Tarsorrhaphy
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Background
Tarsorrhaphy is a safe and relatively simple procedure in which part, or all the upper and lower eyelids are joined together to cover the eye partially or completely. Tarsorrhaphies are highly effective in the management of nonhealing epithelial defects and other corneal surface pathology by creating a more hospitable environment for corneal healing.[1] Tarsorrhaphies can be permanent or temporary, total or partial, and can be further classified into short duration tarsorrhaphies without sutures, temporary suture tarsorrhaphies, permanent suture tarsorrhaphies, and more extensive tarsorrhaphies that involve mobilization of skin or tarsal plate flaps.[2]
A temporary tarsorrhaphy promotes corneal healing during a short period of disease or exposure and is appropriate when improvement over a few weeks is anticipated. Temporary lid closure procedures include conventional suture, glue, temporary external lid weights, and neurotoxin tarsorrhaphies.[1] A "permanent" tarsorrhaphy offers corneal protection when long-term closure is needed and can be released over time if improvement occurs. Tissue excision can still be confined to the posterior lamella to preserve the anterior lamella and lash line which provides a better cosmetic result when opened in the future. Common permanent lid closure options include the lateral or medial tarsorrhaphy and gold or platinum-weighted implants. A pillar tarsorrhaphy is another option for a “permanent,” yet reversible narrowing of the interpalpebral fissures. This involves connecting the upper and lower lid on either side of the pupil to provide adequate closure while permitting examination of the cornea.[3] A neurotoxin tarsorrhaphy, employs the use of botulinum toxin A (Botox©) to create a protective ptosis as a safe and effective temporary tarsorrhaphy with the added benefit of an easier slit-lamp examination.[4][5] Tarsorrhaphies are a safe and highly effective option for treatment of persistent epithelial defects with minimal complications.[6]
Preoperative Considerations
Pre-procedural evaluation:
Prior to the procedure, a full ophthalmic examination should be performed and documented. A thorough slit lamp biomicroscopic examination should document corneal pathology and the size and location of any defects or corneal ulcers. Careful examination of the palpebral conjunctiva using double eversion to look for foreign bodies or keratinization should also be performed. External examination of eyelid abnormalities, the degree of lagophthalmos, and assessment of corneal sensitivity are critical in determining what type of tarsorrhaphy is appropriate (permanent vs. temporary) and deciding on the extent of tarsorrhaphy (lateral vs. medial vs. central vs. total) to be performed. The length of tarsorrhaphy to be performed is determined by gently pinching the upper and lower eyelids together with forceps or manually to achieve desired closure.
Indications:
Corneal exposure secondary to:
- Lagophthalmos due to facial nerve palsies (neurogenic exposure keratopathy), cicatricial damage to the eyelids, anorexia nervosa, leprosy, Ramsay Hunt Syndrome Type 2, orbital tumors, and thyroid eye disease [1][7][8][9]
- Proptotic exposure secondary to thyroid eye disease or other inflammatory orbital disease, and orbital tumors [1]
- Eyelid malposition secondary to trauma, surgery, scaring, and floppy eyelid syndrome
- Inadequate blinking secondary to reduced corneal sensation, Riley Day Syndrome/Familial Dysautonomia, severe brain injury, or prolonged sedation [1]
Non-healing corneal defects or pathology:
- Non-healing epithelial defects
- Neurotrophic corneal ulceration (CN V deficit, herpes simplex virus (HSV) or varicella zoster virus (VZV) keratitis) [1]
- Recalcitrant dry eye
- Progressive corneal thinning or descemetocele
- Limbal stem cell deficiency
- Palytoxin keratitis/keratoconjunctivitis/coral keratitis
In conjunction with other surgeries:
- To prevent conjunctival swelling (chemosis) and exposure after ocular surgery
- To retain a prosthesis, Boston Keratoprosthesis, or other device in patients with anophthalmia or after evisceration or enucleation
- Tarsal kink repair [10]
- In conjunction with globe reconstruction in repair of traumatic globe luxation
- In ocular graft versus host disease in combination with amniotic membrane
Contraindications:
Tarsorrhaphies have few contraindications and can be easily reversed if needed. Relative contraindications for complete tarsorrhaphy include active microbial (bacterial or fungal) keratitis where ongoing visualization of the ocular surface is needed. A partial tarsorrhaphy may be beneficial for ulcer treatment and epithelial defect healing. A complete tarsorrhaphy in the remaining good eye of a monocular patient is generally avoided but may be necessary to preserve vision. Likewise, a bilateral, complete tarsorrhaphy is undesirable, and alternative treatment modalities such as amniotic membrane devices and bandage soft contact or scleral contact lens may be considered to prevent complete visual occlusion.[11]
Surgical Techniques
Temporary Bolster Tarsorrhaphy
Technique
- Pre-operatively, the amount of tarsorrhaphy to be performed is determined.
