration of all borders. Peripapillary halo. Total obscuration on the disc of a segment of a major
blood vessel. |
| Stage 5 - Severe Papilledema: Dome-shaped protrusions representing anterior expansion of the optic nerve head. Peripapillary halo is narrow and smoothly demarcated. Total obscuration of a segment of a major blood vessel may or may not be present. Obliteration of the optic cup. |
Fundoscopic examples
History
When papilledema is suspected, the patient's history must include the symptom of the chief complaint and associated symptoms, questions about possible differential diagnoses, and inquiry about risk factors associated with IIH.
1- Chief complaint and associated symptoms: papilledema can be asymptomatic. However, when symptoms occur, they can include systemic symptoms and visual symptoms.
Systemic symptoms include headaches, nausea, vomiting, and pulsatile tinnitus. Headache characteristics typically are positional, and they are worse in the mornings and when lying down. Pulsatile tinnitus occurs due to turbulence within the venous system from high intracranial pressure and is often described as a "whooshing" sound.
Visual symptoms include transient visual obscurations, which occur from transient ischemia to the optic nerve head. Although they can occur frequently, they are not an ominous sign for visual loss. Long-standing papilledema or fulminant papilledema, however, may result in severe peripheral visual field loss and may progress to involve central vision if rapid and fulminant, or untreated, or presenting late. Intermittent or constant binocular diplopia can occur as a result of unilateral or bilateral sixth nerve palsies, which are non-localizing neurological signs, and are thought to be related to the anatomical course of the sixth cranial nerve. The long course of the 6th cranial nerve is compressed against the clivus by the increased CSF pressure as it makes a sharp turn into the Dorello canal underneath the petroclinoid ligament of Gruber. A hyperopic shift can also occur from a posterior flattening of the globe that causes axial length shortening.
2- Questions about possible differential diagnoses: It is important to include in the history questions about previous space-occupying lesions, hypertension, vasculitis (fever, rash, bowel problems), meningitis ( fever, neck stiffness), cerebral venous thrombosis (personal or family history of hypercoagulable states, smoking, or oral contraceptive pills), and medications that might precipitate high ICP, such as tetracyclines, vitamin A and derivatives, lithium, steroids, or steroid withdrawal.
3- Questions about risk factors that may be associated with IIH: Recent weight gain, thyroid disease, anemia, polycystic ovarian syndrome, renal disease, and obstructive sleep apnea, to name a few.
Physical examination
The physical examination will include a complete neuro-ophthalmic examination and an urgent blood pressure check to rule out malignant hypertension. A neurological examination may be appropriate to exclude other focal neurologic deficits.
In the setting of acute papilledema, the bilateral optic disc swelling seen on the fundus examination is often accompanied by a normal afferent visual function including a normal best corrected visual acuity, color vision, lack of a relative afferent pupillary defect, and normal confrontation visual fields (apart from an enlarged blind spot). However, if central vision was lost without loss of peripheral visual field and with lack of a relative afferent pupillary defect, most likely there has been extension of the fluid into the macula, causing macular edema, in which case an OCT of the macula is useful.
In the setting of acute florid/fulminant papilledema or chronic untreated papilledema, the peripheral visual field can get constricted, and if left untreated it can extend to cause central visual loss.
Ocular motility examination may reveal a unilateral or bilateral 6th nerve palsy as non-localizing signs of increased intracranial pressure.
Diagnostic procedures
Ophthalmic investigations, neuro-imaging, and lumbar puncture are used to produce a diagnosis.
Ophthalmic investigations
- Spectral domain optical coherence tomography of the RNFL (SD-OCT RNFL) may play an objective role in detecting subtle disc swelling and in monitoring for improvement of the condition, but it should be interpreted along with the clinical picture, as sometimes improvement of swelling could occur in cases with optic atrophy.[6] In that sense, an OCT of the ganglion cell layer-inner plexiform layer (GCL-IPL) is more reliable in distinguishing improvement of thickness due to treatment response vs due to atrophy.[7]
- Stereo disc images may be helpful to in following the disc status over time.
- Formal (typically automated) perimetry commonly shows enlarged blind spots and nerve fiber layer-type visual field defects that are similar to those found in glaucoma; for example, a nasal step, an arcuate scotoma, and even a constricted field can be seen in severe cases. In severe chronic papilledema, worsening peripheral field loss may be seen and can eventually involve central vision. Acute fulminant cases may present with significant visual field or acuity loss and should be treated emergently.
- Fluorescein angiography can detect disc leakage in true optic disc edema, as opposed to disc staining without leakage in pseudopapilledema. In addition, autofluorescence or B-scan ultrasound and enhanced depth imaging optical coherence tomography of the optic nerve head may show pseudopapilledema (eg, optic disc drusen).
Neuro-imaging
The immediate objective of an urgent CT scan would be to rule out a space-occupying lesion, and combined with a CT venogram would be to rule out cerebral venous thrombosis in the appropriate context. Moreover, an MRI/MRV with contrast would assess for leptomeningeal enhancement if meningitis is suspected, and would assess features of high intracranial pressure. High intracranial pressure features assessed on MRI and MRV with contrast are not specific to IIH and can occur in any cause of high ICP[8] Neuro-imaging features of high ICP include:
- empty or partially empty sella
- dilated perioptic nerve sheath with prominent CSF
- vertical or horizontal optic nerve tortuosity
- posterior flattening of the globe. In some severe degrees of optic disc swelling, an intraocular optic nerve protrusion might be noted.
In addition:
- One important finding to look for in papilledema patients is tonsillar ectopia as such patients might be at risk of herniation when performing the LP depending on the severity.[8]
- MR venography (MRV) or CTV should also be considered to look primarily for venous sinus thrombosis and secondarily for stenosis.
Lumbar puncture
Lumbar puncture that is done after neuro-imaging demonstrates no risk for herniation; it is done to document the opening pressure and CSF composition (ruling out neoplastic, infectious, or inflammatory causes). At a minimum, opening pressure along with glucose, protein, cell count and differential, and culture are obtained.
Differential diagnosis
Papilledema must be distinguished from other entities that may mimic papilledema. The following entities may cause bilateral disc edema with no high intracranial pressure, and in some cases a normal afferent visual function. A complete history and a thorough examination often will guide the direction of diagnosis.
- Anomalous optic discs: buried drusen, tilted disc, hypoplastic discs
- Diabetic papillopathy
- Hypertensive papillopathy
- Obstructive sleep apnea
- Intraocular or periocular inflammation: posterior scleritis, perineuritis, uveitis with associated optic disc edema ( eg, sarcoidosis, toxoplasmosis, and VKH), neuro-retinitis
- Compressive optic neuropathy (optic nerve sheath meningioma)
- Optic neuritis
- Thyroid ophthalmopathy
- Central retinal vein occlusion
- Nonarteritic ischemic optic neuropathy
- Infiltrative optic neuropathy and tumors (lymphoma, leukemia)
Management
Management goals are: 1) Treating underlying cause, 2) saving vision, and 3) symptomatic relief (eg, headache).[9]
- Treating the underlying cause
- If malignant hypertension was found, then the patient must be sent to the emergency department for further management. Even with malignant hypertension cases, the patient with papilledema must still proceed with neuro-imaging. FISH is a condition that combines both fulminant IIH and systemic hypertension.
- If a mass is present, primary therapy should be directed toward that lesion (eg, surgery)
- If acute cerebral venous thrombosis was seen, the patient must be sent to the thrombosis team immediately. It is best to avoid acetazolamide in cerebral venous thrombosis, as it can precipitate the condition; however, it can later be added on when the patient has started on anti-coagulation therapy.
- Medications (tetracyclines, vitamin A analogues, etc.) are thought to be causative, so they should be discontinued.
- If the final diagnosis was IIH; please see below for management.
- Saving vision and symptomatic relief (eg, for headache)
- Treat the underlying cause.
- Administer medications to lower ICP.
Management of IIH
If the final diagnosis was IIH, a consensus for treatment depends on the severity of the condition. The spectrum of this condition may range from asymptomatic with no visual field defects to fulminant with severe vision loss. If the patient has no immediate visual threat, then a more conservative approach can be taken, with weight reduction and medications. If the patient's condition is vision-threatening, then a more aggressive approach should be taken, such as admission and surgical intervention.
Conservative management
- Weight reduction: a recommended amount of weight loss (5%-10%) was found to improve the symptoms and signs of high ICP[10]
- Avoid precipitating medications.
- Control underlying risk factors (eg, thyroid disease, or obstructive sleep apnea to name a few).
- Stop smoking.
Medical management
- First line of treatment is acetazolamide (a carbonic anhydrase inhibitor). The idiopathic intracranial hypertension trial (IIHT) has provided strong support for safe and effective use of acetazolamide, combined with weight reduction, in effectively treating IIH and improving papilledema in these patients with mild vision loss.[11]
- Topimarate and furosemide (Lasix) are considered second-line agents that can be used if the patient is intolerant to acetazolamide or if acetazolamide does not work.
Surgical management
- If there is visual loss only, optic nerve sheath fenestration can be considered.