- Topical anesthetic is instilled, and the central area of the upper and lower eye lid is injected with local anesthetic (lidocaine 1-2% with epinephrine).
- Two bolsters of the surgeon's choice of size and material (plastic tubing, red robin catheter, cotton wool balls, etc.) are prepared. If it is anticipated that the suture will be removed within 2 weeks and there is no skin compromise, bolsters may not be necessary.
- An absorbable or nonabsorbable 4-0 to 6-0 suture is initially passed through the bolster material.
- The needle is then passed through the upper lid skin 3-4 mm above the lid margin, exiting through the meibomian gland orifices.
- The same suture is passed through the meibomian gland orifices of the opposite lower lid margin and retrieved from the skin 3-4 mm below the lid margin.
- The suture is then passed through the second bolster.
- The needle is then turned around and passed 3-4 mm below the lower lid skin, through the lid margin and retrieved from the meibomian gland orifices.
- The needle is then passed through the meibomian gland orifices of the upper lid margin and retrieved 3-4 mm above the upper lid margin and engages the upper bolster.
- The suture is then tied over the bolster to complete the tarsorrhaphy. The suture should be snug to prevent incomplete lid opposition when intraoperative edema resolves.
- Additional bolsters may be placed depending on the area of coverage desired.[1][12]
In summary: The suture is passed through the bolster, followed by the upper eyelid, then the lower eyelid, then the second bolster. Once the second bolster is engaged, the suture is turned around and placed through the second bolster, then the lower eyelid, then the upper eyelid, and the bolster. The suture is then tied to complete the tarsorrhaphy.[12]
The temporary suture tarsorrhaphy can be placed anywhere along the lid margins. If inspection of the cornea at intervals is desired, a drawstring tarsorrhaphy technique can be utilized.
Drawstring Tarsorrhaphy
Technique
- Pre-operatively, the amount of tarsorrhaphy to be performed is determined.
- Topical anesthetic is instilled, and the central area of the upper and lower eye lid is injected with local anesthetic (lidocaine 1-2% with epinephrine).
- Three bolsters of the surgeon's choice of size and material (plastic tubing, red robin catheter, cotton wool balls, etc.) are prepared.
- An absorbable or nonabsorbable double armed 4-0 to 6-0 suture is initially passed through the bolster material.
- The needle is then passed through the upper lid skin 3-4 mm above the lid margin, exiting through the meibomian gland orifices.
- The same suture is passed through the meibomian gland orifices of the opposite lower lid margin and retrieved from the skin 3-4 mm below the lid margin.
- The second arm of the double armed suture is then passed in an identical fashion along the other side of the bolster and through the eyelid as above
- The final step is to create the drawstring, the 2 suture arms are passed through the 3rd bolster (it is commonplace to make this smaller than the other ones)
- The needles can be removed and then an air knot is created about 3-4cm below the eyelid
- Now the drawstring can be used.
- The smaller bolster can be pulled up to "lock" the 2 larger bolsters in place providing excellent closure
- If the eye needs to be opened the smaller bolster is separated from the larger lower eyelid bolster, which allows the eyelid to be examined or to receive treatment
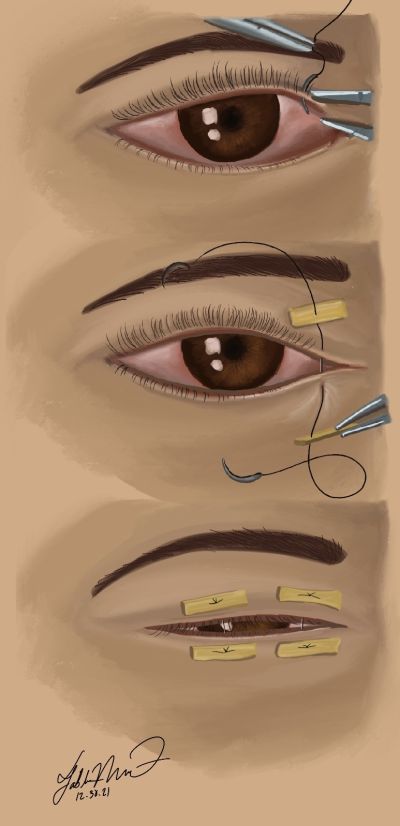
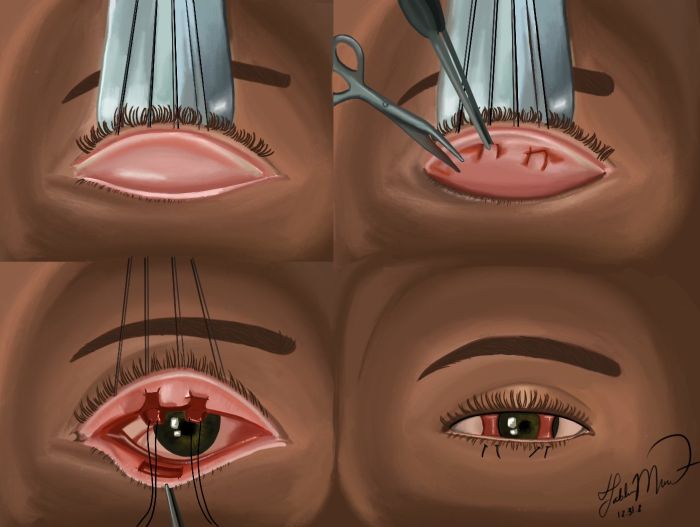
Permanent lateral tarsorrhaphy
Technique
- Pre-operatively, the amount of tarsorrhaphy to be performed is determined.
- Topical anesthetic is instilled, and the central area of the upper and lower eye lid is injected with local anesthetic (lidocaine 1-2% with epinephrine).
- A #11 or #15 Bard-Parker blade is used to make an incision along the grey line of the lateral lower lid of the desired length to a depth of 2 mm.
- Stevens or Westcott scissors are used to dissect between the anterior and posterior lamella to a depth of 3-4 mm making sure to stay parallel to the tarsal plate.
- The mucocutaneous junction of the posterior lamella of the lower lid is excised using Westcott scissors.
- Attention is then turned to the upper lid where the identical technique is used.
- A 5-0 or 6-0 Vicryl suture is then passed partial thickness through the posterior lamella of the lower lid 2-3 mm inferior to the lid margin, to the posterior lamella of the upper lid exiting 2-3 mm superior to the lid margin in an interrupted fashion. It is important to ensure that each suture pass is partial thickness only, as a full thickness pass could cause suture-induced irritation post-operatively. It is also important for the lamellar sutures to exit and enter within the meibomian gland orifices of the eyelid margin to avoid erosion when placed too anteriorly. The number of sutures needed is determined by the length of the tarsorrhaphy.