- If there are headaches only or headaches combined with visual loss, CSF diversion procedures such as a ventriculoperitoneal shunt or a lumboperitoneal shunt can be considered.
- Dural venous sinus stenting may also be considered for patients with increased ICP who have significant venous sinus stenosis, evidenced by a venous sinus pressure high gradient difference.
- Bariatric surgery can be considered for weight loss.
Surgical complications including shunt failure, breakage, or infection in CSF shunting, or visual loss related to surgical trauma in optic nerve fenestration have been reported.
Fulminant IIH
Patients with acute to subacute onset of symptoms within weeks, who present with severe papilledema and severe visual loss (and follow the modified Dandy criteria for diagnosis of IIH) are considered to have fulminant IIH. These patients are critically ill and need to be admitted immediately. These patients will have an expedited MRI/MRV with contrast and a lumbar puncture for CSF opening pressure and composition. They can be started on acetazolamide (up to 4 grams a day), and can also be given a 3-day course of intravenous steroids, which is thought to help reduce the severity of the congestion; however, there is no clear evidence for the latter. A lumbar drain should be considered while waiting for the ultimate surgical intervention, which should be done urgently. Optic nerve sheath fenestration or a CSF diversion procedure can be done to reduce the risk of further visual loss, and with hopes to regain some visual field capacity. These patients need to be followed closely until they pass the critical period and remain stable.
Prognosis
Chronic papilledema leads to axon loss with constriction of the visual field, loss of disc substance and, in the end, loss of central acuity. Patients (especially those with IIH) need to be followed according to the severity of their presentation to prevent vision loss. The most common cause of recurrence of IIH is weight gain.
Additional Resources
- The Neuro-Ophthalmology Virtual Education Library: library.med.utah.edu/NOVEL/
- The North American Neuro-Ophthalmolgy Society (NANOS): www.nanosweb.org
- Porter D, Vemulakonda GA. Blood Pressure. American Academy of Ophthalmology. EyeSmart/Eye health. https://www.aao.org/eye-health/anatomy/blood-pressure-list. Accessed January 31, 2025.
Reference
- ↑ Lee A, Rigi M, Al marzouqi S, Morgan M. Papilledema: epidemiology, etiology, and clinical management 2015;7-47. Available from https://doi.org/10.2147/EB.S69174
- ↑ 2.0 2.1 Keira A Markey, Susan P Mollan, Rigmor H Jensen, Alexandra J Sinclair. Understanding idiopathic intracranial hypertension: mechanisms, management, and future directions. Lancet Neurol. 2016 Jan;15(1):78-91. doi: 10.1016/S1474-4422(15)00298-7. Epub 2015 Dec 8.PMID: 26700907DOI: 10.1016/S1474-4422(15)00298-7
- ↑ Friedman DI, Liu GT, Digre KB. Revised diagnostic criteria for the pseudotumor cerebri syndrome in adults and children. Neurology. 2013 Sep 24;81(13):1159-65. doi: 10.1212/WNL.0b013e3182a55f17. Epub 2013 Aug 21. PMID: 23966248.
- ↑ Jacks A, Miller N. Spontaneous retinal venous pulsation: aetiology and significance. J Neurol Neurosurg Psychiatry. 2003;74:7–9.
- ↑ Reference: Frisén L. Swelling of the optic nerve head: A staging scheme. J Neurol Neurosurg Psychiatry 1982; 45:13-18
- ↑ Wang JK, Kardon RH, Kupersmith MJ, Garvin MK. Automated quantification of volumetric optic disc swelling in papilledema using spectral-domain optical coherence tomography. Invest Ophthalmol Vis Sci. 2012;53:4069– 4075
- ↑ Kardon RH. Role of the macular optical coherence tomography scan in neuro-ophthalmology. J Neuroophthalmol. 2011;31:353–361.
- ↑ 8.0 8.1 Friedman DI, Liu GT, Digre KB. Revised diagnostic criteria for the pseudotumor cerebri syndrome in adults and children. Neurology. 2013 Sep 24;81(13):1159-65. doi: 10.1212/WNL.0b013e3182a55f17. Epub 2013 Aug 21. PMID: 23966248.
- ↑ Mollan SP, Davies B,Silver NC, et al. J Neurol Neurosurg Psychiatry 2018;89:1088–1100.
- ↑ Wong R, Madill SA, Pandey P, Riordan-Eva P. Idiopathic intracranial hypertension: the association between weight loss and the requirement for systemic treatment. BMC Ophthalmol. 2007;7:15.
- ↑ Smith S, Friedman D. The Idiopathic Intracranial Hypertension Treatment Trial: A Review of the Outcomes 2017;1303-1310. Available from https://doi.org/10.1111/head.13144