- The anterior lamella of the lower lid is then sutured to the anterior lamella of the upper lid using absorbable or permanent 5-0 to 6-0 suture in an interrupted fashion.[1][13]
Permanent medial tarsorrhaphy
A medial tarsorrhaphy is another useful technique when applicable. This technique is modified to avoid damage to the canaliculi and involves making a V-shaped incision peripheral to the canaliculi to the upper and lower lids. The medial tarsorrhaphy is advantageous in that it does not interfere with peripheral vision.
Technique
- Pre-operatively, the amount of tarsorrhaphy to be performed is determined.
- Topical anesthetic is instilled, and the central area of the upper and lower eye lid is injected with local anesthetic (lidocaine 1-2% with epinephrine).
- A V-shaped incision just peripheral to the canaliculi to the upper and lower lid is made.
- A bowman prob can be placed into the canaliculus to help identify the canaliculus.
- A #15 Bard-Parker blade is used to cut through the skin and orbicularis adjacent to the canaliculus.
- Dissection of the anterior and posterior lamella is carried out along the length of the incision.
- The posterior lamellae of the upper and lower lids are sutured together using 5-0 or 6-0 Vicryl suture in interrupted fashion.
- The anterior lamella is then sutured together with 6-0 Vicryl suture in interrupted fashion.
Pillar tarsorrhaphy
Technique:
- Topical anesthetic is instilled, and the central area of the upper and lower eye lid is injected with local anesthetic (lidocaine 1-2% with epinephrine).
- 4-0 silk sutures are passed through the upper eyelid at the level of the meibomian gland orifices and the eyelid is everted over a speculum.
- A #15 blade is used to make 2 parallel incisions that are connected at one end to develop two pillars of tarsoconjunctiva tissue, one corresponding to the medial limbus and one corresponding to the lateral limbus.
- The pillars are elevated with Westcott scissors and dissection is carried out above the superior border of the tarsus of the upper eyelid creating a rectangle or pillar on each tongue or pillar of tissue.
- Attention is then directed to the lower eyelid where a rectangle of tarsoconjunctival tissue is excised at the eyelid margin with the #15 blade and Westcott scissors at the level of each of pillar.
- Each arm of a double armed 5-0 Vicryl suture is passed partial thickness through each pillar. Each arm is then placed through the lower eyelid through the exposed area of the previously excised rectangle of tissue and retrieved from the skin to transpose each pillar to the raw surface of the lower eyelid.
- The sutures are tied over cotton bolsters to complete the tarsorrhaphy.
- The 4-0 silk everting sutures are removed, and the eyelids are inspected to ensure appropriate closure.[3][14]
Glue tarsorrhaphy
Use of any tissue glue or cyanoacrylate glue gel can be a simple, alternative method for creating a temporary tarsorrhaphy unstable for invasive procedures, for temporary eyelid apposition for persistent epithelial defects, or for ease of use in any setting.[5][15]
Technique
After washing and preparing the eye thoroughly within the lid margins and fornices, the glue gel of choice is applied along closed lateral eyelid margins using the standard applicator tip supplied with the gel. Using the same applicator tip, the gel is spread medially along the eyelid margins to achieve the desired amount of closure. The glue is allowed to dry for 15 – 20 seconds. Once dry, the eyelids stay closed for approximately 2 weeks.[5][16] Some report longer lasting closure with a recent study in a pediatric population reporting that cyanoacrylate glue tarsorrhaphies last an average of 4.5 weeks (range 0.5-13 weeks).[17] It can be reversed by cutting the eyelashes following the application of lidocaine gel.
Neurotoxin tarsorrhaphy
The use of botulinum toxin injection to create a neurotoxin-induced neuroparalysis of the levator palpebrae has shown to be both safe and efficacious.[18] However, the results can be relatively unpredictable and further studies are needed to determine safe and efficacious dosage for such a procedure. Typically, 5-15 units are injected into the levator muscle to induce a protective ptosis; however, doses ranging from 2.5 units up to 30 units have been reported.[18]
Technique
The upper eyelid is prepped with Betadine solution and the desired concentration is drawn up into a 1 mL insulin or tuberculin syringe. Introduce the needle tip of a 23 to 26-gauge needle just below the superior orbital rim along the mid-pupillary plane and passed against the orbital roof for 1 to 2 cm. The desired amount of botulinum toxin is injected, and the needle and syringe are discarded appropriately. The patient should then be monitored closely for appropriate healing and resolution of ptosis with return of levator function.[18][19] Repeat injection may be necessary.
Postoperative management
Antibiotic ointment is often placed over the eyelids with a patch placed after surgery. The patient should follow up closely after surgical treatment on day 1, week 1, and at intervals appropriate for the underlying condition thereafter. Patients should refrain from eye rubbing or wearing make-up until approved by the physician. Additionally, driving should be restricted until approved by the ophthalmologist. Many patients will experience peripheral vision loss with lateral tarsorrhaphies, so adjustments to the patient’s daily activities should be addressed. Appropriate topical lubricating drops and antibiotics should be prescribed to preserve the cornea and treat the underlying disease.
Complications
As with any surgical procedure, there is a risk of bleeding, infection, swelling, and/or damage to surrounding structures. Other risks include premature separation, the need for reoperation, ankyloblepharon formation, pyogenic or suture granulomas, trichiasis, distichiasis, skin breakdown, lid margin deformities, and premature separation.[6] Pillar tarsorrhaphies have a unique complication of ectropion.[20] Reported side effects of neurotoxin tarsorrhaphies included preseptal hemorrhage, inadvertent injury to the globe, and superior rectus under action resulting in diplopia which can last up to 9 months after injection.[18]
Outcomes
Tarsorrhaphy is a safe and effective procedure that can be used alone or in combination with other surgical techniques to protect the cornea from exposure and for the treatment of nonhealing epithelial defects, corneal surface disease, and other ocular and eyelid disorders. The overall success rate for tarsorrhaphy for nonhealing epithelial defects has been reported to be up to 80 to 100% for complete healing. With standard techniques, the procedure is relatively straightforward with minimal risks and should be considered early when epithelial corneal defects are not responding to medical therapy. The effects of traditional suturing techniques last from 2 to 8 weeks. Ideally, temporary tarsorrhaphies promote healing within 2 weeks and can be removed at that time. If adequate healing has not occurred and the patient requires long-term treatment, a permanent tarsorrhaphy can be performed which can still be opened at a later date.[21]
Additional Resources
https://www.aao.org/oculoplastics-center/facial-nerve-palsy-2
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 Rajak S, Rajak J, Selva D. Performing a tarsorrhaphy. Community Eye Health. 2015;28(89):10-11.
- ↑ Cosar CB, Cohen EJ, Rapuano CJ, Maus M, Penne RP, Flanagan JC, Laibson PR. Tarsorrhaphy: clinical experience from a cornea practice. Cornea. 2001 Nov;20(8):787-91. doi: 10.1097/00003226-200111000-00002. PMID: 11685052.
- ↑ 3.0 3.1 Tanenbaum M, Gossman MD, Bergin DJ, Friedman HI, Lett D, Haines P, McCord CD Jr. The tarsal pillar technique for narrowing and maintenance of the interpalpebral fissure. Ophthalmic Surg. 1992 Jun;23(6):418-25. PMID: 1513540.
- ↑ Khairy H. Botulinum toxin A-induced ptosis: A safe and effective alternative to surgical tarsorrhaphy for corneal protection. Journal of the Egyptian Ophthalmological Society. 2014;107(1):20-22. doi:10.4103/2090-0686.134937.
- ↑ 5.0 5.1 5.2 Ehrenhaus M, D'Arienzo P. Improved Technique for Temporary Tarsorrhaphy With a New Cyanoacrylate Gel. Arch Ophthalmol. 2003;121(9):1336–1337. doi:10.1001/archopht.121.9.1336.
- ↑ 6.0 6.1 Tzelikis PF, Diniz CM, Tanure MA, Trindade FC. Tarsorrafia: aplicações em um Serviço de Córnea [Tarsorrhaphy: applications in a Cornea Service]. Arq Bras Oftalmol. 2005 Jan-Feb;68(1):103-7. Portuguese. doi: 10.1590/s0004-27492005000100019. Epub 2005 Mar 30. PMID: 15824813.
- ↑ Fu L, Patel BC. Lagophthalmos. In: StatPearls. Treasure Island (FL): StatPearls Publishing; August 10, 2020.
- ↑ Hogeweg M, Keunen JE. Prevention of blindness in leprosy and the role of the Vision 2020 Programme. Eye. 2005: 19, 1099–1105.
- ↑ Lee V, Currie Z, Collin JRO. Ophthalmic Management of Facial Nerve Palsy. Eye. 2004; 18: 1225-1234.
- ↑ Naik MN, Honavar SG, Bhaduri A, Linberg JV. Congenital horizontal tarsal kink: a single-center experience with 6 cases. Ophthalmology. 2007 Aug;114(8):1564-1568. DOI: 10.1016/j.ophtha.2006.12.001. PMID: 17367861.
- ↑ Grob S, Charlson E, Tao JP. Tarsorrhaphy (Temporary and Permanent). Ophthalmic Plastic Surgery: Tricks of the Trade. 2020:279-281. doi:10.1055/b-0039-173373.
- ↑ 12.0 12.1 Allen, R. “Temporary bolster tarsorrhaphy.” Oculoplastics Surgery Techniques. University of Iowa Health Care. Ophthalmology and Visual Sciences Video Library.
- ↑ Allen, R. “Lateral tarsorrhaphy.” Oculoplastics Surgery Techniques. University of Iowa Health Care. Ophthalmology and Visual Sciences Video Library.
- ↑ Allen, R. “Pillar tarsorrhaphy.” Oculoplastics Surgery Techniques. University of Iowa Health Care. Ophthalmology and Visual Sciences Video Library.
- ↑ Donnenfeld ED, Perry HD, Nelson DB. Cyanoacrylate temporary tarsorrhaphy in the management of corneal epithelial defects. Ophthalmic Surg. 1991;22591- 593.
- ↑ Allen, R. “Glue tarsorrhaphy.” Oculoplastics Surgery Techniques. University of Iowa Health Care. Ophthalmology and Visual Sciences Video Library.
- ↑ Trivedi D, McCalla M, Squires Z, Parulekar M. Use of cyanoacrylate glue for temporary tarsorrhaphy in children. Ophthalmic Plast Reconstr Surg. 2014 Jan-Feb;30(1):60-3. doi: 10.1097/IOP.0000000000000011. PMID: 24398490.
- ↑ 18.0 18.1 18.2 18.3 Kasaee A, Musavi MR, Tabatabaie SZ, et al. Evaluation of efficacy and safety of botulinum toxin type A injection in patients requiring temporary tarsorrhaphy to improve corneal epithelial defects. Int J Ophthalmol. 2010;3(3):237-240. doi:10.3980/j.issn.2222-3959.2010.03.13.
- ↑ Ellis MF, Daniell M. An evaluation of the safety and efficacy of botulinum toxin type A (BOTOX) when used to produce a protective ptosis. Clin Exp Ophthalmol. 2001;29(6):394–399.
- ↑ Steiner GC, Gossman MD, Tanenbaum M. Modified tarsal pillar tarsorrhaphy. American Journal of Ophthalmology. 1993 Jul;116(1):103-104. DOI: 10.1016/s0002-9394(14)71755-6. PMID: 8328528.
- ↑ Cassel GH, Billig MD, Randall HG. The Eye Book: A Complete Guide to Eye Disorders and Health. Baltimore, MD: Johns Hopkins University Press, 1998.